Indications & Contraindications
Indications
- Blunt splenic injury AAST grade III–V with contrast blush or pseudoaneurysm on CTA in hemodynamically stable or stabilizing patients
- Grade I–III with direct vascular injury — contrast extravasation, pseudoaneurysm (PSA), or arteriovenous fistula (AVF) on CTA regardless of grade
- Grade IV–V injuries regardless of direct signs of vascular injury — referred for angiography per WTA algorithm
- Grade I–II without vascular injury — typically observation only
- Proximal vs distal embolization: proximal preferred for multifocal/diffuse injury; distal (superselective) for focal extravasation or PSA; combined for AAST IV–V
Contraindications
- Hemodynamic instability refractory to resuscitation — patient requires emergent splenectomy
- Peritonitis / signs of hollow viscus injury requiring laparotomy
- Grade V shattered spleen without residual vascularization
- Uncorrectable coagulopathy
- Relative: severe contrast allergy (prep with steroids if time permits)
- Relative: inappropriate triage to nonoperative management (up to 40% of major complications from SAE occur in inappropriately triaged patients)
AAST Splenic Injury Grading
| Grade | Hematoma | Laceration / Vascular |
|---|---|---|
| I | Subcapsular, <10% surface area | Capsular tear, <1 cm depth |
| II | Subcapsular 10–50%; intraparenchymal <5 cm | 1–3 cm depth, no trabecular vessel |
| III | Subcapsular >50% or expanding; ruptured | >3 cm depth or involving trabecular vessels |
| IV | — | Segmental/hilar vessel injury, >25% devascularization |
| V | — | Shattered spleen or hilar vascular injury with complete devascularization |
Pre-Procedure Planning
Imaging & Labs
- CTA abdomen/pelvis: review AAST grade, identify contrast blush, PSA, AVF, extent of hemoperitoneum, and associated injuries
- Arterial phase improves sensitivity for active splenic hemorrhage — request if not already obtained
- Evaluate splenic artery origin (celiac trunk vs aortic origin variant ~8%) and degree of tortuosity
- Labs: CBC (serial H/H), PT/INR, PTT, fibrinogen, type & screen / crossmatch
- Lactate, base deficit — markers of resuscitation adequacy
Coordination & Preparation
- Communicate with trauma surgery — confirm patient appropriate for nonoperative management; OR on standby for potential splenectomy
- Activate massive transfusion protocol (MTP) if needed — pRBC, FFP, platelets available in IR suite
- Ensure large-bore IV access ×2, arterial line if hemodynamically borderline
- Consent: risks of splenic infarction, rebleeding, abscess, need for splenectomy, contrast reaction
Relevant Anatomy
Splenic Artery
- Origin: celiac trunk (most common), arising from abdominal aorta at ~T12 level; variant origins include directly off aorta (~8%) or SMA (<1%)
- Course: tortuous path along the superior border of the pancreas toward the splenic hilum
- Dorsal pancreatic artery: first major branch, arises within first few cm of splenic artery origin; connects to transverse pancreatic artery — critical collateral pathway
- Greater pancreatic artery (pancreatica magna): arises more distally; its connection to the dorsal pancreatic artery via the transverse pancreatic artery is the key collateral for proximal embolization
- Terminal branches: polar arteries (superior and inferior) divide into segmental branches at the hilum
Collateral Supply & End-Organ Considerations
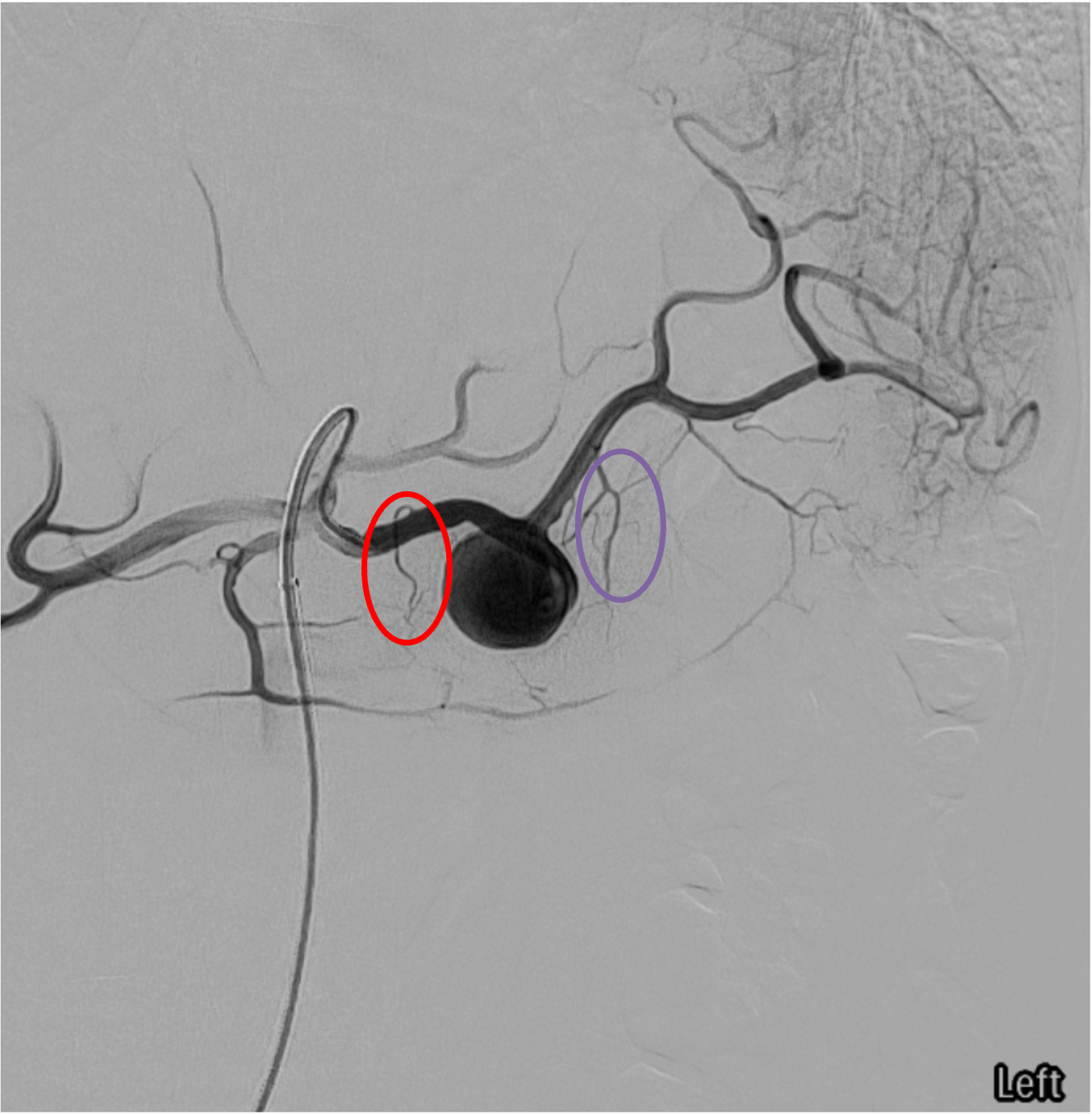
- Short gastric arteries: connect splenic artery to left gastric artery; major collateral pathway maintaining splenic perfusion after proximal embolization
- Left gastroepiploic artery: distal branch of splenic artery; anastomoses with right gastroepiploic artery (from GDA) along the greater curvature of the stomach
- Dorsal pancreatic–transverse pancreatic–greater pancreatic pathway: maintains perfusion distal to proximal coil/plug placement
- Spleen is effectively an end-organ with limited intrinsic collateral — distal embolization carries higher infarction risk than proximal
- Proximal embolization reduces perfusion pressure while preserving splenic function via collateral pathways
Ideal Site for Proximal Embolization
The midsplenic artery, distal to the origin of the dorsal pancreatic artery but proximal to the origin of the greater pancreatic artery, is the ideal location for proximal coil or plug placement. This preserves the dorsal pancreatic–transverse pancreatic–greater pancreatic artery collateral pathway, which along with the short gastric arteries and gastroepiploic arcade, maintains adequate splenic parenchymal perfusion at reduced systolic pressure, allowing the spleen to heal.
Technique
Arterial Access
Celiac Trunk Selection & Angiography
Selective Splenic Artery Catheterization
Strategy Decision: Proximal vs Distal vs Combined
Proximal Embolization Technique
Distal / Superselective Embolization Technique
Completion Angiography & Closure
Community Cards
Landmarks & Key Findings
Angiographic Landmarks
- Celiac trunk: first major anterior branch of abdominal aorta at ~T12/L1; trifurcates into left gastric, common hepatic, and splenic arteries
- Splenic artery origin: typically leftward from celiac trunk; identify tortuosity early for catheter selection
- Dorsal pancreatic artery: first major branch — marks the proximal boundary for safe coil placement
- Greater pancreatic artery: marks the distal boundary — coils placed between these two landmarks
- Short gastric arteries: small branches to gastric fundus; key collaterals visible on post-embolization runs
Signs of Vascular Injury on Angiography
- Active extravasation: contrast blush that persists and increases in density on subsequent phases; irregular, amorphous contrast pooling outside the vascular lumen
- Pseudoaneurysm (PSA): focal, well-circumscribed contrast collection that fills and empties with arterial phase; contained rupture
- Arteriovenous fistula (AVF): early opacification of splenic or portal vein during arterial phase
- Vessel truncation / irregularity: abrupt cutoff or spasm of intrasplenic branches suggesting parenchymal disruption
Troubleshooting
Unable to Advance Catheter to Target
Extremely common in elderly patients. Switch to coaxial microcatheter system (2.4–2.8 Fr) over a shapeable microwire. Consider using a guide catheter or long sheath for additional support. Simmons or SOS catheter may provide better purchase in the celiac trunk. If tortuosity is extreme and proximal embolization is planned, deploy coils where catheter reaches in the midsplenic artery.
Diffuse Extravasation From Multiple Branches
Switch strategy to combined approach: perform proximal embolization (coils/plug in midsplenic artery) to reduce overall perfusion pressure, then superselectively embolize the most active bleeding sites. If too many sites to individually treat, proximal embolization alone may suffice as it reduces pressure to all branches.
Ongoing Extravasation After Initial Embolization
Re-evaluate with repeat angiography. Check for collateral supply feeding the bleeding site (short gastric, gastroepiploic). Consider additional coiling of collateral feeders. If persistent despite repeat embolization, communicate with trauma surgery for conversion to splenectomy. Do not delay surgical intervention for uncontrolled hemorrhage.
Small-Caliber or Spastic Splenic Artery
Vasospasm can mimic small vessel caliber. Administer intra-arterial nitroglycerin 100–200 mcg to relieve spasm. Wait 1–2 minutes and re-image. Use hydrophilic microcatheter and wire to navigate. Avoid excessive catheter manipulation which worsens spasm. If true small caliber, vascular plug may not be deliverable — use coils via microcatheter.
Coil Pack Migrating in High-Flow Splenic Artery
High-flow environment of the splenic artery can lead to coil migration. Anchor first coil securely; slightly oversize coils relative to vessel diameter (1–2 mm larger). Deploy multiple coils to create a dense pack. Consider vascular plug as an alternative — single device often sufficient for proximal occlusion. Gelatin sponge slurry can supplement coils for faster stasis.
Complications
Minor / Expected Complications
- Partial splenic infarction — expected with embolization, especially distal; usually asymptomatic or self-limited LUQ pain; more common with dSAE than pSAE
- Post-embolization syndrome — low-grade fever, LUQ pain, leukocytosis for 2–5 days; supportive management with antipyretics and analgesics
- Pleural effusion — left-sided; usually small and self-resolving; monitor clinically
- Coil/plug migration (minor) — if asymptomatic and does not cause ischemia; monitor
Major Complications
- Rebleeding requiring re-embolization or splenectomy — failure rate ~8% overall; higher for grade IV–V without SAE; repeat angiography + embolization vs surgical rescue
- Splenic abscess (~5%) — infected infarct; presents with fever, LUQ pain, leukocytosis; CT-guided drainage or splenectomy
- Pancreatitis — if coils migrate to or occlude pancreatic branches; monitor lipase
- Overwhelming post-splenectomy infection (OPSI) — if total splenic infarction results in functional asplenia; encapsulated organisms (Streptococcus pneumoniae, Neisseria meningitidis, H. influenzae); lifelong risk; requires vaccination
- Access-site complication — pseudoaneurysm, hematoma at CFA puncture site
Pearls & Pitfalls
References & Resources
Proximal vs Distal Approach Summary
- Proximal: shorter procedure time, lower minor complication rate, reduces perfusion pressure, preserves collateral flow; cannot easily re-embolize distally
- Distal: selective, treats focal injury, higher minor complication rate (infarction, abscess); can use coils, particles, Gelfoam, NBCA
- Both techniques show similar clinical efficacy for splenic salvage
Primary References
- Haan JM, Bochicchio GV, Kramer N, Scalea TM. Nonoperative management of blunt splenic injury: a 5-year experience. J Trauma. 2005;58(3):492–498.
- Bhullar IS, Frykberg ER, Siragusa D, et al. Selective angiographic embolization of blunt splenic traumatic injuries in adults decreases failure rate of nonoperative management. J Trauma Acute Care Surg. 2012;72(5):1127–1134.
- Habash M, Ceballos D, Gunn AJ. Splenic artery embolization for patients with high-grade splenic trauma: indications, techniques, and clinical outcomes. Semin Intervent Radiol. 2021;38(1):105–112.
- Banerjee A, Duane TM, Wilson SP, et al. Trauma center variation in splenic artery embolization and spleen salvage: a multicenter analysis. J Trauma Acute Care Surg. 2013;75(1):69–74.
- Schnuriger B, Inaba K, Konstantinidis A, et al. Outcomes of proximal versus distal splenic artery embolization after trauma: a systematic review and meta-analysis. J Trauma. 2011;70(1):252–260.
- Arvieux C, Frandon J, Tidadini F, et al. Splenic arterial embolization to avoid splenectomy (SPLASH) study group. JAMA Surg. 2020;155(12):1102–1111.