Indications & Contraindications
Indications
- Heavy menstrual bleeding (menorrhagia) — most common indication; ~90% report improvement post-UFE
- Bulk symptoms — pelvic pressure/pain, urinary frequency, abdominal distention
- Failed or intolerant of medical therapy (hormonal, GnRH agonists)
- Desire for uterine preservation as alternative to myomectomy or hysterectomy
- Multiple fibroids (treats all fibroids simultaneously, unlike myomectomy)
Contraindications
- Pregnancy
- Active pelvic infection / endometritis
- Pedunculated subserosal fibroid with stalk <50% of fibroid diameter (risk of detachment and peritonitis)
- Known or suspected gynecologic malignancy
- IUD in place (remove prior to procedure)
- Relative: Desire for future fertility — myomectomy generally preferred; limited data on pregnancy outcomes post-UFE
- Relative: Very large fibroid uterus extending above umbilicus (may have suboptimal response)
Pre-Procedure Planning
Imaging & Labs
- MRI pelvis with and without contrast — map fibroid number, size, location; exclude adenomyosis and malignancy; assess vascularity and enhancement pattern
- Labs: CBC, BMP, coagulation panel (PT/INR, PTT)
- Pregnancy test (serum beta-hCG) — mandatory
- TSH if menorrhagia workup (exclude thyroid cause)
- Type & screen not routinely required
Patient Preparation
- GYN clearance — gynecologist evaluation and agreement on management plan; recent Pap smear; endometrial biopsy if indicated
- NPO per institutional protocol (moderate sedation)
- Pain management plan: arrange PCA pump (morphine or hydromorphone), ketorolac 30 mg IV, ondansetron 4 mg IV
- Discuss overnight admission vs same-day discharge based on institutional protocol
Relevant Anatomy
Uterine Artery
- Arises from the anterior division of the internal iliac artery (IIA)
- Courses medially along the pelvic floor, crossing over the ureter (“water under the bridge”) before reaching the lateral aspect of the uterus
- Gives off the cervicovaginal branch (supplies cervix and upper vagina) before ascending along the lateral uterine body
- Ascending segment gives fibroid branches and ultimately anastomoses with the ovarian artery at the uterine fundus (ovarian-uterine anastomosis)
- The horizontal segment of the uterine artery is the target for catheterization — embolize PAST the cervicovaginal branch takeoff
Variant Anatomy & Collaterals
- Variant origin: uterine artery may arise from the external iliac artery or share a common trunk with the obturator artery
- Ovarian artery supply: 4–8% of patients have significant collateral supply to fibroids via the ovarian arteries (from the aorta); may require additional embolization from an aortic approach
- Utero-ovarian anastomosis types (Razavi classification): Type I — intramyometrial, Type II — tubal, Type III — through ovarian parenchyma; Types Ib and III have higher risk of non-target ovarian embolization
- Bilateral embolization is required — unilateral embolization produces suboptimal results due to cross-collateral supply
Key Angiographic Relationships
On selective internal iliac artery angiography, the uterine artery is identified as a medially coursing branch from the anterior division. The cervicovaginal branch arises early from the uterine artery and courses inferiorly toward the cervix. Microcatheter position must be confirmed distal to this branch before embolization to avoid cervical necrosis. At the fundus, look for the ascending uterine artery anastomosing with the descending ovarian artery — if this is prominent, particle size selection is critical to avoid retrograde ovarian embolization.
Technique
Access & Catheters
- Right common femoral artery (CFA) access — single puncture, bilateral catheterization
- 5F vascular sheath
- 5F Cobra (C2) catheter or Roberts uterine catheter for IIA selection
- Microcatheter 2.4–2.8F (e.g., Progreat, Renegade) for superselective uterine artery catheterization
- 0.035” Glidewire and 0.018” microwire
Embolic Agents
- Tris-acryl gelatin microspheres (Embosphere) — preferred; calibrated, predictable penetration
- Size: 500–700 µm or 700–900 µm
- PVA particles (500–710 µm) — acceptable alternative
- Avoid particles <500 µm — risk of deep penetration into ovarian branches and non-target embolization
- Mix with contrast (50:50 with saline/contrast) for fluoroscopic visualization during injection
Medications
- Cefazolin 1g IV pre-procedure
- Moderate sedation (midazolam + fentanyl) or PCA pump
- Ketorolac 30 mg IV (at start of procedure)
- Ondansetron 4 mg IV PRN nausea
- Nitroglycerin 100–200 µg IA (available for uterine artery spasm)
- Heparin per institutional protocol (optional)
Arterial Access
Contralateral IIA Catheterization (Left Side First)
Superselective Microcatheter Placement
Embolization — Left Uterine Artery
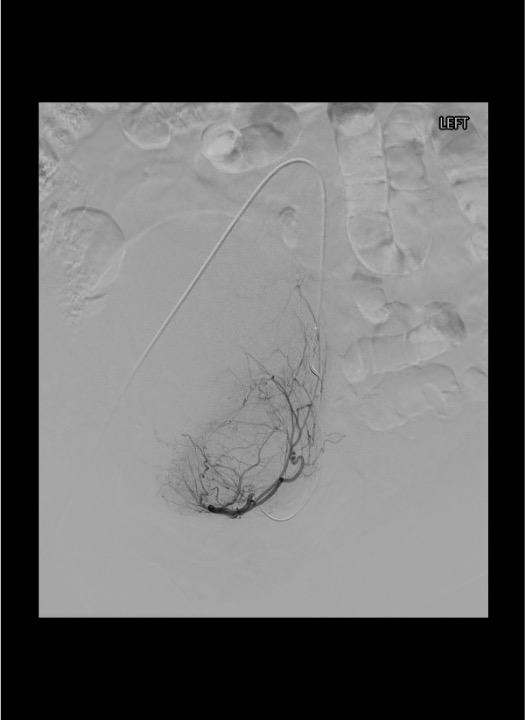
Ipsilateral IIA Catheterization — Waltman Loop Technique
Option A — Waltman Loop (preferred):
- Retract the Cobra/RUC to just above the aortic bifurcation with the tip pointing cephalad.
- Advance the catheter forward while applying gentle clockwise torque — the tip encounters the opposite aortic wall and begins to buckle, forming a reverse-curve (the "loop").
- Continue advancing until a stable J-shaped loop is formed in the distal aorta, with the tip now pointing caudally toward the ipsilateral iliac.
- Pull back slowly on the catheter — in reverse-curve configuration, traction causes the tip to advance distally (paradoxical motion). The loop "walks" down into the ipsilateral IIA.
- Confirm stable IIA position with a hand injection of contrast. Steep ipsilateral oblique (30–45°) to separate anterior and posterior divisions.
Option B — RUC "Push into Aorta, Rotate & Pull":
- Advance a stiff 0.035″ wire deep into the contralateral external iliac to anchor the system.
- Push the catheter body deep into the contralateral iliac, gaining slack in the aortic segment.
- Retract the wire back to the aorta; the catheter tip will prolapse off the bifurcation.
- With clockwise torque, redirect the tip into the ipsilateral common iliac → advance into IIA.
Once the ipsilateral IIA is selected: obtain selective angiogram, identify right uterine artery, advance microcatheter past the cervicovaginal branch.
Embolization — Right Uterine Artery
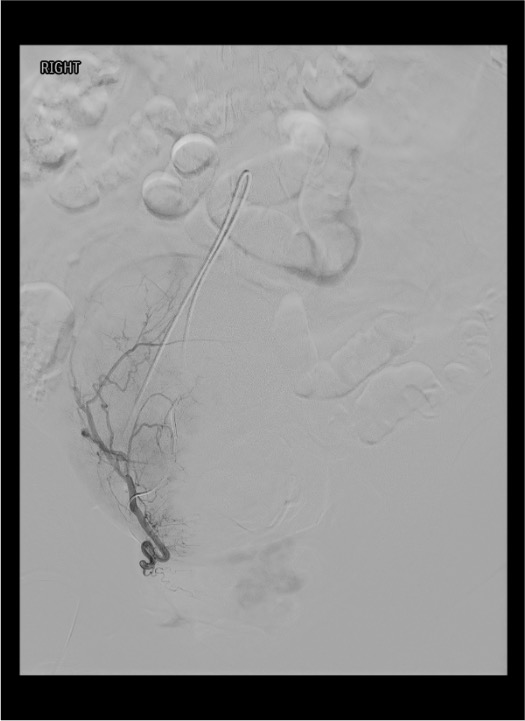
Completion & Closure
Community Cards
Angiographic Landmarks
Uterine Artery Identification
- IIA angiogram: uterine artery courses medially from the anterior division toward the midline pelvis
- Characteristic “hook” or “U-turn” as it crosses over the ureter and turns superiorly along the lateral uterus
- Fibroids demonstrate a dense vascular blush on arterial phase — confirms fibroid vascularity and supply from this uterine artery
- Small or spastic uterine artery may be difficult to identify — use nitroglycerin IA if needed
Critical Branch Points
- Cervicovaginal branch takeoff: arises from the uterine artery early in its course; supplies cervix and upper vagina; must embolize DISTAL to this branch
- Ovarian artery collateral supply: look for retrograde filling of ovarian artery at the fundus on late-phase images; if prominent, consider aortogram to evaluate for accessory ovarian artery supply requiring separate embolization
- Anastomotic arcade: ascending uterine artery connects with descending ovarian artery at the fundus — this is the route for potential non-target ovarian embolization
Troubleshooting
Small or Spastic Uterine Artery Not Visible on IIA Angiogram
Administer nitroglycerin 100–200 µg IA directly into the anterior division of the IIA. Wait 30–60 seconds and repeat angiogram. If still not visible, try a more selective anterior division injection. Consider variant anatomy — the uterine artery may arise from the external iliac or share a trunk with the obturator artery. Review pre-procedure MRA if available.
Fibroids Receiving Significant Collateral Supply from Ovarian Arteries
If prominent ovarian artery supply is identified on pre-procedure MRA or on angiography (retrograde filling at fundus), selective catheterization of the ovarian artery from the aorta is required for complete embolization. Use a Cobra or SOS catheter from the femoral approach to select the ovarian artery origin from the anterior aorta (typically at L2 level, below the renal arteries). Embolize with same particles. Be cautious to avoid reflux into ovarian branches.
Unable to Catheterize One Uterine Artery
Unilateral embolization is suboptimal but may be acceptable in select cases. The un-embolized side will continue to supply the fibroids via cross-midline collaterals. Options: (1) attempt from contralateral femoral access, (2) attempt radial approach, (3) accept unilateral result and plan for potential repeat procedure. Document clinical rationale. Outcomes with unilateral embolization are inferior to bilateral.
Uterine Artery Spasm During Catheterization or Embolization
Administer nitroglycerin 100–200 µg IA through the microcatheter. Wait 1–2 minutes. Avoid aggressive catheter manipulation. If spasm persists, pull microcatheter back slightly and wait. Warm saline flush may help. If spasm is recalcitrant, consider completing embolization from a more proximal position (still distal to cervicovaginal branch) or aborting that side and reattempting at a later date.
Embolic Material Refluxing Proximal to Uterine Artery Origin
Stop injection immediately. The endpoint has been reached or exceeded. Non-target embolization of IIA branches (gluteal, internal pudendal) can cause buttock pain or ischemia. Allow time for flow to re-establish. Confirm on angiogram that no major IIA branches are occluded. Document and monitor for buttock/perineal symptoms post-procedure.
Complications
Common / Expected
- Post-embolization syndrome (virtually universal) — pain, low-grade fever, nausea, malaise lasting 5–7 days; manage with PCA, ketorolac, ondansetron, and supportive care
- Pelvic cramping / pain — most significant in first 24–48 hours; can be severe; adequate pain protocol is essential
- Vaginal discharge — common in weeks following procedure; usually resolves spontaneously
- Transient amenorrhea — may last 1–3 months; menses typically return
Serious Complications
- Fibroid expulsion (~5%) — submucosal fibroids may undergo transcervical passage; can present with pain, bleeding, discharge weeks to months post-procedure; may require GYN intervention
- Infection / endometritis (<2%) — fever >101.5°F persisting beyond 1 week, purulent discharge, elevated WBC; requires antibiotics, possible hospitalization
- Premature ovarian failure — ~1% if <45yo; higher risk if >45yo; due to non-target embolization of ovarian branches via utero-ovarian anastomosis; presents as amenorrhea with elevated FSH
- Amenorrhea (permanent) — rare in patients <40; more common in perimenopausal patients
- Non-target embolization — ovarian artery (premature menopause), gluteal branches (buttock pain/claudication), cervical necrosis (if embolized proximal to cervicovaginal branch)
Pearls & Pitfalls
References & Resources
Standards of Practice
- CIRSE SoP: GYN/Obstetric Hemorrhage → Rand T et al., CVIR Endovascular 2020 · DOI
Primary References
- Spies JB, et al. The EMMY Trial: long-term results of uterine artery embolization vs hysterectomy for treatment of symptomatic uterine fibroids. Am J Obstet Gynecol. 2005;193:1618–1629.
- Goodwin SC, Spies JB, et al. Uterine artery embolization for treatment of leiomyomata: long-term outcomes from the FIBROID Registry. J Vasc Interv Radiol (JVIR). 2008;112:1301–1309.
- Popovic M, et al. Uterine artery embolization for the treatment of symptomatic fibroids: current evidence and future directions. Cardiovasc Intervent Radiol. 2020;43:897–903.
- Stewart JK. Uterine artery embolization for uterine fibroids: a closer look at misperceptions and challenges. Tech Vasc Interventional Rad. 2021;24:100725.
- White AM, Spies JB. Uterine fibroid embolization: update on reported outcomes. Tech Vasc Interventional Rad. 2006;9:2–6.
- Razavi MK, et al. Angiographic classification of ovarian artery-to-uterine artery anastomoses: initial observations in uterine fibroid embolization. Radiology. 2002;224:707–712.