Indications
| Type | Clinical Context |
|---|---|
| Diagnostic | New-onset ascites of unclear etiology; suspected spontaneous bacterial peritonitis (SBP); unexplained clinical deterioration in a patient with known ascites |
| Therapeutic | Symptomatic tense ascites causing dyspnea, abdominal pain, or early satiety; ascites refractory to diuretics |
Contraindications
| Type | Contraindication |
|---|---|
| Absolute | Hemodynamic instability; clinically evident fibrinolysis or DIC; no safe ultrasound access window |
| Relative | Overlying skin infection or cellulitis at access site; prior abdominal surgery with suspected tethered bowel; pregnancy (use ultrasound guidance, avoid gravid uterus); massive organomegaly encroaching on access site |
Coagulopathy in cirrhosis: Mild-to-moderate coagulopathy and thrombocytopenia are common in cirrhotic patients and are generally not contraindications to paracentesis. The INR does not reliably reflect bleeding risk in liver disease — these patients have a rebalanced hemostatic system. Routine laboratory correction is not required per SIR guidelines.
Relevant Anatomy
Access Site
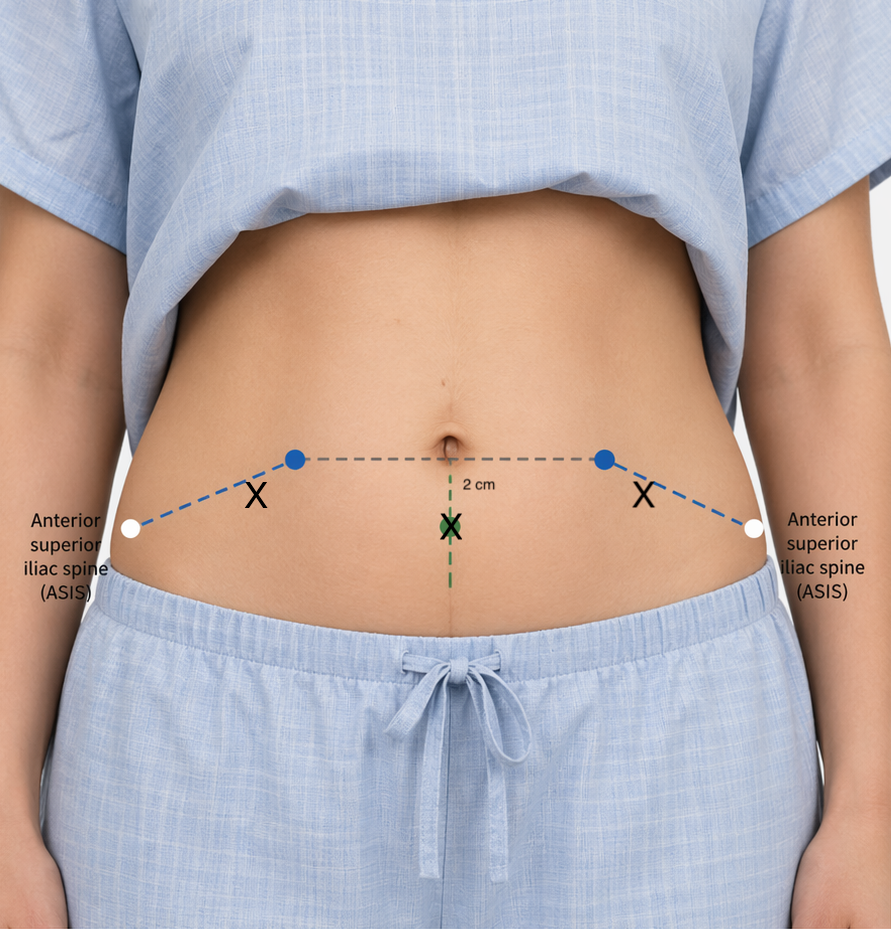
The preferred access site is the left or right lower quadrant, approximately 3 cm medial and 3 cm superior to the anterior superior iliac spine (ASIS), lateral to the rectus sheath. This approach avoids the inferior epigastric artery, which runs deep within the rectus abdominis muscle.
A midline infraumbilical approach through the avascular linea alba is an alternative when lateral access is not feasible. Final access site is always determined by real-time ultrasound — the anatomic landmark guides initial probe placement only.
Structures at Risk
- Inferior epigastric artery — runs within the rectus sheath; avoid by accessing lateral to the rectus (confirmed with color Doppler)
- Distended bladder — patient should void or have a urinary catheter placed prior to low midline access
- Tethered bowel — surgical scars may cause bowel adhesion to the anterior abdominal wall; ultrasound confirmation of a clear window is essential
- Splenomegaly / hepatomegaly — confirm organ margins on ultrasound before committing to LLQ or RLQ access
Pre-Procedure Checklist
Imaging Review
- Confirm ascites volume and distribution on prior cross-sectional imaging or bedside ultrasound
- Identify largest accessible fluid pocket; note any loculations
- Document prior abdominal surgical scars (risk of tethered bowel at access site)
Labs
- SIR Category 1 — routine labs not required for simple paracentesis in stable patients
- Check INR and platelet count only if there is a known bleeding diathesis, clinically suspected DIC, or severe liver dysfunction beyond baseline cirrhosis
- If checked: target INR <2.0, platelets >50,000/µL (though correction rarely needed before this procedure)
Anticoagulation Management
- Hold anticoagulation when medically appropriate prior to procedure
- Heparin infusion: hold 4–6 hours; LMWH: hold 24 hours; warfarin: hold 5 days; direct oral anticoagulants (DOACs): hold 24–48 hours depending on agent and renal function
- Antiplatelet agents (except aspirin): hold per institutional protocol
Consent Considerations
Discuss the following risks with the patient: bleeding (including vessel injury), infection, bowel perforation, persistent ascites leak at the puncture site, and post-paracentesis circulatory dysfunction (PPCD) — particularly relevant for large-volume drainage without albumin replacement.
Patient Preparation
- Patient voids prior to procedure (or urinary catheter placed if unable)
- IV access required for large-volume (therapeutic) paracentesis — needed for albumin infusion
- Plan which fluid analysis tests to send before starting (see Fluid Analysis section below)
Equipment Overview
The following categories of equipment are used. Specific device selection, sizes, and configuration are part of the detailed procedural approach.
- Imaging guidance: Ultrasound with sterile probe cover and gel
- Access: Paracentesis needle-catheter kit (sheathed single-step or modified Seldinger)
- Local anesthesia: Local anesthetic agent and needle
- Sterile preparation: Antiseptic solution, sterile drape, sterile gloves
- Fluid collection (diagnostic): Syringes and specimen tubes
- Fluid collection (therapeutic): Vacuum drainage bottles and connecting tubing
Procedure Overview
The following is a high-level summary of the procedural steps. Full step-by-step technique, equipment setup, and periprocedural management are available in RadCall Pro.
- Ultrasound survey — identify the largest accessible fluid pocket, confirm no bowel or vessels in the needle path, mark the skin entry site, and measure depth to fluid
- Sterile preparation — antiseptic prep and draping of the access site
- Local anesthesia — anesthetize skin and soft tissue down to the peritoneum under ultrasound guidance
- Needle access — advance the access needle into the peritoneal fluid under real-time ultrasound guidance and confirm fluid return
- Specimen collection — aspirate fluid for diagnostic labs (see Fluid Analysis below); for cytology, adequate volume is needed
- Therapeutic drainage — connect catheter to vacuum drainage system; drain until patient is comfortable or target volume reached
- Removal and dressing — remove catheter, apply sterile dressing; document fluid color, clarity, and total volume removed
Complications
| Complication | Rate | Recognition & Management |
|---|---|---|
| Ascites leak at puncture site | ~5% | Most common complication; typically self-limited; wound closure techniques or ostomy bag as temporizing measure; optimize technique to reduce tract patency |
| Abdominal wall hematoma / bleeding | <1% | Vessel injury (inferior epigastric artery); recognizable on CT as hyperdense collection in abdominal wall; conservative management vs. embolization for large or expanding hematomas |
| Bowel perforation | <1% | Typically self-sealing with small-caliber needle; monitor for signs of peritonitis (fever, worsening abdominal pain, leukocytosis); CT abdomen if perforation suspected |
| Post-paracentesis circulatory dysfunction (PPCD) | Up to 20% without albumin | Hypovolemia, hyponatremia, and renal impairment occurring 12–72 hours after large-volume drainage; prevented by albumin replacement (6–8 g per liter removed in cirrhotic patients) |
| Infection | Rare | Cellulitis or peritonitis from contamination; strict sterile technique is preventive |
Post-Procedure Care & Imaging
Monitoring
- Vital signs every 30 minutes for 1 hour post-procedure (especially after large-volume drainage)
- Monitor puncture site for bleeding, expanding hematoma, or persistent fluid leak
- No routine post-procedure imaging required for uncomplicated paracentesis
Albumin Replacement
- For >5 L removed in cirrhotic patients: administer 25% human albumin at 6–8 g per liter of ascitic fluid removed
- Begin albumin infusion during or immediately after drainage — do not delay
- Albumin replacement significantly reduces PPCD, hyponatremia, and 30-day mortality in this population
Imaging Findings to Report
- If follow-up CT or ultrasound is obtained: evaluate for residual or recurrent ascites, new abdominal wall hematoma, pneumoperitoneum (bowel perforation), or peritoneal free air
- Pneumoperitoneum after paracentesis is uncommon — if seen, correlate with clinical picture and consider bowel perforation
Ascitic Fluid Analysis Reference
| Test | Threshold | Interpretation |
|---|---|---|
| SAAG (serum albumin − ascites albumin) | ≥ 1.1 g/dL | Portal hypertension (cirrhosis, heart failure, Budd-Chiari, portal vein thrombosis) — 97% accuracy |
| SAAG | < 1.1 g/dL | Non-portal hypertensive etiology: malignancy, TB peritonitis, pancreatitis, nephrotic syndrome |
| Cell count — PMN | ≥ 250/mm³ | Spontaneous bacterial peritonitis (SBP) — begin empiric antibiotics immediately (e.g., cefotaxime) |
| Total protein | ≥ 2.5 g/dL | Exudate: malignancy, TB, pancreatitis; <2.5 g/dL = transudate (cirrhosis, heart failure) |
| Glucose | Lower than serum | Markedly low or undetectable → suspect bowel perforation or infection |
| Amylase | Elevated vs. serum | Pancreatic ascites or bowel perforation |
| Bilirubin | Ascites > serum | Biliary or bowel perforation |
| Triglycerides | > 200 mg/dL | Chylous ascites: lymphatic obstruction, malignancy, post-surgical |
| Cytology | — | Malignant cells; sensitivity ~60–75%; adequate volume needed for cellularity |
| Gram stain / culture | — | Inoculate blood culture bottles at bedside — increases SBP culture yield 50–70% vs. standard tubes |
Fluid Appearance
- Clear / straw-colored: Typical transudative ascites (cirrhosis, heart failure)
- Turbid / cloudy: Infection (SBP) or elevated cell count
- Milky / opalescent: Chylous ascites — check triglycerides
- Pink / bloody: Traumatic tap, malignancy, or hemorrhagic ascites — send fluid hematocrit
- Brown: Elevated bilirubin — biliary leak or bowel perforation
When to Escalate
- Persistent bright-red bloody aspirate that does not clear — stop the procedure; monitor hemodynamics; obtain CT abdomen if vessel injury suspected; interventional or surgical consultation if hematoma is expanding
- Signs of bowel perforation post-procedure (new fever, worsening peritoneal signs, leukocytosis) — CT abdomen/pelvis; surgical consultation
- Hemodynamic instability during or after procedure — IV fluid resuscitation; hold further drainage; urgent medical evaluation
- SBP on fluid analysis (PMN ≥ 250/mm³) — notify the primary team immediately for empiric antibiotic initiation; do not wait for culture results
References
- Runyon BA; AASLD Practice Guideline Committee. Management of adult patients with ascites due to cirrhosis. Hepatology. 2013;57(4):1651–1653.
- European Association for the Study of the Liver (EASL). EASL Clinical Practice Guidelines for decompensated cirrhosis. J Hepatol. 2018;69(2):406–460.
- Patel IJ et al. Society of Interventional Radiology Consensus Guidelines for the Periprocedural Management of Thrombotic and Bleeding Risk in Patients Undergoing Percutaneous Image-Guided Interventions. J Vasc Interv Radiol. 2019;30(8):1168–1184.