Indications & Contraindications
Indications
- Renal hemorrhage — trauma (blunt/penetrating), post-biopsy bleeding, post-surgical hemorrhage
- Angiomyolipoma (AML) >4 cm — prophylactic embolization to prevent rupture; emergent for acute hemorrhage (rupture risk ~50% when ≥4 cm)
- Pseudoaneurysm — post-biopsy, post-partial nephrectomy, post-nephrostomy
- Pre-operative devascularization — large RCC before nephrectomy to reduce intraoperative blood loss
- Arteriovenous fistula / malformation — post-biopsy AVF, congenital AVM with hematuria or hypertension
- Renal artery aneurysm — ≥1.5–2 cm, symptomatic, women of childbearing age, polyarteritis nodosa
- Palliative — unresectable renal tumors (pain, hematuria control)
- End-stage renal disease — polycystic kidneys with intractable pain/hematuria for transplant prep; irreversible transplant rejection
Contraindications
- Uncorrectable coagulopathy (INR >1.5, platelets <50K) — relative in emergent hemorrhage
- Severe contrast allergy without adequate premedication
- Renal artery stenosis as cause of symptoms (better treated with angioplasty)
- Relative: solitary kidney — superselective embolization mandatory; maximize nephron preservation
- Relative: severely impaired renal function — risk/benefit discussion; hydration critical
- Relative: pregnancy — radiation exposure considerations
Embolic Agent Selection by Indication
| Indication | Preferred Embolic | Technique |
|---|---|---|
| Pseudoaneurysm / AVF | Coils (microcoils) | Superselective — coil across neck or feeding artery |
| AML (elective) | Coils + microspheres/PVA | Superselective to feeding artery |
| Trauma / acute hemorrhage | Coils ± Gelfoam | Coils to injured segmental artery; Gelfoam for temporary occlusion |
| Tumor devascularization (RCC) | PVA / microspheres + coils | Particles first (arteriolar level), then coil main feeders |
| AVM (complex) | Ethanol / NBCA glue ± coils | Sclerosant to ablate nidus; coils to reduce flow first |
| Total embolization (ESRD) | Ethanol/PVA + coils | Peripheral particles first, then coils in main renal artery |
Pre-Procedure Planning
Imaging & Labs
- CTA abdomen — identify bleeding source, renal arterial anatomy, number of renal arteries (accessory arteries in 25–30%), AML vascularity, aneurysm morphology
- Assess for replaced/aberrant renal arteries and collateral supply
- Labs: Creatinine / GFR (baseline renal function), CBC, PT/INR, platelets
- Type & screen — recommended for hemorrhage cases
Patient Preparation
- Hydration — especially critical if solitary kidney or transplant kidney; 1 L NS IV before start
- NPO 6h before (moderate sedation)
- IV access; Foley catheter for prolonged cases or total embolization
- Consent: discuss renal function impact (infarction risk), post-embolization syndrome (pain, fever, nausea), non-target embolization risk
Relevant Anatomy
Renal Arterial Supply
- Renal arteries arise from the aorta at L1–L2 level; right renal artery courses posterior to IVC
- Anterior division — supplies upper, middle, and lower anterior segments (4 segmental arteries)
- Posterior division — supplies posterior segment (single segmental artery)
- Segmental arteries are end-arteries — no collaterals between segments; any embolization causes infarction of the supplied territory
- Segmental → interlobar → arcuate → interlobular arteries
Variants & Key Points
- Accessory renal arteries present in 25–30% of population — must identify on CTA/aortogram; may supply bleeding territory
- Early branching of main renal artery (prehilar branching) in ~10% — may require multiple selective catheterizations
- AML vascularity: dysplastic, tortuous feeding arteries with microaneurysms; characteristic “onion skin” appearance of abnormal vessels
- Pseudoaneurysms: focal contrast pooling on angiogram; contained rupture of arterial wall — often at biopsy tract
Angiographic Landmarks
On flush aortogram, identify renal artery origins at L1–L2 (tennis racket or Omni-Flush catheter positioned with tip just above renal artery origins). Selective renal angiogram defines anterior/posterior division and segmental branches. In the nephrographic phase, assess for parenchymal defects (prior infarcts), mass effect (tumor), and early venous filling (AVM/AVF). Superselective catheterization into the feeding segmental or interlobar artery using microcatheter allows precise identification of the target lesion before embolization.
Supplies & Setup
Access & Catheters
- 5F vascular sheath (6–8F if balloon-assisted technique)
- 5F renal curve catheter — C2 Cobra (primary), SOS Omni, or RC-2
- Microcatheter (2.4–2.8F) + 0.018″ microwire for superselective access
- Flush catheter for aortogram (Tennis Racket or Omni-Flush)
- 0.035″ guidewire (angled Glidewire, Bentson)
- Standard access kit: micropuncture set, lidocaine 1%
Embolic Agents
- Coils: pushable and/or detachable (0.018″ microcoils for superselective; 0.035″ for main artery)
- Microspheres / PVA: 300–900 μm particles for tumor devascularization and AML
- Gelfoam: pledgets or slurry for temporary occlusion (benign hemorrhage, pre-nephrectomy)
- Ethanol 98–99%: for sclerosis of AVM nidus or total renal ablation (use with balloon occlusion)
- NBCA glue: for high-flow AVM when rapid occlusion needed
Medications
- Moderate sedation: midazolam 1–2 mg + fentanyl 50–100 mcg IV
- Nitroglycerin 100–200 mcg IA (for vasospasm)
- Heparinized saline flush
- Cefazolin 1g IV (if tumor/abscess risk)
- IV fluid: NS at 100–150 mL/h during and post-procedure
- Ketorolac 15–30 mg IV PRN (post-embolization pain)
Procedure Steps
Vascular Access
Flush Aortogram
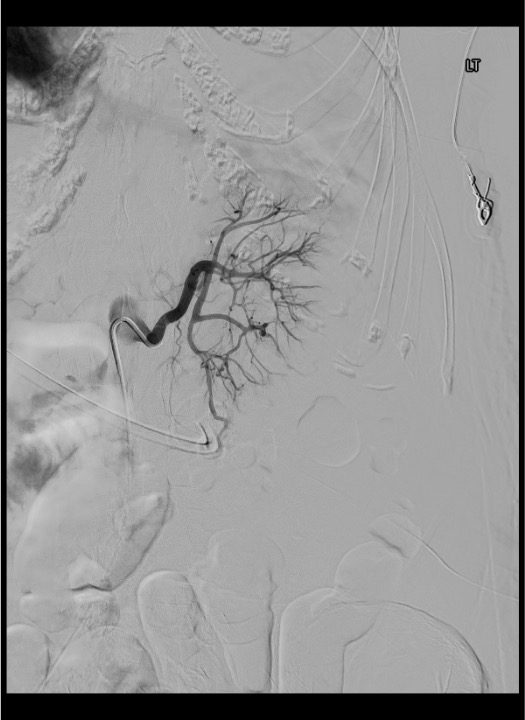
Selective Renal Artery Catheterization
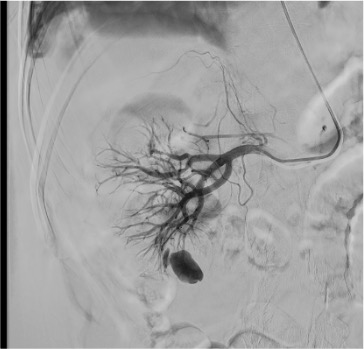
Superselective Catheterization
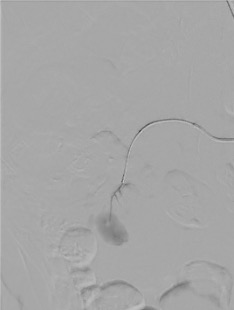
Embolization
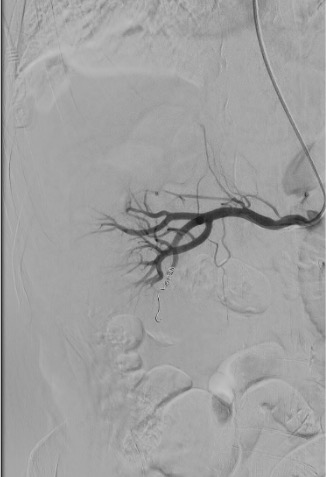
Completion Angiogram
Sheath Removal & Hemostasis
Community Cards
Troubleshooting
Renal Artery Spasm During Catheterization
Common with wire/catheter manipulation in renal artery branches. Administer nitroglycerin 100–200 mcg IA through the catheter. Wait 1–2 minutes for resolution. Avoid forceful catheter advancement during spasm. If persistent, allow catheter to sit without manipulation for several minutes.
Bleeding Territory Supplied by Accessory Renal Artery
If bleeding persists after embolization of the main renal artery branches, suspect an accessory renal artery supplying the hemorrhage territory. Return to aortogram to identify additional renal arteries. Accessory arteries (present in 25–30%) may arise from the aorta at any level from T12 to the iliac bifurcation. Selectively catheterize and embolize the accessory artery feeding the lesion.
Cannot Advance Microcatheter to Target
Ensure stable 5F guide catheter position in the main renal artery. Use a hydrophilic microwire (0.014–0.018″) to navigate tortuous or spastic segmental branches. Consider catheter exchange to a reverse-curve catheter (SOS) for better purchase. In difficult anatomy (large tumor mass effect, aortic aneurysm), try alternative catheter shapes (Simmons, RC-2). If vessel too small for microcatheter, consider particle embolization from a more proximal position.
Embolic Material Reflux into Non-target Vessels
Stop embolization immediately. Inject slowly and under continuous fluoroscopy. Ensure catheter is positioned distal enough to prevent reflux. For high-flow lesions (AVM), consider balloon occlusion of the parent artery during embolization to prevent reflux. Use appropriately sized coils (oversize by 20–30% relative to vessel diameter). If coil migrates, retrieve with snare device.
Continued Bleeding After Embolization
Repeat angiogram from the main renal artery to reassess. Evaluate for: incomplete embolization of target, collateral supply from lumbar or capsular arteries, accessory renal artery, or new bleeding site. Deploy additional embolic material as needed. In refractory cases, consider more proximal embolization (accepting greater parenchymal loss) or surgical consultation.
Complications
Common / Expected
- Post-embolization syndrome (>90% with total embolization) — flank pain, fever, nausea/vomiting, elevated WBC; onset 1–3 days post-procedure; self-limited over several days; treat with analgesics, antipyretics, antiemetics
- Renal infarction — expected consequence of segmental artery embolization (end-arteries); superselective technique limits infarction to <10% of parenchyma in best cases
- Access site complications — groin hematoma (<2%), pseudoaneurysm; manual compression or closure device
Serious Complications
- Non-target embolization — reflux of embolic material into aorta causing spine, lower extremity, or bowel infarction; prevented by proper catheter positioning and slow injection
- Renal function loss — clinically significant with subselective embolization (up to 15–50% parenchymal infarction); rarely requires dialysis unless solitary kidney
- Abscess formation — infected infarct; low incidence; prophylactic antibiotics for tumor embolization
- Coil migration — occurs in <2% of cases; retrievable with endovascular snare
- Incomplete embolization — recurrent bleeding; usually apparent on completion angiogram; additional embolization required
Pearls & Pitfalls
References & Resources
Key Technical Points
- Superselective embolization results in <10% non-target parenchymal infarction
- Technical success rate for emergent renal artery embolization: ~93%
- Incomplete embolization and coil migration occur in <2% of cases
- Recanalization of embolized arteries is rare with current embolic agents
Primary References
- Ginat DT, Saad WEA, Turba UC. Transcatheter renal artery embolization: clinical applications and techniques. Tech Vasc Interventional Rad. 2009;12:224–239.
- Sauk S, Zuckerman DA. Renal artery embolization. Semin Intervent Radiol. 2011;28(4):396–406.
- Schwartz MJ, Smith EB, Trost DW, Vaughan ED Jr. Renal artery embolization: clinical indications and experience from over 100 cases. BJU Int. 2007;99(4):881–886.
- Jain S, et al. Renal angiomyolipoma embolization: a systematic review. Cardiovasc Intervent Radiol. 2018;41(10):1485–1498.
- Somani BK, Nabi G, Thorpe P, et al. Image-guided biopsy-diagnosed renal cell carcinoma: critical appraisal of technique and long-term follow-up. Eur Urol. 2007;51(5):1218–1228.