Indications & Contraindications
Indications
- Acute osteoporotic VCF with significant height loss (>30%) — VADs (including SpineJack) emerged specifically for cases of significant vertebral height loss where balloon kyphoplasty is inadequate
- Young patients where durable height restoration is desired — titanium implant remains in situ indefinitely; balloon kyphoplasty deflates before cement, allowing partial height recollapse
- Traumatic fractures — SpineJack indicated for compression fractures from osteoporotic, traumatic, and tumor-related causes; level range T5–L5
- Primary or secondary bone tumors — SpineJack can be used with appropriate oncologic indication alongside cementoplasty for tumor stabilization
- Cases where kyphosis correction is a priority — SpineJack provides up to 40% height gain and significant kyphotic angle reduction (mean 11–24 degrees in published series)
Contraindications
- Absolute: Retropulsed fragments causing neurologic deficit (surgery) · Active spinal infection / osteomyelitis · Uncorrectable coagulopathy · PMMA allergy
- Relative: Posterior wall disruption with canal compromise — device expansion may worsen retropulsion · Spinal instability requiring surgical fixation · Severe osteoporosis with very low bone density (device may not achieve purchase)
- OsseoFix absolute contraindication note: Retropulsed fragments and dural sac/cord compression are absolute contraindications for OsseoFix — similar caution applies to all VADs
SpineJack vs. Balloon Kyphoplasty — Key Advantages
- Durable height restoration: SpineJack remains in situ permanently — balloon deflation before cement in kyphoplasty allows partial cavity collapse; SpineJack maintains full expansion
- Mechanical (not hydraulic) expansion: Craniocaudal expansion is controlled mechanically — provides predictable force application; balloon expansion is hydraulic and less directional
- Superior height restoration and kyphosis correction: Preliminary and multicenter studies (Noriega et al, SAKOS study, Spine J 2019) show SpineJack non-inferior to kyphoplasty for pain with superior height restoration
- 81.5% pain relief at 48 hours in Noriega et al registry study (n=108); 90%+ decrease in analgesia use at 12 months
- Cost: Most expensive of the three options; requires implant-specific instrumentation
Pre-Procedure Checklist
Relevant Anatomy
Transpedicular / Parapedicular Access
- Bilateral approach mandatory: SpineJack requires bilateral implant placement — two cannulae, two devices, simultaneous expansion
- Standard transpedicular approach (pedicle width ≥4–5 mm); parapedicular approach for narrower pedicles
- Level range: T5–L5 (VBS system); SpineJack generally applied T4–L5
- Working cannula (implant-specific trocar) advanced to posterior one-third of vertebral body — confirmed on lateral fluoroscopy
SpineJack Device Anatomy
- Titanium implant, 5 mm diameter in collapsed state; expands craniocaudally via mechanical compression of the device handle
- Expansion creates a cavity in the vertebral body within which SpineJack resides — analogous to balloon kyphoplasty cavity but implant remains
- Mechanical (not hydraulic) expansion = operator-controlled, directional craniocaudal force — compacts trabecular bone vertically
- PMMA injected through the working cannula simultaneously on both sides, filling the cavity around the in-situ implant
- Device locked in expanded position before cement injection — ensures height is maintained during cementing
Drill Path & Template
- After working cannula placed at posterior one-third of vertebral body: inner trocar replaced with guidewire, then bone drill (reamer)
- Drill advances to desired implant position — terminates just short of the anterior vertebral body cortex
- Drill removed; template device "cleans" the desired implant site and confirms dimensions
- Process repeated on contralateral side before any device insertion
- Both implant expanders inserted through working cannulae, expanded simultaneously under fluoroscopy
Technique
Supplies
Steps — SpineJack Vertebral Augmentation
Prophylactic antibiotics & positioning
Bilateral pedicle entry — working cannulae
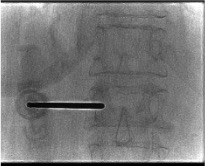
Guidewire and bone drill (reamer)
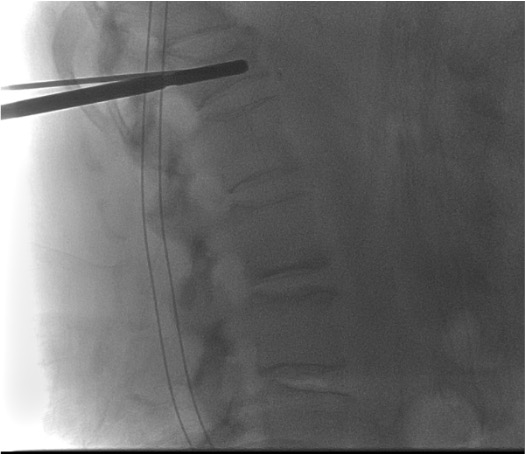
Template device — site preparation
Insert SpineJack expanders bilaterally
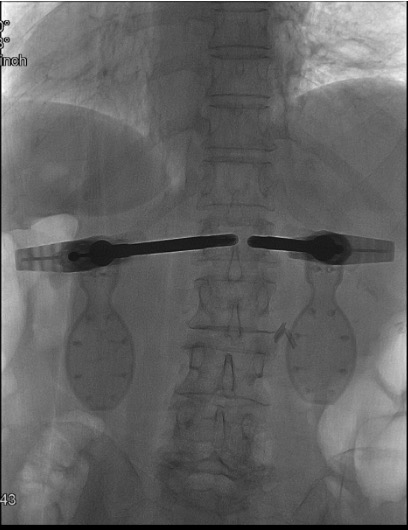
Simultaneous mechanical expansion — height restoration
Lock devices in expanded position
Simultaneous PMMA cement injection
Cannula removal & final imaging
Vertebral Body Stenting (VBS) System — Alternative VAD
- VBS (DePuy Synthes): Expandable titanium stent deployed using the same balloon as conventional kyphoplasty — stent remains expanded in place after balloon removal (unlike conventional kyphoplasty where only cement remains)
- Bipedicular approach; suitable for T5–L5 compression fractures
- After balloon inflation and stent expansion: balloon is removed but stent stays — PMMA injected through same cannula into stent cavity
- Evidence shows greater vertebral height reduction after VBS vs. standard kyphoplasty (stent holds height after balloon removal)
- Significant pain reduction and height restoration maintained at 12 months in published series
- Mechanism: Hydraulic (balloon-based) vs. SpineJack mechanical — both leave permanent implant; different engineering approach
Troubleshooting
Device does not expand / insufficient height gain
Likely cause: Chronic fracture with sclerotic bone, very dense trabecular architecture, or device positioned in wrong location (e.g., in the fracture endplate rather than in trabecular bone).
Next step: Confirm device tip position on lateral fluoroscopy — ensure both implants are within trabecular bone at the same level and aimed at the fracture cleft. Attempt additional expansion force. If maximum expansion reached without adequate height gain: lock device in partially expanded position and proceed with cement. Partial restoration still provides pain relief and structural stabilization.
Asymmetric expansion — one device advances faster
Likely cause: Asymmetric fracture morphology, one device in denser bone, or slight difference in starting positions.
Next step: Monitor both devices simultaneously on AP fluoroscopy during expansion. Slow the advancing device and apply more force on the lagging side to achieve symmetric height restoration. Small asymmetries are acceptable — do not force symmetric expansion at the cost of superior end-plate breach.
Cement leak around device
Likely cause: Posterior cortical breach, cement injected at too low viscosity, or fracture line extending to posterior cortex.
Next step: Stop injection immediately. Wait for polymerization at leak site (1–2 min). Resume cautiously if leak self-seals. The SpineJack implant should provide additional containment vs. vertebroplasty — persistent posterior leak despite locked implant suggests unrecognized posterior wall compromise. Post-procedure CT mandatory.
Superior end-plate breakthrough during expansion
Likely cause: Excessive expansion force, device positioned too cranially, or weakened end-plate from tumor/osteoporosis.
Next step: Stop expansion immediately. Lock device at current position. Assess extent of end-plate disruption on fluoroscopy. Intradiscal cement filling is a risk during subsequent cement injection — monitor carefully and stop if intradiscal filling seen on AP fluoroscopy. Post-procedure CT will characterize extent of end-plate involvement.
Complications
Implant-Related Complications
- Device malposition: Implant too posterior (canal risk) or too anterior (cortical breach) — confirmed on post-procedure CT; surgical removal in rare symptomatic cases
- Implant migration: Rare with proper technique; risk higher in very osteoporotic bone
- Infection (deep implant infection): Rare but serious; prophylactic antibiotics mandatory. Requires prolonged antibiotics or explantation if confirmed
- End-plate disruption: Aggressive expansion may breach end plate; intradiscal cement risk
Cement & Procedure Complications
- Cement extravasation: Epidural, foraminal, intradiscal, venous/PE — same spectrum as vertebroplasty and kyphoplasty; reduced but not eliminated by implant containment
- Adjacent vertebral fracture: Stiffened augmented level increases adjacent level stress — counsel patients
- Failed height restoration: Chronic/sclerotic fractures; partial restoration still provides pain relief
- Neurologic injury: Rare; from cement epidural leak or device malposition — emergent surgical consult if new deficit
Emergent Escalation Triggers
- New neurologic deficit post-procedure → emergent spine surgery consult + CT spine
- Hypoxia / chest pain post-procedure → CT chest angiography (cement PE)
- Fever + back pain after 48–72h → CBC, CRP, blood cultures; consider implant infection
- Severe unremitting pain escalation → CT spine (epidural collection or device malposition)
Post-Procedure Care
Recovery & Monitoring
- Recover supine 1–2 hours; complete neurologic exam before discharge
- Ambulation on post-procedure day 1 — demonstrated in published SpineJack case series
- Pain score documented at 24–48 hours; expected significant improvement
- Case series: pain reduction from 6.5/10 pre-procedure to 1/10 post-procedure day 1; kyphotic angle reduced by 11–24 degrees
- Most patients discharged same day or post-procedure day 1 (institutional preference)
Imaging & Follow-up
- Post-procedure CT to confirm implant positioning, height restoration, and cement distribution — mandatory for implant procedures
- Upright weight-bearing AP and lateral X-ray at 1 month to confirm maintained height (key advantage of SpineJack: height durability)
- Resume anticoagulation: 24 hours post-procedure
- Osteoporosis management: bisphosphonates, denosumab, or teriparatide — essential to prevent adjacent fractures
- Physical therapy referral for core strengthening and fall prevention
- Report fever, new neurologic symptoms, or new acute back pain immediately
Critical Pearls
Related Resources
Vertebral Augmentation Comparison
| Feature | Vertebroplasty | Balloon Kyphoplasty | SpineJack |
|---|---|---|---|
| Mechanism | Direct cement injection | Balloon inflation creates cavity, then cement | Titanium implant expands craniocaudally, then cement |
| Height restoration | Minimal | Moderate; partial recollapse possible | Superior — up to 40%; durable (implant permanent) |
| Cement leak risk | Highest (direct injection into cancellous bone) | Lower (contained cavity) | Low (implant scaffold contains cement) |
| Sedation | MAC / Local | MAC / General | General / MAC |
| Antibiotics | Not routine | Not routine | Cefazolin 1g IV — required |
| Implant remains | No | No (balloon removed) | Yes — titanium; permanent |
| Cost | Lowest | Moderate | Highest |
| Best for | VCF without significant height loss; malignant fractures; low-cost centers | Fractures with >15% height loss; intact posterior wall | Significant height loss; young patients; durable restoration; tumor cases |
| Follow-up imaging | CT (optional) | CT + upright XR at follow-up | CT (mandatory) + upright XR at 1 month |
Vertebral Augmentation Devices (VADs) Overview
| Device | Manufacturer | Mechanism | Level Range | Notes |
|---|---|---|---|---|
| SpineJack | Stryker Corp. | Mechanical — craniocaudal expansion | T4–L5 | SAKOS trial (non-inferior to BKP, superior height restoration) |
| VBS (Vertebral Body Stenting) | DePuy Synthes | Hydraulic (balloon) — stent expands, balloon removed | T5–L5 | Greater height retention than standard BKP after balloon removal |
| OsseoFix | Alphatec Spine | Mechanical compression of trabecular bone via titanium mesh | T6–L5 | Less PMMA required; interlocking bone-titanium effect; absolute CI: retropulsed fragments |
Primary Reference
Prologo JD, Ray CE Jr., eds. Advanced Pain Management in Interventional Radiology: A Case-Based Approach. Thieme; 2024. DOI: 10.1055/b000000387
Chapter 18 (Dalili et al — Osteoporotic Fracture III: Vertebral Augmentation Devices)
Additional references: Noriega et al, Spine J 2019 (SAKOS study — SpineJack vs. BKP) · Noriega et al, BioMed Res Int 2015 (108 SpineJack registry, 1-year results) · Vanni et al, J Spine Surg 2016 (third-generation VADs) · Werner et al, J Bone Joint Surg Am 2013 (VBS vs. kyphoplasty RCT) · Disch & Schmoelz, Spine 2014 (VBS augmentation model)