PAD Classification
Rutherford Classification
| Category | Clinical Description | Notes |
|---|---|---|
| 0 | Asymptomatic | ABI often normal or mildly reduced; atherosclerosis present on imaging |
| 1 | Mild claudication | Walk >200 m; symptoms reproducible with exertion |
| 2 | Moderate claudication | Walk 50–200 m before symptoms |
| 3 | Severe claudication | Walk <50 m; significant functional limitation |
| 4 | Ischemic rest pain | ABI typically <0.4; pain worst at night, relieved by dependency; CLI begins here |
| 5 | Minor tissue loss | Focal ulceration or gangrene not exceeding toes; CLI requiring urgent revascularization |
| 6 | Major tissue loss | Tissue loss above the transmetatarsal level; limb threatened; urgent revascularization or amputation planning |
Categories 4–6 = Critical Limb Ischemia (CLI) — requires urgent revascularization to prevent major limb loss. ABI <0.4 or absolute ankle pressure <50 mmHg supports CLI diagnosis.
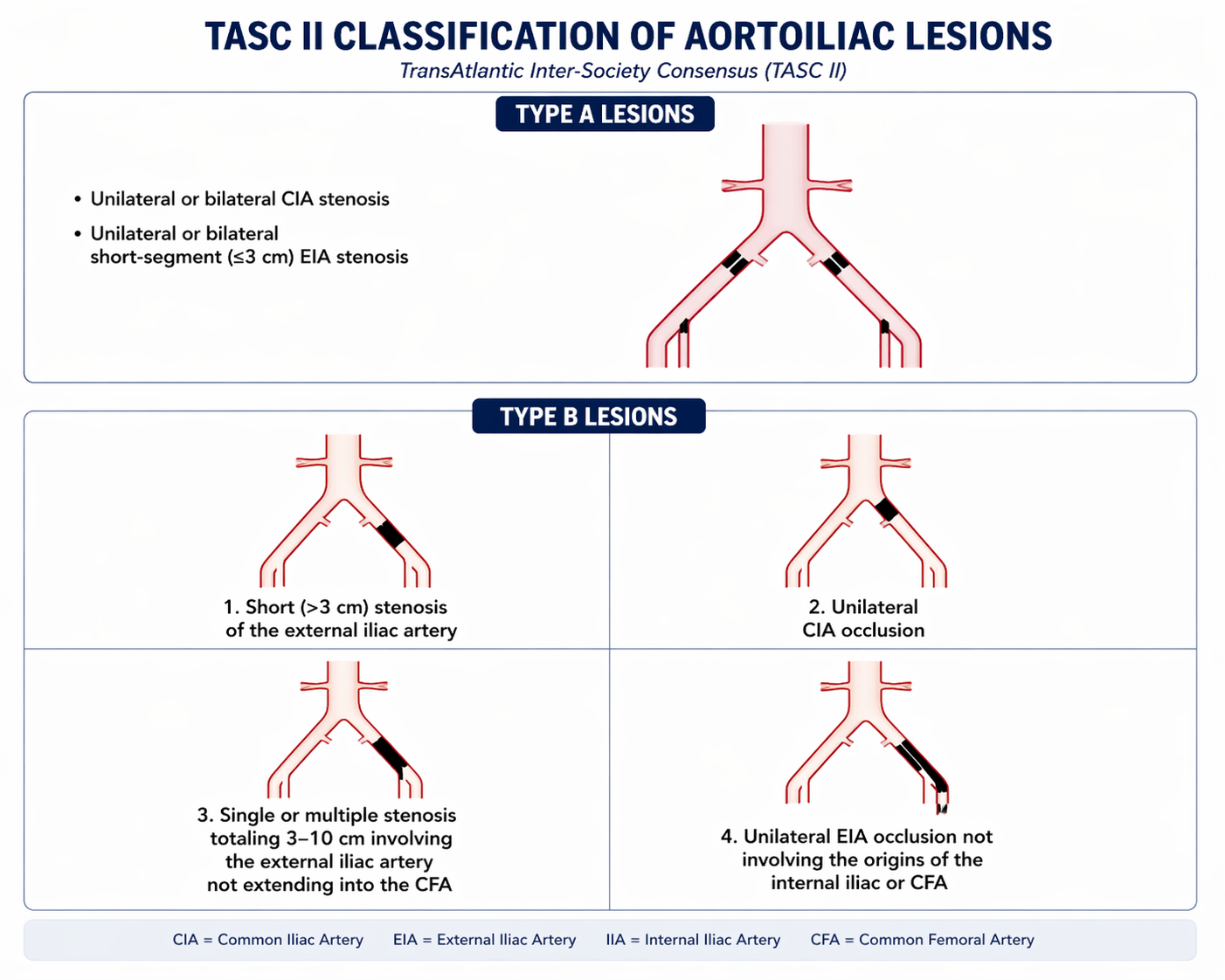
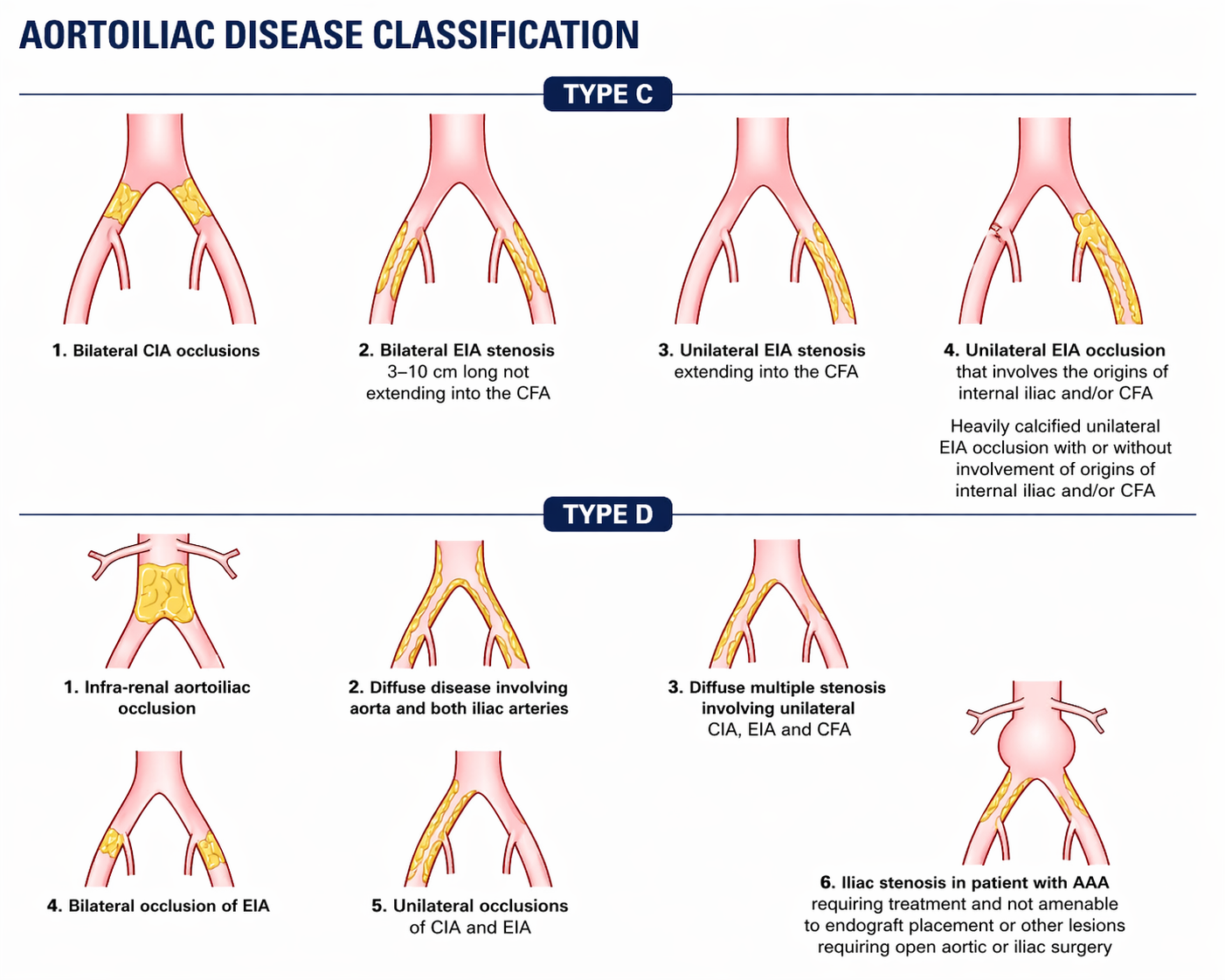
TASC II Classification (SFA/Popliteal Example)
| Type | Lesion Characteristics | Preferred Strategy |
|---|---|---|
| A | Single stenosis ≤10 cm; single occlusion ≤5 cm | Endovascular first-line |
| B | Multiple stenoses/occlusions each ≤5 cm; single stenosis or occlusion 5–15 cm not involving the popliteal; calcified stenosis ≤5 cm | Endovascular preferred |
| C | Multiple stenoses or occlusions >15 cm total; recurrent stenosis/occlusion after two prior endovascular interventions | Surgery often preferred; endovascular acceptable in poor surgical candidates |
| D | CTO of common femoral or SFA >20 cm involving popliteal; CTO of popliteal and proximal trifurcation | Surgery preferred; endovascular for high-risk surgical patients |
Indications
| Indication | Clinical Context |
|---|---|
| Lifestyle-limiting claudication (Rutherford 2–3) | Failed 3-month supervised exercise therapy and pharmacotherapy; TASC A–B lesions; quality-of-life impairment documented |
| Critical limb ischemia (Rutherford 4–6) | Rest pain, tissue loss, or gangrene — revascularization to prevent major amputation; urgent; TASC A–D all considered; revascularize any patency target |
| Acute limb ischemia (Rutherford IIa/IIb) | Catheter-directed thrombolysis ± mechanical thrombectomy followed by angioplasty of culprit lesion; Rutherford III (irreversible) → surgical embolectomy |
| Pre-amputation optimization | BTK revascularization to heal wound or optimize amputation level; may allow more distal amputation and better rehabilitation potential |
| Subclavian steal / upper extremity ischemia | Subclavian stenosis proximal to vertebral origin causing vertebrobasilar symptoms or arm claudication; stenting from brachial or femoral access |
Contraindications
- Acute limb ischemia Rutherford class III (irreversible ischemia) — paralysis, anesthesia, rigid muscle; primary surgical intervention required; endovascular approach inappropriate
- Severe contrast allergy without pre-medication — CO2 angiography as alternative in confirmed allergy; pre-medicate if using iodinated contrast
- Uncorrectable coagulopathy — INR >3.0 or platelets <50,000 in elective setting; correct before proceeding
- No distal target vessel — no patent runoff to revascularize; discuss with vascular surgery for bypass candidacy (vein conduit, pedal bypass)
- Isolated aortoiliac disease with excellent surgical candidacy and TASC D lesion — aortobifemoral bypass offers superior long-term patency and is preferred when operative risk is acceptable
Relevant Anatomy
Aortoiliac Segment (Inflow)
The aorta bifurcates at the level of L4 into the right and left common iliac arteries (CIA). The CIA divides into the external iliac artery (EIA) and internal iliac artery. The EIA continues under the inguinal ligament to become the common femoral artery (CFA). Aortoiliac disease is the "inflow" problem — must be treated before or concurrent with infrainguinal intervention. CIA and EIA disease is typically approached from the contralateral femoral access. Balloon-expandable stents are preferred for iliac bifurcation lesions where precise placement is critical; self-expanding stents are preferred for the EIA given flexion at the hip.
Femoral-Popliteal Segment (Most Common Endovascular Target)
The CFA bifurcates at the groin into the superficial femoral artery (SFA) and the profunda femoris (deep femoral) artery. The SFA traverses Hunter's canal through the adductor hiatus — the mechanical stress at this location makes it the most common site of atherosclerotic occlusion and endovascular restenosis. The popliteal artery begins at the adductor hiatus and extends to the tibial trifurcation. Avoid stenting across the knee joint — stent fracture and occlusion rates are markedly elevated in this region. The Viabahn stent-graft is an option for recurrent SFA disease.
Infrapopliteal / Below-the-Knee Segment
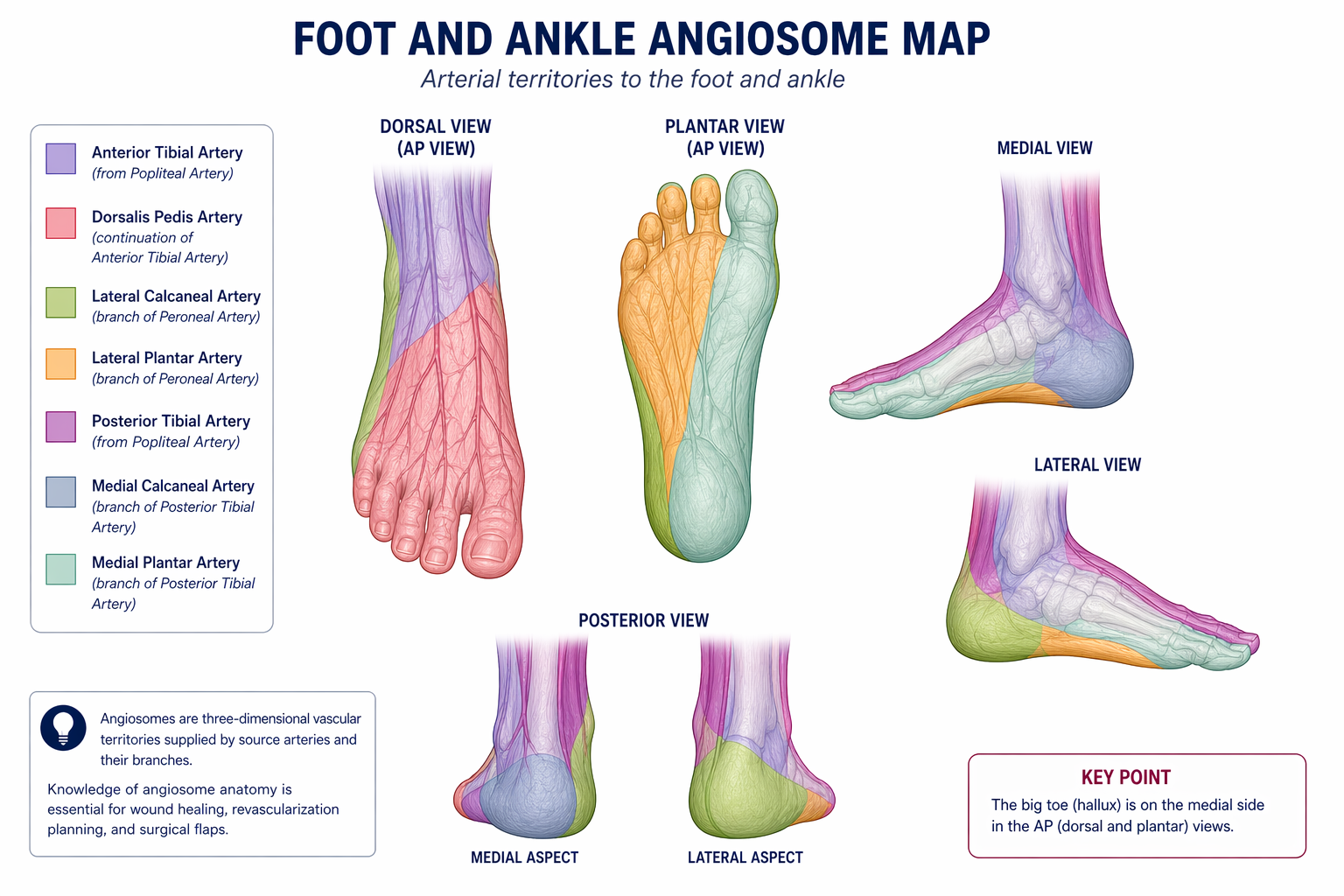
The popliteal artery trifurcates into the anterior tibial artery (travels through the interosseous membrane to the dorsum of the foot), posterior tibial artery (supplies the plantar surface and medial ankle), and peroneal artery (runs along the fibula; ends at the ankle without direct foot supply). These vessels are tortuous and small-caliber (2–4 mm). Long chronic total occlusions (CTOs) are common in CLI. Retrograde tibial or dorsal pedal access is used for complex lesions not crossable antegradely. Angiosome-directed revascularization — matching the revascularized vessel to the wound's angiosome territory — improves wound healing outcomes (supported by BEST-CLI subanalyses).
Pre-Procedure Checklist
Imaging Review
- Duplex ultrasound with ankle-brachial index (ABI) and waveform analysis — required before intervention; localizes disease level and confirms hemodynamic significance
- CTA runoff or MRA runoff to map disease extent from aorta to pedal vessels, characterize calcification pattern (medial vs. intimal), and guide device selection
- Assess contralateral access anatomy for crossover sheath placement if needed (iliac tortuosity, prior interventions)
- Wound photography and documentation for CLI patients
Medical Optimization
- Antiplatelet therapy: aspirin 81–325 mg daily; add clopidogrel 75 mg (dual antiplatelet) peri-procedurally; clopidogrel load 300–600 mg if not pre-loaded
- Statin therapy: all PAD patients regardless of cholesterol level (pleiotropic and plaque-stabilizing benefit)
- Supervised exercise therapy documented for claudicants before elective intervention
- Smoking cessation counseling — active smoking dramatically worsens patency; document cessation status
Labs
- CBC (hemoglobin, platelet count), creatinine/eGFR (contrast nephropathy risk), INR/PT
- Hold metformin 24–48 h pre-procedure if eGFR <45 or large contrast volume anticipated
- Intra-procedural anticoagulation: UFH 70–100 U/kg IV bolus at time of arterial access; target ACT 250–350 sec; check ACT 30 min after bolus and redose as needed
Consent
- Access site complications: hematoma (5–10%), pseudoaneurysm (~1–2%), AV fistula
- Procedural risks: dissection requiring stent, distal embolization ("trash foot"), vessel rupture, thrombosis
- Contrast nephropathy: pre-hydration; CO2 angiography alternative in advanced CKD
- Need for unplanned surgical intervention; restenosis (DCB reduces but does not eliminate); limb loss risk in CLI
- Paclitaxel DCB: discuss theoretical mortality signal (not confirmed in large registries) per informed consent best practice
Procedure Overview
The following is a high-level summary. Full access technique, wire crossing strategies for CTOs, device selection tables, drug-coated balloon sizing, and stent deployment protocols are available in RadCall Pro.
- Access — antegrade ipsilateral femoral access for SFA and BTK disease; micropuncture technique (21-gauge needle, 0.018" wire) to minimize hematoma; antegrade approach requires puncture above the inguinal ligament into the CFA with a caudal needle angle; confirm CFA position on fluoroscopy using the femoral head as a landmark (CFA overlies the medial two-thirds of the femoral head); introduce 6 Fr sheath; contralateral crossover access for iliac or proximal CFA lesions
- Diagnostic angiogram — roadmap the affected segment from above the lesion through runoff vessels; identify stenosis/occlusion, collateral vessels, and distal runoff; CO2 angiography if contrast use must be minimized; biplane or oblique views to separate overlapping vessels at trifurcation
- Anticoagulation — UFH 70–100 U/kg IV bolus; check ACT after 5 minutes; target ACT 250–350 sec; re-dose as needed for long cases
- Lesion crossing — hydrophilic wire (0.035" for above-knee; 0.018" for BTK) with low-profile support catheter; intraluminal crossing preferred; subintimal crossing technique for long CTOs (controlled re-entry with Outback or Pioneer re-entry catheter); confirm intraluminal distal wire position under fluoroscopy before treatment (no subintimal balloon inflation)
- Pre-dilation — balloon sized to reference vessel diameter (typically 0.5 mm undersized to reduce dissection risk); inflate to nominal or rated burst pressure based on lesion response; assess for flow-limiting dissection, elastic recoil, or calcification requiring additional strategy
- Atherectomy (if planned) — directional (HawkOne) for eccentric, soft/mixed plaque; rotational/orbital (Diamondback 360°, Jetstream) for calcified lesions; laser (Spectranetics ELCA) for in-stent restenosis or heavily calcified lesions; embolic protection device (SpiderFX or FilterWire) strongly recommended, especially for BTK atherectomy; atherectomy precedes DCB to reduce dissection and improve paclitaxel delivery
- Drug-coated balloon (DCB) — size balloon to vessel diameter (1:1 balloon-to-vessel ratio); slow, prolonged inflation (minimum 3 minutes) at nominal pressure to ensure paclitaxel transfer; do not use DCB in popliteal below P3 segment or BTK vessels outside of clinical trials — insufficient safety and efficacy data; observe for flow-limiting dissection post-DCB before concluding
- Stenting (if needed — provisional strategy) — reserve stenting for residual stenosis >30%, flow-limiting dissection (type C or above), or elastic recoil; nitinol self-expanding stents (Absolute Pro, Innova, SMART) for SFA — accommodate vessel movement and resist fracture; balloon-expandable stents (Express LD, iCast) for CFA or iliac — precise deployment at bifurcation; avoid stent placement across the knee joint; post-dilate with appropriately sized balloon; confirm full stent apposition
- Completion angiogram — assess treated segment in two projections; confirm residual stenosis <30%, patent inline runoff to foot, absence of flow-limiting dissection or distal embolism; treat any complications before sheath removal
Drug-Coated Balloon Evidence
Key Trials and Data
- IN.PACT SFA trial (2015): DCB vs. plain balloon angioplasty in SFA and proximal popliteal artery disease; 12-month primary patency 82.2% vs. 52.4% (p<0.001); benefit sustained through 5-year follow-up; established DCB as standard of care for femoropopliteal disease
- Paclitaxel mortality signal (Katsanos 2018 meta-analysis): pooled meta-analysis suggested a late (2- and 5-year) all-cause mortality increase with paclitaxel-coated balloons and stents in the femoropopliteal segment; generated significant controversy and FDA review; subsequent large independent registry analyses (ILLUMINATE Pivotal, REAL PTX 2020, SVS VQI registry, ISAR-STATH) did not confirm a mortality signal; FDA and society guidance supports continued use with documented informed consent; ongoing surveillance recommended
- BTK DCB: BELOW trial and LUTONIX BTK trial demonstrated mixed results without clear benefit in infrapopliteal disease; BTK DCB is not routinely recommended outside of clinical trials; plain balloon angioplasty remains standard for BTK revascularization
Atherectomy Devices
| Device Type | Mechanism | Best Use Case | Notes |
|---|---|---|---|
| Directional (HawkOne / TurboHawk) | Rotating blade excises and collects plaque into nosecone chamber | Eccentric, non-calcified or mixed plaque; SFA and popliteal | Requires embolic protection device; DEFINITIVE AR trial supports directional atherectomy + DCB combination |
| Rotational (Jetstream) | Differential cutting and aspiration of plaque; active aspiration of debris | Moderate calcification; SFA; in-stent restenosis | Integrated aspiration reduces embolic burden; embolic protection still recommended |
| Orbital (Diamondback 360°) | Diamond-coated crown sands calcified plaque with centrifugal force | Heavily calcified lesions; all infrainguinal segments | Accommodates severe circumferential calcification; embolic protection recommended; crown size selected by vessel diameter |
| Laser (Spectranetics ELCA) | Pulsed UV laser energy (308 nm) causes photochemical and photothermal plaque ablation | In-stent restenosis (drug of choice); heavily calcified lesions; BTK; occluded stents | Requires saline or CO2 flush during activation (no blood in lasing field); smaller plaque fragments; embolic protection for BTK use |
Atherectomy is not recommended as monotherapy — always follow with DCB or angioplasty to optimize the treated segment. The DEFINITIVE AR trial (2017) demonstrated that directional atherectomy followed by DCB improved 12-month primary patency compared to DCB alone for complex femoropopliteal lesions.
Complications
| Complication | Rate | Management |
|---|---|---|
| Distal embolization ("trash foot") | 2–5% | Embolic protection device for atherectomy; aspiration thrombectomy (AngioJet, Export catheter) for acute macroembolism; catheter-directed tPA infusion for microvascular embolism; surgical consultation for limb-threatening embolism |
| Vessel dissection | Common post-angioplasty | Graded by NHLBI classification A–F; flow-limiting dissection (C and above) requires stenting; provisional stenting preferred over primary stenting; prolonged balloon tamponade (3–5 min) for borderline dissections |
| Vessel rupture / perforation | <1% | Covered stent-graft (Viabahn for SFA; iCast for iliac/popliteal) for containment; prolonged balloon tamponade at rupture site while preparing covered stent; surgical backup for uncontrolled extravasation or covered stent failure |
| Acute thrombosis | 1–3% | UFH bolus; aspiration thrombectomy; catheter-directed tPA infusion (0.5–1 mg/hr); evaluate for underlying culprit lesion (inadequately treated stenosis, stent underexpansion) |
| Access site hematoma / pseudoaneurysm | Hematoma 5–10%; pseudoaneurysm ~1–2% | Manual or mechanical compression; ultrasound-guided thrombin injection (200–1,000 U) for pseudoaneurysm; surgical repair for large or rapidly expanding hematomas |
| Restenosis | 20–40% at 12 months (plain PTA); 10–20% with DCB | Repeat PTA or DCB; atherectomy for in-stent restenosis; Viabahn covered stent-graft for recurrent SFA restenosis (VIPER trial); surveillance duplex essential for early detection |
| Contrast nephropathy | Variable (1–20% depending on baseline eGFR) | Pre-hydration with isotonic saline (1 mL/kg/hr 6–12 h before and after); minimize contrast volume; CO2 angiography as primary imaging in eGFR <30; avoid nephrotoxic medications peri-procedurally; N-acetylcysteine evidence weak but low risk |
Post-Procedure Care
- Dual antiplatelet therapy: aspirin 81–325 mg daily (lifelong) + clopidogrel 75 mg daily for 1–3 months post-stent; consider extended DAPT for complex or BTK revascularization
- Antithrombotic therapy — VOYAGER PAD (2020, 6,564 patients): rivaroxaban 2.5 mg BID + aspirin vs. aspirin alone post-revascularization; significant reduction in major adverse limb events and major cardiovascular events (HR 0.85, 95% CI 0.76–0.96); also reduced acute limb ischemia; consider for patients who underwent revascularization and have acceptable bleeding risk
- Statin therapy: all PAD patients regardless of baseline LDL; high-intensity preferred
- Duplex ultrasound surveillance: at 1 month and 6 months post-procedure, then annually; assess peak systolic velocity ratios at treated segments; velocity ratio >2.5 suggests significant restenosis requiring intervention
- ABI measurement: at 1 month; improvement of >0.1 from baseline ABI indicates hemodynamic success; toe-brachial index (TBI) useful in heavily calcified vessels (non-compressible)
- Wound care (CLI patients): coordinate with vascular surgery, wound care team, or podiatry; off-loading, infection control, debridement as needed; wound healing may take weeks to months after revascularization
- Smoking cessation: mandatory — active smoking dramatically worsens arterial patency; pharmacotherapy (varenicline, NRT) and behavioral support
- Access site monitoring: vital signs and access site checks every 30 minutes for 2 hours post-procedure; antegrade femoral access carries higher hematoma risk than retrograde — monitor closely
When to Escalate
- Acute limb ischemia Rutherford III during procedure — hard neurologic findings (paralysis, anesthesia, muscle rigidity); emergent surgical embolectomy or bypass; notify vascular surgery immediately
- Vessel rupture not controlled by covered stent — maintain balloon tamponade; vascular surgery for open repair; ensure blood products available
- Failed wire crossing after multiple attempts — do not persist beyond 60–90 minutes; vascular surgery for hybrid approach (surgical endarterectomy + endovascular extension) or open bypass
- Impending major amputation with no endovascular target — BEST-CLI data supports surgical bypass (vein conduit) in patients with good conduit and high-complexity tibial lesions (GLASS stage III); multidisciplinary limb salvage team discussion
- Worsening CLI despite technically successful revascularization — re-evaluate patency with duplex; vascular surgery for repeat revascularization or amputation planning; wound care team; consider hyperbaric oxygen in select cases
- Distal embolization not responding to aspiration or thrombolytics — vascular surgery for surgical thromboembolectomy
References
- Gerhard-Herman MD et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease. J Am Coll Cardiol. 2017;69(11):e71–e126.
- Tepe G et al. Drug-coated balloon versus standard percutaneous transluminal angioplasty for treatment of SFA and popliteal artery (IN.PACT SFA). Circulation. 2015;131(5):495–502.
- Bonaca MP et al. Rivaroxaban in peripheral artery disease after revascularization (VOYAGER PAD). N Engl J Med. 2020;382(21):1994–2004.
- Norgren L et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg. 2007;45(Suppl S):S5–S67.
- Farber A et al. Surgery or endovascular therapy for chronic limb-threatening ischemia (BEST-CLI). N Engl J Med. 2022;387(25):2305–2316.
- Katsanos K et al. Risk of death following application of paclitaxel-coated balloons and stents in the femoropopliteal artery (meta-analysis). J Am Heart Assoc. 2018.