Nomenclature and Classification
The term pelvic congestion syndrome is increasingly replaced by the broader term pelvic venous disorders (PeVD), as the former fails to specify the underlying pathophysiology.[4] The SVP classification (Symptoms–Varices–Pathophysiology), developed by the American Vein & Lymphatic Society, provides a standardized framework:[5]
| Domain | Scale | Description |
|---|---|---|
| S — Symptoms | S0–S3 | Graded across anatomic zones from the renal vein to the lower extremities |
| V — Varices | V0–V3 | Based on location: renal hilum, pelvic plexuses, extrapelvic transitional veins |
| P — Pathophysiology | A / H / E | Anatomy (A), Hemodynamics (H: reflux R vs. obstruction O), Etiology (E: nonthrombotic NT vs. thrombotic T) |
Example designations: classic bilateral ovarian reflux PCS = S₂V₂P(BGV,R,NT); nutcracker syndrome = S₁V₁P(LRV,O,NT).
Indications and Contraindications
| Indication | Context |
|---|---|
| Chronic pelvic pain >6 months with PeVD features | Pain worse upright/premenstrually, post-coital ache, dyspareunia, dysmenorrhea; other causes excluded (endometriosis, adhesive disease, interstitial cystitis) |
| Documented ovarian vein reflux or pelvic varices | TVUS, MR/CT venography, or catheter venography confirmation |
| Vulvar, perineal, or atypical lower-extremity varicosities | Originating from pelvic escape points (inguinal, obturator, pudendal, clitoral) |
| Recurrent lower-extremity varicosities after saphenous ablation | Pelvic source feeding atypical collaterals |
| Nulliparous patients with documented reflux and symptoms | Published data show VAS reduction 8.5 → 2.4 at 90 days and 90% satisfaction |
| Type | Contraindication |
|---|---|
| Absolute | Pregnancy · Active pelvic infection · Uncorrectable coagulopathy |
| Relative | Subclinical dilated veins without symptoms (12% prevalence on CT in asymptomatic women — clinical correlation essential) · Severe contrast allergy · Renal impairment · Fertility concerns — AMH declines 9–14% at 12 months; monitor in patients <30 |
Relevant Anatomy
The pelvic venous plexus receives tributaries from the uterus, ovaries, broad ligament, and adjacent pelvic viscera. Pathologic reflux originates from:
- Left ovarian (gonadal) vein: drains into the left renal vein at a near-perpendicular angle; valvular incompetence common; most frequently refluxing vessel. Nutcracker compression of the left renal vein can contribute to left-sided hypertension.
- Right ovarian vein: drains directly into IVC; less commonly refluxing but bilateral treatment is frequently required.
- Internal iliac tributaries: obturator, internal pudendal, inferior gluteal, and lateral sacral veins; contribute to parauterine varices and pelvic escape points. Untreated iliac tributaries are the leading cause of recurrence.
- Secondary obstruction: left renal vein compression (nutcracker syndrome) and left common iliac vein compression (May-Thurner syndrome) drive pelvic venous hypertension from outflow obstruction — these require stenting, not embolization, as first-line treatment.
Diagnosis
Transvaginal Duplex Ultrasound (TVUS) — First-Line
TVUS is the recommended first-line imaging tool (SVS/AVF Grade 1C). Key diagnostic criteria:[1][6][7]
| Parameter | Abnormal Threshold |
|---|---|
| Ovarian vein diameter | >6–8 mm |
| Ovarian vein flow velocity | ≤3 cm/s |
| Myometrial vein diameter | >5 mm |
| Valsalva-evoked flow direction | Flow reversal or caudal reflux >1 s |
| Parauterine/pelvic veins | Tortuous dilated networks |
A validated ultrasound-based scoring system integrating these parameters achieves an AUC of 0.861 for predicting venography-confirmed PeVD, with a positive predictive value of 93% and negative predictive value of 92%.[6]
CT and MR Venography
Useful for pre-treatment planning and identification of structural causes. CT venography identifies nutcracker anatomy (left renal vein compression between the aorta and SMA) and May-Thurner anatomy (left common iliac vein compression by the right iliac artery). MR venography demonstrates tortuous parauterine varices without ionizing radiation and is preferred in young patients.[2][8]
Retrograde Venography — Gold Standard
Retrograde venography remains the diagnostic gold standard and is recommended when intervention is planned (SVS/AVF Grade 1C).[8] Laparoscopy has limited sensitivity for PeVD because CO&sub2; pneumoperitoneum compresses pelvic veins and underestimates the degree of venous congestion.[1]
Beard Venographic Criteria
Venographic diagnosis of PCS is established when ≥3 of the following 4 Beard criteria are met:[3] Score ≥5 on the image-based scoring system: 91% sensitivity, 89% specificity. See the PeVD Imaging guide for complete US, MRI, CT, and IVUS protocols.
| # | Criterion |
|---|---|
| 1 | Ovarian vein diameter >6 mm |
| 2 | Contrast retention >20 seconds after selective injection |
| 3 | Opacification of ipsilateral and/or contralateral pelvic venous plexus, or filling of vulvovaginal veins |
| 4 | Filling of thigh or perineal varicosities |
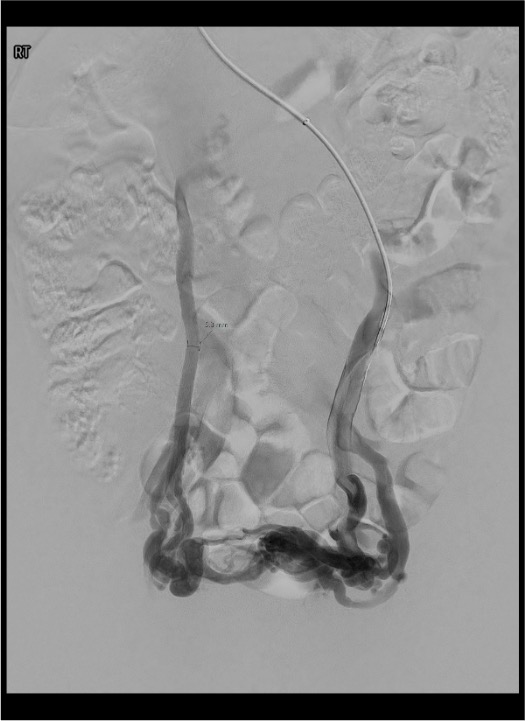
Important caveat: Dilated, refluxing pelvic veins are found in approximately 12% of asymptomatic women on CT. A positive venogram alone is insufficient — clinical correlation with a consistent pain syndrome is essential before proceeding with embolization.[18][19] If no improvement occurs after initial embolization, a repeat procedure is unlikely to be effective.[9]
Procedure Overview
The following is a high-level summary. Full catheter and microcatheter selection, coil sizing tables, STS foam preparation ratios, and internal iliac tributary targeting algorithms are available in RadCall Pro.
Step 1 — Identify Pathophysiology Before Intervening
Treatment depends on distinguishing primary reflux from secondary obstruction:[2]
- Primary reflux (incompetent gonadal/internal iliac valves) → transcatheter embolization
- Secondary obstruction (nutcracker, May-Thurner) → treat the obstruction first with stenting before considering gonadal vein embolization; embolizing a hypertensive outflow-obstructed system leads to early recurrence
Step 2 — Access and Venographic Assessment
- Access: right internal jugular vein (preferred — favorable angle for left renal/ovarian vein) or right common femoral vein; 5–6 Fr sheath.
- Left renal venogram: 5 Fr cobra or reverse-curve catheter; identify ovarian vein ostium; selective left renal venogram with Valsalva. Evaluate for nutcracker anatomy (renal vein diameter ratio, pressure gradient >3 mmHg).
- Left ovarian venogram: advance into gonadal vein; Valsalva to confirm reflux and map pelvic collaterals and escape points.
- Right ovarian venogram: direct IVC catheterization; right gonadal vein origin smaller and more oblique.
- Bilateral internal iliac venograms: with Valsalva; identify refluxing tributaries (pudendal, obturator, inferior gluteal) and escape points. These are the main driver of recurrence if untreated.
Step 3 — Embolization (Primary Reflux)
- Ovarian vein embolization: advance microcatheter to the pelvic inlet; deploy coils or inject foam sclerosant distally; retract and occlude the trunk up to ~1 cm from the renal vein ostium (prevents coil migration into renal vein or IVC).
- Sandwich technique (preferred): distal coils or foam → mid-trunk sclerosant → proximal coil anchor. Lowest recurrence in comparative series.
- Collateral occlusion: superselective catheterization and embolization of refluxing internal iliac tributaries — this step is critical and is the main determinant of long-term success.
- Completion venogram: confirm stasis, absence of reflux on Valsalva, and no non-target filling.
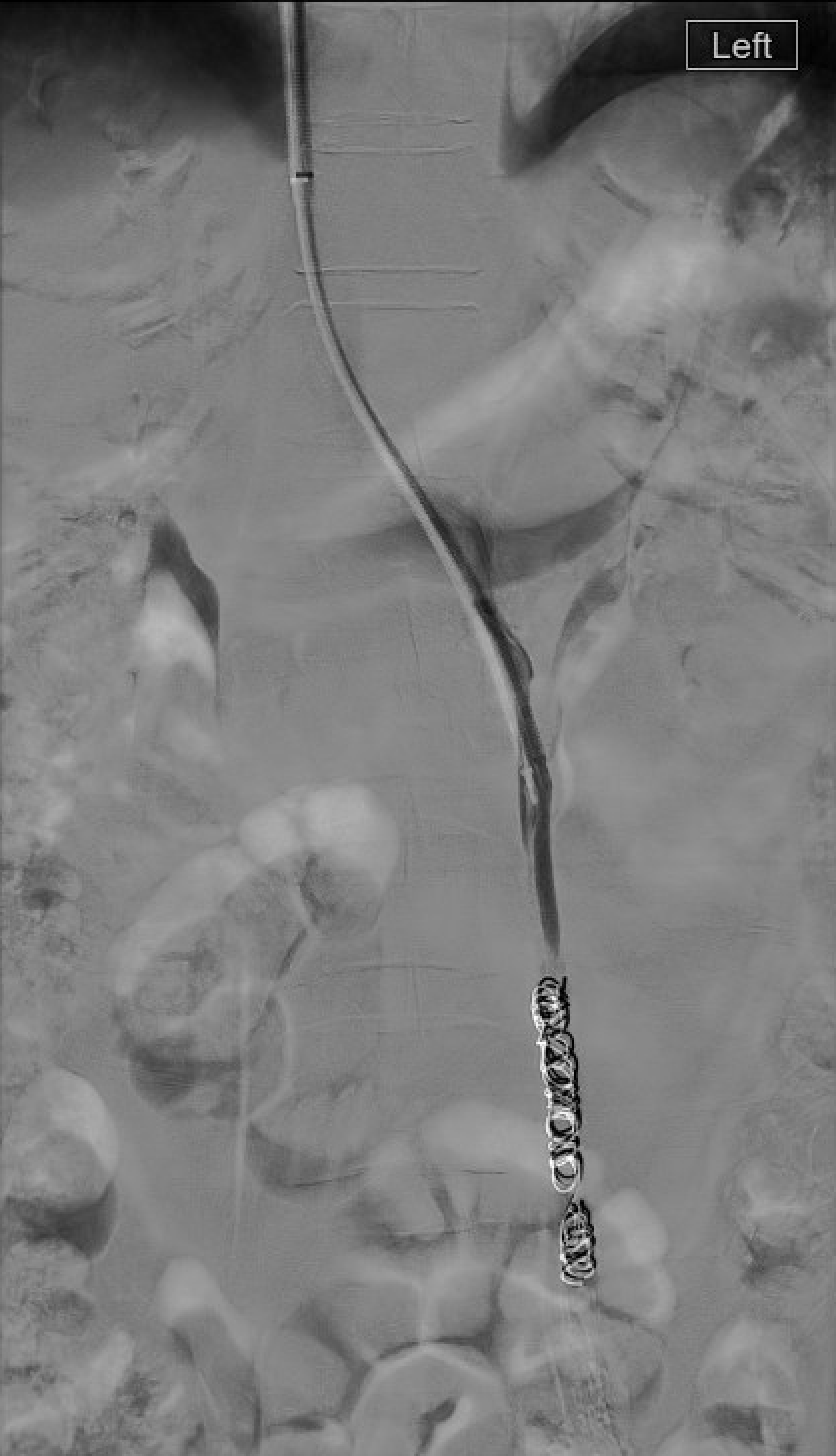
Embolic Agent Selection
| Agent | Role | Notes |
|---|---|---|
| Coils (pushable/detachable) | Proximal and distal anchors in gonadal vein trunk | Precise placement; sized 20–30% larger than vein diameter |
| Polidocanol or STS foam (3%, 1:4 air ratio) | Sclerose parauterine and pudendal varices | Penetrates small collaterals; coil + polidocanol foam combination shows 76% clinical effectiveness |
| Amplatzer vascular plug | Single-device proximal trunk occlusion | Requires 4–6 Fr delivery; not for tortuous or small-caliber veins |
| n-BCA glue | Tortuous iliac tributaries | Operator-dependent; catheter adherence risk |
Iliac Vein Stenting (Secondary Obstruction)
When PeVD is caused by May-Thurner syndrome or nutcracker syndrome, treat the obstruction first before addressing gonadal vein reflux:[2][3][15]
- A single-center series (n=45 women, 38% MTS alone, 53% MTS + nutcracker) treated with bilateral iliac vein balloon angioplasty ± stenting ± ovarian vein embolization showed significant reductions in pain scores and analgesic use.[16]
- Iliac vein stenting for MTS without acute DVT (n=34) achieved 68% clinical success; female sex correlated with better outcomes.[17]
- Nonthrombotic iliac vein lesions (NIVLs) generally maintain excellent stent patency but require careful selection to avoid over-treating normally functioning veins (SIR position statement).[15]
Complications
| Complication | Rate | Management |
|---|---|---|
| Post-embolization syndrome (pelvic pain, low-grade fever) | 5–15% | Self-limited 2–7 days; NSAIDs; transient worsening of baseline pain |
| Pelvic vein thrombosis | ~7% (coil + foam series)[12] | Anticoagulation; most resolve; risk higher with foam sclerosant |
| Coil migration to pulmonary circulation | ~1.3%[11] | Usually asymptomatic; snare retrieval if large or symptomatic; prevented by appropriate sizing and distal anchor placement |
| Recurrence / persistent symptoms | 5–11%[11][12] | Usually untreated internal iliac tributaries; repeat embolization; second procedure unlikely effective if first failed[9] |
| Ovarian reserve decline (AMH) | 9–14% at 12 months[14] | Consistent with physiological aging; monitor AMH in patients <30 |
| Non-target embolization | Rare | Preserve utero-ovarian anastomoses; careful microcatheter positioning |
| Access-site hematoma / contrast nephropathy | Low | Standard precautions |
Post-Procedure Care
- Same-day discharge after 2–4h bed rest (jugular or femoral access).
- Analgesia: NSAIDs for post-embolization syndrome; typically resolves within 7 days; scrotal/pelvic support.
- Follow-up: pain assessment (VAS) at 4–6 weeks and 3–6 months; Doppler US if recurrent symptoms.
- AMH monitoring: baseline and 12-month measurement in patients desiring future fertility, especially those under 30.
- Second stage: if symptoms persist after ovarian vein embolization alone and internal iliac reflux was not addressed, stage an internal iliac tributary session. If initial embolization failed comprehensively, repeat embolization is unlikely to be effective.[9]
Evidence Summary
- De Gregorio MA et al (2020, JVIR) — largest series, n=520, mean follow-up 59 months: VAS improved from 7.6 pretreatment to 0.9 at 5 years. Symptom recurrence 5%; minor complications 10.9%; major complications 2.1% (including 1.3% coil migration to lung).[11]
- Daniels JP et al (2016, JVIR) — systematic review of 21 prospective series, n=1,308: ~75% early substantial pain relief, generally increasing and sustained over time, with low reintervention rates.[10]
- Wang Y et al (2026, Frontiers in Medicine) — coil + polidocanol foam, n=158: 100% technical success, 76% clinical effectiveness at final follow-up, 7% complication rate (primarily pelvic vein thrombosis). Parity ≥3 and estrogen-dependent disease were independent risk factors for poor prognosis.[12]
- Szmygin M et al (2024, CVIR) — nulliparous patients, n=21: mean VAS reduced from 8.5 to 2.4 at 90 days; 90% patient satisfaction.[13]
- Karakaya HC et al (2025, Reprod Biol Endocrinol) — ovarian reserve, n=81: AMH declined 9–14% at 12 months post-embolization — consistent with physiological aging; changes in patients under 30 warrant monitoring.[14]
- Champaneria R et al (2016, Health Technology Assessment) — systematic review: heterogeneous but consistent improvement across published series; prospective RCT data remain limited.[19]
- Mansouri-Tehrani MM et al (2026, J Ultrasound Med): TVUS-based scoring system achieves AUC 0.861 for predicting venography-confirmed PeVD; PPV 93%, NPV 92%.[6]
References
- Krambeck C, Tesch K, Watrowski R, Maass N, Alkatout I. Pelvic Congestion Syndrome: The Gynecological Perspective. J Clin Med. 2026;15(4):1655.
- Bookwalter CA, VanBuren WM, Neisen MJ, Bjarnason H. Imaging Appearance and Nonsurgical Management of Pelvic Venous Congestion Syndrome. Radiographics. 2019;39(2):596–608.
- Barge TF, Uberoi R. Symptomatic Pelvic Venous Insufficiency: A Review of Current Controversies. Clin Radiol. 2022;77(6):409–417.
- Knuttinen MG, Machan L, Khilnani NM, et al. Diagnosis and Management of Pelvic Venous Disorders: Expert Panel Narrative Review. AJR. 2023;221(5):565–574.
- Meissner MH, Khilnani NM, Labropoulos N, et al. The SVP Classification of Pelvic Venous Disorders. J Vasc Surg Venous Lymphat Disord. 2021;9(3):568–584.
- Mansouri-Tehrani MM, Shahnazari R, Iraji H, et al. Optimizing Venographic Referrals in Pelvic Congestion Syndrome: A Prospective Diagnostic Accuracy Study. J Ultrasound Med. 2026. doi:10.1002/jum.70199.
- Expert Panel on GYN and OB Imaging; Shampain KL, Feldman MK, et al. ACR Appropriateness Criteria® Postmenopausal Subacute or Chronic Pelvic Pain: Update 2025. J Am Coll Radiol. 2026.
- Gloviczki P, Comerota AJ, Dalsing MC, et al. Clinical Practice Guidelines of the SVS and AVF for Varicose Veins and Chronic Venous Disease. J Vasc Surg. 2011;53(5 Suppl):2S–48S.
- Rochon PJ, Reghunathan A, Kapoor BS, et al. ACR Appropriateness Criteria® Lower Extremity Chronic Venous Disease. J Am Coll Radiol. 2023;20(11S):S481–S500.
- Daniels JP, Champaneria R, Shah L, et al. Effectiveness of Embolization or Sclerotherapy of Pelvic Veins for Chronic Pelvic Pain: Systematic Review. J Vasc Interv Radiol. 2016;27(10):1478–1486.
- De Gregorio MA, Guirola JA, Alvarez-Arranz E, et al. Pelvic Venous Disorders in Women Due to Pelvic Varices: Treatment by Embolization — Experience in 520 Patients. J Vasc Interv Radiol. 2020;31(10):1560–1569.
- Wang Y, Li F, Niu G, et al. Feasibility, Safety, and Effectiveness of Coil and Foam Embolization in Venous-Origin Chronic Pelvic Pain. Front Med. 2026;13:1741818.
- Szmygin M, Pyra K, Bérczi V, et al. Endovascular Treatment of Pelvic Venous Congestion Syndrome in Nulliparous Patients. Cardiovasc Intervent Radiol. 2024;47(6):822–828.
- Karakaya HC, Dirican Ozen B, Eskin P, et al. Efficacy of Endovenous Embolization for Pelvic Congestion Syndrome and Its Impact on Ovarian Reserve. Reprod Biol Endocrinol. 2025;23(1):132.
- Vedantham S, Weinberg I, Desai KR, et al. SIR Position Statement on Chronic Iliofemoral Venous Obstruction With Endovascular Stents. J Vasc Interv Radiol. 2023;34(10):1643–1657.
- Wu WC, Hsu WH, Chang TC, Huang LW. Pelvic Congestion Syndrome Due to Central Venous Outflow Obstruction: May-Thurner and Nutcracker Syndromes. Int J Gynaecol Obstet. 2025. doi:10.1002/ijgo.70268.
- Ahmed O, Ng J, Patel M, et al. Endovascular Stent Placement for May-Thurner Syndrome in the Absence of Acute DVT. J Vasc Interv Radiol. 2016;27(2):167–173.
- Jurga-Karwacka A, Karwacki GM, Schoetzau A, et al. A Forgotten Disease: Pelvic Congestion Syndrome as a Cause of Chronic Lower Abdominal Pain. PLoS One. 2019;14(4):e0213834.
- Champaneria R, Shah L, Moss J, et al. The Relationship Between Pelvic Vein Incompetence and Chronic Pelvic Pain: Systematic Reviews. Health Technol Assess. 2016;20(5):1–108.
- Related IR guides: uterine fibroid embolization, varicocele embolization, IVC filter placement.