Indications
BCLC Staging & Treatment — 2026
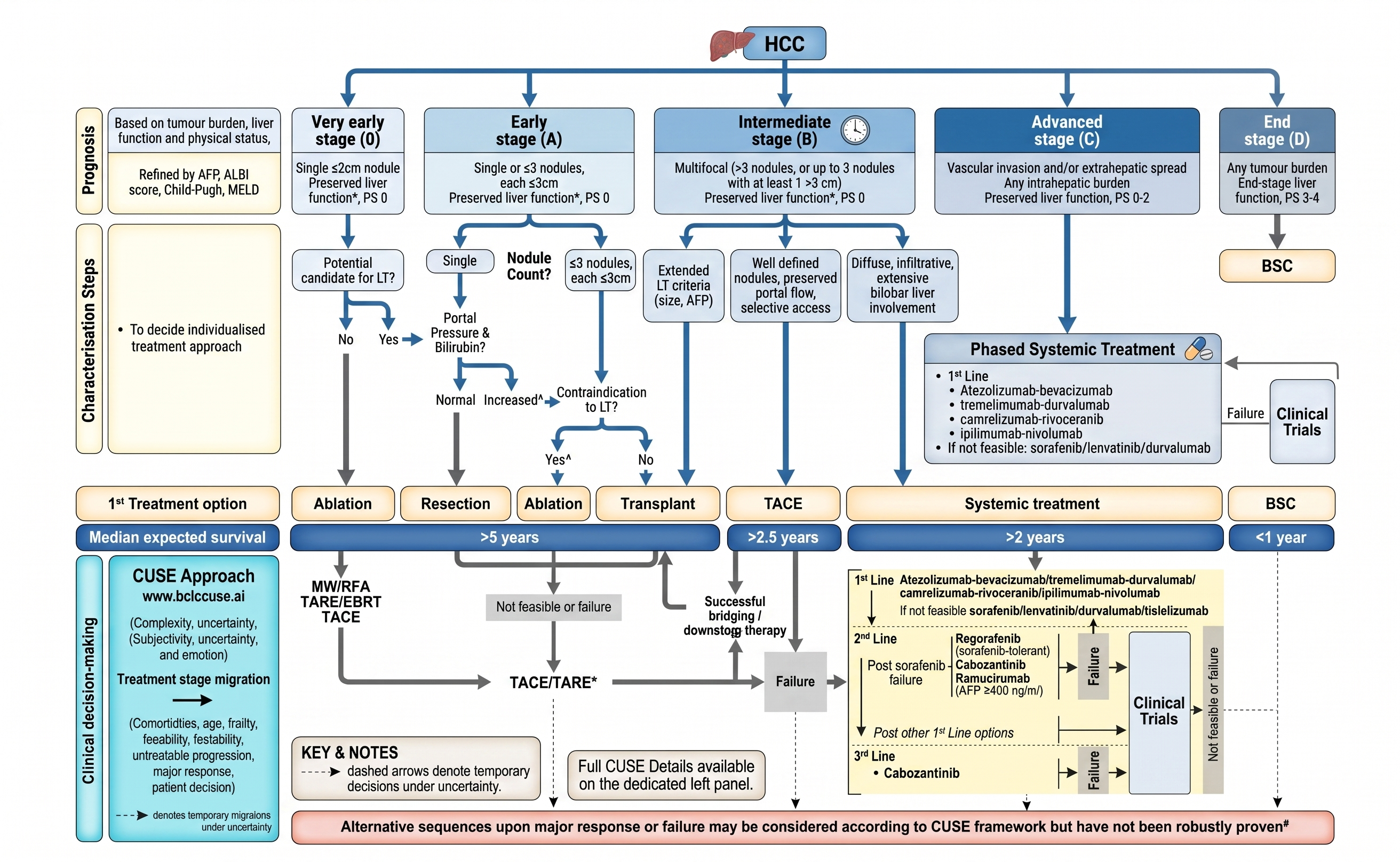
| Indication | Clinical Context |
|---|---|
| HCC — unresectable, BCLC Stage B (intermediate) | Standard of care per EASL/AASLD guidelines; multinodular disease without vascular invasion or extrahepatic spread; Child-Pugh A or selected B7 patients |
| Bridge to liver transplantation | Prevent HCC dropout from transplant waitlist; maintain tumor within Milan criteria while awaiting organ; TACE is effective and does not preclude subsequent transplant |
| Downstaging for transplant eligibility | Reduce tumor burden to within transplant criteria; coordinate with hepatology and transplant surgery team |
| Residual HCC after ablation | Salvage TACE for incompletely treated tumors after radiofrequency or microwave ablation |
| Hepatic metastases (selected) | Neuroendocrine tumors (NET) are most responsive; colorectal metastases less so; discuss at multidisciplinary tumor board |
Contraindications
| Type | Contraindication |
|---|---|
| Absolute | Decompensated cirrhosis (Child-Pugh C); bilirubin >3 mg/dL; INR >2; hepatic encephalopathy; ECOG performance status >2; tumor replacement >50% of liver volume; complete biliary obstruction without external drainage |
| Relative | Extensive main portal vein thrombosis (segmental/branch PVT — proceed with caution and multidisciplinary discussion); prior bilio-enteric anastomosis (greatly elevated cholangitis risk — prophylactic antibiotics mandatory); Child-Pugh B8–9 (very high risk; extreme caution and case-by-case multidisciplinary decision) |
Multidisciplinary tumor board review is strongly recommended before every TACE session. Child-Pugh score must be recalculated before each treatment — liver function can change significantly between sessions. Sorafenib or other systemic therapy should be discussed for patients progressing beyond TACE-amenable disease.
Relevant Anatomy
Hepatic Arterial Supply and HCC Vascularity
Normal hepatic blood supply: celiac trunk → common hepatic artery → proper hepatic artery → right and left hepatic arteries at the porta hepatis. Hepatic arterial anatomy varies in approximately 25% of patients — the most common variant is a replaced right hepatic artery arising from the superior mesenteric artery (~15%); replaced left hepatic from the left gastric artery (~5%). A complete celiac angiogram and superior mesenteric angiogram are essential to identify all tumor-feeding vessels before treatment.
HCC is hypervascular — the tumor receives ~90–100% of its blood supply from hepatic arteries, compared to the normal liver where 75% comes from the portal vein. This differential vascularity allows superselective targeting of tumor-feeding vessels with embolic material while relatively sparing the surrounding normal parenchyma, which has dual blood supply.
Non-Target Embolization Risks
Critical structures at risk for non-target embolization include the right gastric artery (lesser curvature of stomach — gastric ulceration), cystic artery (cholecystitis), falciform artery (anterior abdominal wall injury), and phrenic artery contributions (which can supply large dome lesions). Superselective catheterization of the tumor-feeding segmental or subsegmental vessel minimizes exposure of non-target structures. Stasis is the angiographic endpoint — particles can reflux beyond stasis into non-target vessels if injection is continued past this point.
Pre-Procedure Checklist
Imaging Review
- Contrast-enhanced CT or MRI liver within 4 weeks — assess tumor size, number, vascularity, and LI-RADS characterization; confirm BCLC staging; assess portal vein patency and hepatic vein involvement
- Calculate Child-Pugh score before every TACE session — this should be a standing requirement, not assumed from prior visit
Labs
- CBC, comprehensive metabolic panel (bilirubin, LFTs, creatinine), coagulation panel, AFP
- Portal vein patency confirmation — Doppler US or cross-sectional imaging; main PVT dramatically increases ischemia risk
- Pre-procedure IV hydration for contrast and chemotherapy nephroprotection
Consent Considerations
Discuss: post-embolization syndrome (majority of patients — fever, RUQ pain, nausea for 24–72 hours), hepatic failure or decompensation, cholecystitis (if cystic artery at risk), biliary injury (higher risk with prior ERCP stent or bilio-enteric anastomosis), gastric ulceration from non-target embolization, treatment failure or progression, contrast nephropathy, access-site complications, and the need for multiple sessions.
Procedure Overview
The following is a high-level summary. Full step-by-step technique, cTACE vs. DEB-TACE selection, and stasis endpoint assessment are available in RadCall Pro.
- Vascular access — common femoral artery access (standard) or radial access; systemic heparinization after sheath placement
- Diagnostic arteriogram — celiac angiography to map hepatic arterial anatomy and identify variants; superior mesenteric angiography to identify replaced hepatic artery branches; identify all tumor-feeding vessels
- Selective hepatic angiography — advance catheter to right or left hepatic artery; confirm tumor hypervascularity (blush) and identify dominant tumor-feeding segmental or subsegmental vessels
- Superselective catheterization — advance microcatheter to the segmental or subsegmental tumor-feeding artery; superselective positioning minimizes non-target liver damage and reduces post-embolization syndrome severity
- Embolization — deliver cTACE (lipiodol-chemotherapy emulsion followed by bland embolic) or DEB-TACE (drug-eluting beads loaded with doxorubicin); endpoint is near-stasis with cessation of antegrade tumor-feeding flow; do not continue embolization beyond stasis
- Completion angiography — confirm stasis; assess for non-target embolization; document final angiographic result; plan contralateral or additional segmental treatment as appropriate
Complications
| Complication | Rate | Recognition & Management |
|---|---|---|
| Post-embolization syndrome | Majority; expected | Fever, RUQ pain, nausea/vomiting within 24–48 hours; normal response to tumor ischemia; scheduled NSAIDs, antiemetics, analgesics; distinguish from cholangitis (persistent fever, RUQ pain with jaundice) or hepatic failure (bilirubin rise, encephalopathy) |
| Hepatic failure / decompensation | ~3–5% (higher with marginal function) | Rising bilirubin, coagulopathy, encephalopathy post-TACE; aggressive supportive care; hepatology consultation; liver transplant evaluation for acute-on-chronic liver failure; prevented by careful Child-Pugh selection |
| Cholecystitis | ~5% (if cystic artery in treatment field) | RUQ pain, fever, leukocytosis; CT confirms gallbladder edema or wall thickening; cholecystostomy for severe cases; prevented by identifying and protecting the cystic artery |
| Biliary injury / biloma | Higher with biliary stent or anastomosis | Biliary necrosis from hepatic artery chemoembolization; prophylactic antibiotics for at-risk patients; ERCP or percutaneous drainage if biloma develops |
| Non-target embolization | Rare with superselective technique | Gastric ulceration, abdominal wall injury; prevented by superselective microcatheter positioning and stopping at stasis endpoint |
| Access-site complications | ~1–2% hematoma, pseudoaneurysm | Standard femoral access complications; ultrasound-guided thrombin injection for pseudoaneurysm; compression for hematoma |
Post-Procedure Care
Monitoring
- Vital signs every 4 hours; temperature monitoring for post-embolization syndrome and cholangitis
- Liver function tests at 24 hours and at 1 week post-procedure — track bilirubin trend
- Pain management: scheduled NSAIDs and opioids as needed for post-embolization pelvic pain; antiemetics for nausea
- Fever within 48 hours — expected; persistent fever >48–72 hours or fever with jaundice/RUQ tenderness suggests cholangitis; blood cultures and CT evaluation
Response Assessment
- mRECIST criteria at 4–6 weeks on contrast-enhanced MRI or CT — viable tumor is defined by arterial phase enhancement; necrosis (no enhancement) indicates treatment response; size reduction is secondary
- Complete response (CR): no arterial enhancement in treated lesions; partial response (PR): ≥30% decrease in sum of viable tumor diameters; stable disease (SD) or progressive disease (PD) per mRECIST definitions
- AFP response (if elevated at baseline) can supplement imaging assessment; rising AFP with imaging response may indicate new lesions
- Repeat TACE sessions are planned based on response — viable tumor or untreated lesions identified at follow-up imaging
When to Escalate
- Hepatic decompensation post-TACE (rising bilirubin, encephalopathy) — hepatology consultation; aggressive supportive care; liver transplant evaluation for acute-on-chronic liver failure; reconsider future TACE candidacy
- Suspected cholangitis (persistent fever, jaundice, RUQ pain) — blood cultures; IV antibiotics; CT for biliary assessment; ERCP or percutaneous biliary drainage for obstruction
- Tumor progression on mRECIST or failure to achieve response — multidisciplinary tumor board discussion; options include systemic therapy (sorafenib, lenvatinib), Y-90 radioembolization, ablation for small residual lesions, or clinical trial enrollment
- TACE-refractory disease — defined as lack of response after 2 properly performed TACE sessions; escalate to systemic therapy discussion with oncology and hepatology
Evidence Base for Liver-Directed Therapy in HCC
The evidence informing the choice of locoregional therapy in HCC spans multiple RCTs, meta-analyses, and registry studies. The following is an organized summary by modality, relevant when counseling patients or participating in multidisciplinary tumor board discussions.
1. TACE vs. Best Supportive Care — Foundational Evidence
TACE became the standard of care for intermediate-stage (BCLC B) HCC based on two landmark RCTs (Llovet 2002, Lo 2002) and a subsequent meta-analysis demonstrating improved overall survival compared with best supportive care. A systematic review of 101 studies (12,372 patients) reported an ORR of 52.5% and median survival of 19.4 months with conventional TACE. Median OS with TACE ranges from 16–40 months depending on patient selection. The NCCN and AASLD guidelines both endorse TACE as a primary locoregional option for unresectable HCC.[1,2,4]
2. cTACE vs. DEB-TACE vs. Bland Embolization (TAE)
- PRECISION V trial (212 patients, advanced HCC): DEB-TACE showed higher rates of complete response, objective response, and disease control vs. conventional TACE, but was not statistically superior (P = .11). Doxorubicin-related side effects were higher with conventional TACE.[3]
- Multiple RCTs and meta-analyses have shown similar responses and safety profiles between cTACE and DEB-TACE, with no consistent superiority of either approach.[2]
- A meta-analysis of RCTs comparing TACE (both types) to bland TAE also did not demonstrate superiority of chemoembolization over bland embolization — the embolic effect itself may be the primary mechanism.[3,5]
3. TARE — Y-90 Radioembolization
| Trial | Design | Key Result |
|---|---|---|
| LEGACY[6] | Single-arm, 162 pts, CTP-A, solitary HCC ≤8 cm | ORR 88.3% (mRECIST); DoR ≥6 mo in 76.1%; 3-yr OS 86.6% with radiation segmentectomy approach → FDA approval of Y-90 glass microspheres (2021) |
| TRACE[7] | Phase II RCT, 72 pts, BCLC A–B; Y-90 glass vs. DEB-TACE | TARE: TTP 17.1 vs. 9.5 mo (HR 0.36); OS 30.2 vs. 15.6 mo (HR 0.48). Trial terminated early after meeting primary endpoint |
| SARAH[8] | Phase III RCT, 459 pts, advanced HCC; Y-90 resin vs. sorafenib | No significant OS difference (8.0 vs. 9.9 mo; HR 0.86, P = .18). Better safety profile with TARE. Post-hoc: patients receiving ≥100 Gy had OS 14.1 vs. 6.1 mo — dosimetry matters |
| SIRveNIB[9] | Phase III RCT, Asia-Pacific, advanced HCC; Y-90 resin vs. sorafenib | No OS difference despite greater tumor response with TARE — consistent with SARAH findings |
| DOSISPHERE-01[2] | RCT; personalized dosimetry (>205 Gy to tumor) vs. standard dosimetry | Personalized: ORR 76.6% vs. 22.2%; downstaging to surgery 35% vs. 3.5%; OS 26.6 vs. 10.7 mo. Dosimetry optimization is critical — standard dosimetry underdelivers |
TARE vs. TACE — pooled data: A 2025 meta-analysis (6 studies, ~443 patients) found TARE significantly reduced the hazard of death compared to TACE (HR 0.68, 95% CI 0.55–0.86) with superior PFS (HR 0.54) and no increase in severe toxicity.[10] However, a 2025 multicenter retrospective study (279 patients) found TACE outperformed TARE in early-stage disease (mOS 60 vs. 25 months for BCLC 0/A), underscoring the importance of patient selection.[11]
4. TARE as Bridge to Transplant
Two large 2026 studies support TARE over TACE in the bridge-to-transplant setting:
- UNOS database study (5,677 patients, 2026): TARE as first locoregional therapy was associated with a 22% decreased hazard of waitlist dropout vs. TACE (HR 0.78), higher complete necrosis on explant (35.3% vs. 20.2%), and fewer treatment sessions required.[12]
- Meta-analysis (10,661 patients, 2026): TARE required fewer sessions, had lower grade 3/4 bilirubin toxicity, higher complete necrosis rates, and superior recurrence-free survival compared to TACE for bridging and downstaging.[13]
5. Ablation (RFA/MWA) vs. Resection
| Study | Design | Finding |
|---|---|---|
| SURF trial[14] | Phase III RCT, 302 pts, Japan, HCC ≤3 cm; surgery vs. RFA | No significant difference in 5-yr OS (74.6% vs. 70.4%; HR 0.96) or RFS (42.9% vs. 42.7%). 90% solitary tumor, ~65% ≤2 cm |
| Chen 2006, Huang 2010[15] | Earlier RCTs (lower risk of bias) | Resection more effective than RFA for OS (HR 0.56) and 2-yr survival |
| 2026 meta-analysis[16] | 25 studies, 10,322 patients | Resection: superior 3- and 5-yr OS and RFS overall; for tumors ≤2 cm, outcomes comparable between modalities |
The AASLD recommends an ablation-first strategy may be considered for tumors <3 cm with good hepatic reserve, particularly in patients with significant surgical risk.[2]
6. Radiation Therapy and HAIC vs. TACE
A 2024 systematic review of 40 RCTs (11,576 patients) established a hierarchical efficacy structure for locoregional therapies:[17]
Efficacy hierarchy (PFS and OS, network meta-analysis):
Surgery + adjuvant therapy > surgery alone > RT ≈ HAIC > TACE ≈ TARE ≈ TAE ≈ TKI monotherapy
RT vs. TACE: HR 0.35 (PFS and OS). HAIC vs. TACE: HR 0.57 (PFS), 0.58 (OS).
7. TACE + Systemic Therapy Combinations
| Trial | Regimen | Result |
|---|---|---|
| TACTICS[18] | TACE + sorafenib vs. TACE alone (phase II RCT, Japan) | PFS improved (22.8 vs. 13.5 mo; HR 0.66); OS not significantly different (36.2 vs. 30.8 mo), likely confounded by high crossover in control arm (76.3%) |
| EMERALD-1[19] | TACE + durvalumab ± bevacizumab vs. TACE + placebo (phase III RCT) | Improved PFS vs. TACE + placebo — first positive phase III trial of immunotherapy combined with TACE |
| CHANCE2005/CARES-005[20] | TACE + camrelizumab + rivoceranib vs. TACE alone (phase II RCT, 200 pts) | PFS 10.8 vs. 3.2 mo (HR 0.34) — significant benefit with dual systemic blockade added to TACE |
Key takeaways for MDT discussion: The choice of locoregional modality depends on extent/location of disease, hepatic reserve, and institutional capabilities — no single modality is universally preferred (NCCN). Critical emerging themes include: personalized dosimetry for TARE (DOSISPHERE-01 fundamentally changed dose targets); TARE over TACE for bridge-to-transplant (UNOS 2026, meta-analysis 2026); and combined locoregional + immunotherapy as the new frontier (EMERALD-1, CHANCE2005). Y-90 is not superior to sorafenib for advanced HCC in phase III trials (SARAH, SIRveNIB), but TARE remains appropriate in selected patients including those with segmental/lobar portal vein thrombosis.[4]
References
- Villanueva A. Hepatocellular Carcinoma. N Engl J Med. 2019;380(15):1450–1462.
- Singal AG, Llovet JM, Yarchoan M, et al. AASLD Practice Guidance on Prevention, Diagnosis, and Treatment of Hepatocellular Carcinoma. Hepatology. 2023;78(6):1922–1965.
- Thornton LM, Abi-Jaoudeh N, Lim HJ, et al. Combination and Optimal Sequencing of Systemic and Locoregional Therapies in Hepatocellular Carcinoma: Proceedings From the SIR Foundation Research Consensus Panel. J Vasc Interv Radiol. 2024;35(6):818–824.
- National Comprehensive Cancer Network. Hepatocellular Carcinoma. Updated 2026-03-10.
- Katsanos K, Kitrou P, Spiliopoulos S, et al. Comparative Effectiveness of Different Transarterial Embolization Therapies for Unresectable HCC: A Network Meta-Analysis. PloS One. 2017;12(9):e0184597.
- Salem R, Johnson GE, Kim E, et al. Yttrium-90 Radioembolization for the Treatment of Solitary, Unresectable HCC: The LEGACY Study. Hepatology. 2021;74(5):2342–2352.
- Dhondt E, Lambert B, Hermie L, et al. Y-90 Radioembolization Versus Drug-Eluting Bead Chemoembolization for Unresectable HCC: Results From the TRACE Phase II RCT. Radiology. 2022;303(3):699–710.
- Vilgrain V, Pereira H, Assenat E, et al. Efficacy and Safety of Selective Internal Radiotherapy With Y-90 Resin Microspheres Compared With Sorafenib in Locally Advanced HCC (SARAH): Phase 3 Trial. Lancet Oncol. 2017;18(12):1624–1636.
- Walton M, Wade R, Claxton L, et al. Selective Internal Radiation Therapies for Unresectable HCC: Systematic Review, Network Meta-Analysis and Economic Evaluation. Health Technol Assess. 2020;24(48):1–264.
- de Alcântara JPTL, Götz GWXDR. TARE With Y-90 and SIRT Versus Conventional TACE for HCC: A Systematic Review and Meta-Analysis. Acad Radiol. 2025;32(11):6739–6750.
- Sanai FM, Alzanbagi A, Arabi M, et al. TACE Outperforms Radioembolization in Early- and Intermediate-Stage HCC: A Multicenter Retrospective Study. Cancers. 2025;17(13):2254.
- Kim NG, Yao FY, Kwong AJ, Mehta N. Y-90 Radioembolization Is Associated With a Lower Risk of Liver Transplant Waitlist Dropout Than Chemoembolization in HCC. J Hepatol. 2026;S0168-8278(26)00020-6.
- Xie M, Zhen Y. Efficacy of TARE and TACE as Downstaging or Bridging Strategies for HCC Before Liver Transplantation: A Systematic Review and Meta-Analysis. Cardiovasc Intervent Radiol. 2026.
- Kawaguchi Y, Hasegawa K, Kashiwabara K, et al. Surgery Versus Ablation for HCC: SURF-RCT Trial. J Clin Oncol. 2025;JCO2402030.
- Heimbach JK, Kulik LM, Finn RS, et al. AASLD Guidelines for the Treatment of Hepatocellular Carcinoma. Hepatology. 2018;67(1):358–380.
- Dai LA, Sun M, Li T, Wei D, Zou RC. Comparison of Surgical Resection and RFA for Small HCC (≤3 cm): An Updated Meta-Analysis. Syst Rev. 2026.
- Patel KR, Menon H, Patel RR, et al. Locoregional Therapies for Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. JAMA Netw Open. 2024;7(11):e2447995.
- Kudo M, Ueshima K, Ikeda M, et al. TACTICS: Final OS Data From a Phase II Trial of TACE + Sorafenib vs. TACE Alone. J Clin Oncol. 2021;39(Suppl 3):270.
- Sangro B, Kudo M, Erinjeri JP, et al. Durvalumab With or Without Bevacizumab With TACE in HCC (EMERALD-1): Phase 3 Study. Lancet. 2025;405(10474):216–232.
- Zhu HD, Fan WJ, Zhao C, et al. TACE Combined With Camrelizumab and Rivoceranib for Unresectable HCC (CHANCE2005/CARES-005): Phase II Trial. J Clin Oncol. 2026;44(11):959–969.