Indications
Procedure Types
| Procedure Type | Purpose | Common Injectates |
|---|---|---|
| Diagnostic arthrography | MR arthrogram (labral tear, cartilage defect evaluation); CT arthrogram (when MRI contraindicated) | Dilute gadolinium (MR) or iodinated contrast (CT) |
| Aspiration | Synovial fluid analysis: cell count, crystal analysis, Gram stain, culture | N/A |
| Therapeutic — corticosteroid | Inflammatory arthritis, OA pain relief, adhesive capsulitis | Corticosteroid + local anesthetic |
| Therapeutic — viscosupplementation | Knee osteoarthritis (HA approved for knee) | Hyaluronic acid |
| Therapeutic — PRP | Emerging evidence for OA; not yet standard of care | Platelet-rich plasma (autologous) |
Indications by Joint
| Joint | Common Indications |
|---|---|
| Hip | Osteoarthritis; avascular necrosis; MR arthrography for labral tear; femoroacetabular impingement; synovitis |
| Shoulder | Osteoarthritis; adhesive capsulitis (hydrodilation); labral tear arthrography; rotator cuff-related arthropathy |
| Knee | Osteoarthritis; rheumatoid arthritis; large effusion aspiration; viscosupplementation; post-traumatic effusion |
| Ankle | Post-traumatic arthritis; inflammatory arthritis; MR arthrography for osteochondral defect; instability evaluation |
| Wrist / Elbow | Inflammatory arthritis; post-traumatic pain; arthrography for ligament/cartilage evaluation |
Contraindications
| Type | Contraindication |
|---|---|
| Absolute | Active septic arthritis (aspiration acceptable, corticosteroid injection absolutely contraindicated); overlying cellulitis or skin infection; systemic bacteremia; allergy to proposed injectate (contrast, steroid, anesthetic) |
| Relative | Uncontrolled diabetes (transient glucose elevation 24–48 h); coagulopathy; prosthetic joint (higher infection risk — requires strict sterile technique and clinical team discussion); recent corticosteroid injection at same site within 3 months |
Never inject corticosteroid into a joint you suspect may be septic. If infection is a possibility, perform aspiration for cell count, Gram stain, and culture first. Withhold steroid until infection is excluded.
Relevant Anatomy
Joint Access Summary
| Joint | Volume | Preferred Guidance | Approach | Injection Site / Landmark |
|---|---|---|---|---|
| Hip | 10–20 mL (MR arthro: 12–15 mL) | Fluoroscopy | Anterior approach | Femoral head-neck junction; confirm intra-articular with ring sign — contrast outlines femoral head circumferentially; iliopectineal bursa communicates with joint in ~15% (acceptable) |
| Shoulder | 10–15 mL | Ultrasound or fluoroscopy | Posterior (preferred) or anterior rotator interval | Posterior approach: needle tip at posterior joint capsule in the infraspinatus / teres minor interval, adjacent to glenoid labrum; real-time US fluid tracking into glenohumeral joint confirms placement |
| Knee | 5–10 mL | Ultrasound | Lateral suprapatellar (preferred) | Suprapatellar pouch visible as anechoic space between quadriceps tendon and femur; confirm with real-time fluid spread within pouch on US; effusion makes this the most straightforward joint to access |
| Ankle | 3–6 mL | Fluoroscopy or ultrasound | Anteromedial approach | Tibiotalar joint space between tibialis anterior tendon and medial malleolus; avoid anterior tibial neurovascular bundle; confirm with contrast or real-time US |
| Wrist | 1–3 mL | Fluoroscopy | Dorsal approach to radiocarpal joint | Lister's tubercle as dorsal reference landmark; radiocarpal joint just distal to radius on AP fluoroscopy; note multiple compartments — radiocarpal, midcarpal, and DRUJ each separate and may require individual injection for arthrography |
| Elbow | 3–5 mL | Fluoroscopy or ultrasound | Lateral (radiohumeral) approach | Triangle formed by lateral epicondyle, radial head, and capitellum; elbow flexed 90°; posterior fat pad elevation on lateral radiograph suggests intra-articular effusion and facilitates access |
Fluoroscopic Injection Sites
Hip
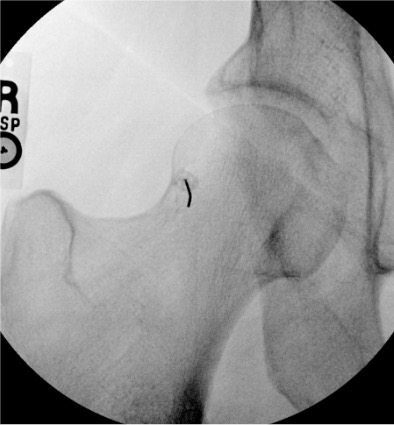
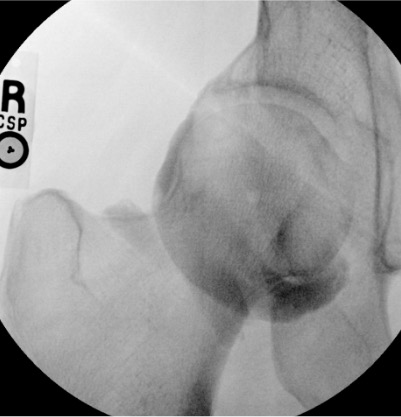
Needle placementPost-injection
Shoulder
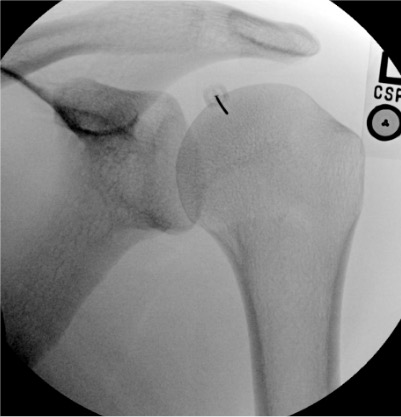
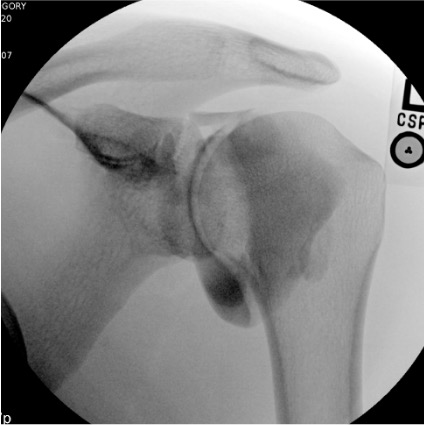
Needle placementPost-injection
Knee
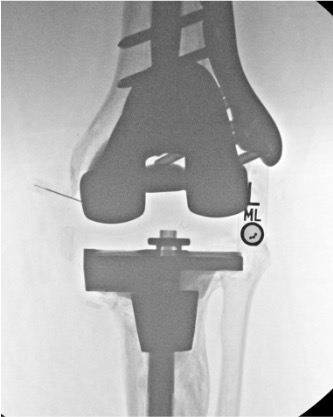
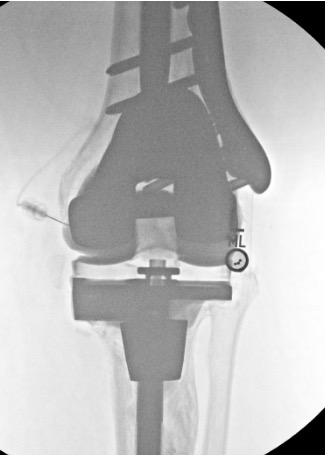
Needle placementPost-injection
Ankle
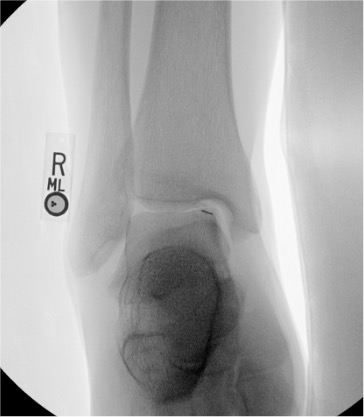
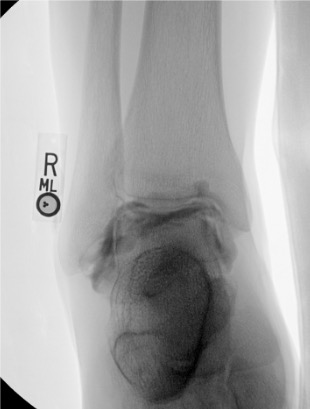
Needle placementPost-injection
Wrist
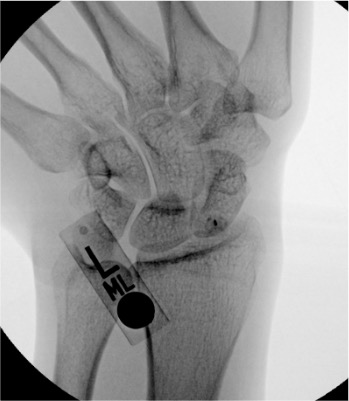
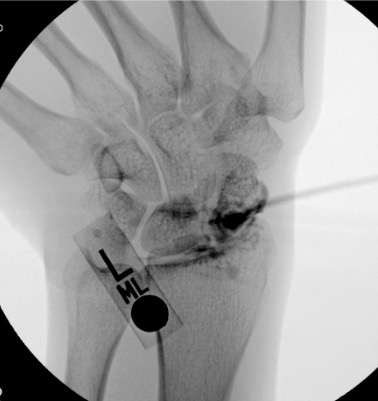
Needle placementPost-injection
Elbow
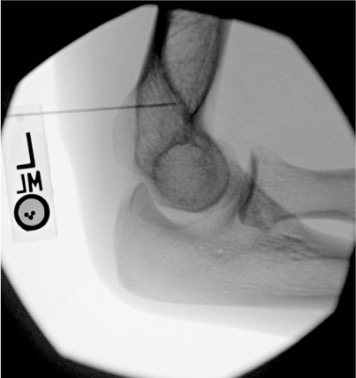
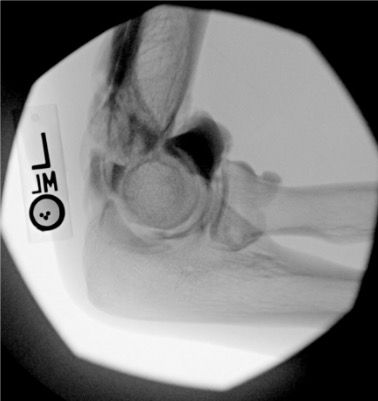
Needle placementPost-injection
Pre-Procedure Checklist
Prior Imaging Review
- Weight-bearing radiographs for all joints: establishes baseline joint space, alignment, and degree of arthritic change
- MRI or CT as available: evaluate for labral tears, cartilage defects, AVN, effusion, or prior arthroplasty hardware
Labs
- WBC, ESR, CRP if infection concern is present
- HbA1c in diabetic patients — document baseline glucose control and counsel on expected transient glucose elevation
- Coagulation profile if patient is anticoagulated (low bleeding risk procedure — holds typically not required for superficial joints)
Confirm Clinical Indication
- Therapeutic vs. diagnostic vs. combined arthrogram-injection
- Steroid vs. hyaluronic acid vs. PRP — confirm indication and insurance/formulary approval as applicable
- Confirm no prior steroid injection at this joint within 3 months
Consent Discussion Points
- Post-injection flare: 5–10% (transient pain increase 24–48 h); warn patient before discharge
- Infection / septic arthritis: <0.001%
- Transient glucose elevation in diabetics: 24–48 h
- Skin depigmentation or subcutaneous fat atrophy (steroid tracking to skin)
- Tendon rupture (from direct tendon injection — prevented by confirmation of intra-articular position)
- Cartilage injury
- Contrast allergy (for arthrography procedures)
- Steroid arthropathy with repeated injections >3–4 per year
Equipment Overview
- Imaging guidance unit: fluoroscopy C-arm (hip, ankle, wrist) or ultrasound with sterile probe cover (shoulder, knee)
- Joint access needle (size appropriate for joint — no specific sizes in this reference)
- Injectate preparation supplies: syringes, mixing needles
- Sterile prep and drape
- Specimen tubes for aspiration (cell count, crystal analysis, culture)
- Sterile occlusive dressing
Procedure Overview
- Review prior imaging and confirm procedure type and target joint
- Position patient appropriately for the target joint and guidance modality
- Image-guided needle placement: fluoroscopy (hip — anterior approach to femoral head-neck junction) or ultrasound (shoulder, knee — real-time visualization)
- Confirm intra-articular position before injecting:
- Fluoroscopy: inject small amount of iodinated contrast — "ring sign" at femoral head-neck junction for hip; contrast fills joint space outlining cartilage
- Ultrasound: real-time visualization of fluid tracking into joint space confirms intra-articular placement
- If effusion is present: aspirate fully before injection to avoid diluting injectate and to obtain diagnostic specimen
- Inject therapeutic agent after confirming intra-articular position
- Document: injection date, joint, agent administered, and patient response
- Discharge with post-injection instructions
Complications
| Complication | Rate | Recognition & Management |
|---|---|---|
| Post-injection flare | 5–10% | Transient pain increase within 24–48 h of injection. Self-resolving. Manage with NSAIDs, ice, and rest. Warn patient before discharge — this is expected in up to 1 in 10 patients and does not indicate failure of the procedure. |
| Vasovagal reaction | Uncommon | Monitor patient in supine or reclined position after procedure. IV fluids; supportive care. |
| Tendon rupture | Rare | Caused by direct intratendinous steroid injection. Prevented by confirming intra-articular position with contrast or US before injection. |
| Septic arthritis | <0.001% | Fever and severe joint pain within days to weeks post-injection. Mandatory joint aspiration for cell count, Gram stain, and culture. Broad-spectrum IV antibiotics; orthopedic or IR consultation for drainage. |
| Glucose elevation in diabetics | Common with corticosteroids | Transient elevation 24–48 h post-injection. Counsel patients to monitor home glucose and adjust insulin per their diabetes management plan. |
| Skin depigmentation / fat atrophy | Uncommon | Superficial steroid tracking from injection site. More common with superficial joints. Permanent but cosmetic only. |
| Steroid arthropathy | With repeated injections >3–4/year | Cartilage thinning and subchondral bone loss. Limit to 3–4 injections per joint per year. Document injection dates and track frequency. |
| Contrast allergy | Rare | For arthrography procedures using iodinated contrast. Confirm allergy history; pre-medicate if prior mild reaction. Anaphylaxis: epinephrine and emergency management per standard protocol. |
Post-Procedure Care
- Rest the injected joint for 24 hours; avoid strenuous weight-bearing activity for 24–48 hours
- Ice application for post-injection flare (20 minutes on, 20 minutes off)
- Diabetic patients: monitor home glucose for 24–48 hours; contact prescribing physician for glucose management if needed
- Follow-up at 4–6 weeks to assess treatment response and determine need for repeat injection or alternative management
- Document injection date, joint, agent, and response in the patient's record for steroid frequency tracking
When to Escalate
- Fever and worsening joint pain within days to weeks post-injection: suspect septic arthritis — urgent joint aspiration with cell count and culture; orthopedic or IR consultation; IV antibiotics; surgical or IR drainage if confirmed
- Progressive joint destruction on follow-up imaging: orthopedic surgery referral for arthroplasty evaluation
- Failure to respond to repeat injections: reassess diagnosis; consider MRI; orthopedic consultation for surgical options