Indications / Contraindications
Indications
- Malignant biliary obstruction (pancreatic cancer, cholangiocarcinoma, HCC, metastatic disease, lymphoma)
- Failed or unsuitable ERCP — post-surgical anatomy, complete obstruction, duodenal obstruction
- Pre-op biliary decompression (controversial; used in select centers)
- Relief of jaundice for chemotherapy eligibility
- Cholangitis from malignant obstruction (urgent)
- Biliary stent dysfunction after prior ERCP stent
Contraindications
- Absolute: Uncorrectable coagulopathy
- Absolute: Massive uncontrolled ascites (relative — transperitoneal approach riskier)
- Relative: Multiple intrahepatic obstructions precluding adequate drainage
- Relative: Diffuse hepatic replacement by tumor (minimal functional liver)
- Relative: Short life expectancy (<1 month) — risk/benefit discussion essential
Malignant Causes by Frequency
- Pancreatic head cancer (~40%): Ampullary region obstruction — classic "double duct sign" on MRCP
- Hilar cholangiocarcinoma (Klatskin tumor): Confluence obstruction → bilateral intrahepatic duct involvement; classified by Bismuth system
- HCC with biliary invasion: Hemobilia and jaundice common
- Colorectal / gastric mets to hepatic hilum: Portal lymphadenopathy pattern
- Lymphoma (periportal nodes): Often responds to chemotherapy — drainage may be temporary
Bismuth Classification (Hilar Tumors)
- Type I: Below CHD confluence — unilateral drainage usually sufficient
- Type II: At confluence — attempt to cross both ducts
- Type IIIa/IIIb: Extends into right or left duct — ipsilateral drainage; contralateral if possible
- Type IV: Bilobar involvement — bilateral drainage rarely provides full benefit; prioritize functional liver volume
Pre-Procedure Checklist
Relevant Anatomy
Biliary Tree Overview
- Right hepatic duct (RHHD) from right liver + left hepatic duct (LHD) from left liver → common hepatic duct (CHD) → cystic duct joins → common bile duct (CBD) → ampulla of Vater (drains with pancreatic duct into duodenum)
- Hilar confluence = junction of R + L hepatic ducts. Primary target in Klatskin tumors.
- Segment II/III ducts (left system) are most peripheral, easily visualized from subxiphoid approach, and provide the best angle for crossing hilar/pancreatic head obstructions
Left Hepatic Approach (Preferred)
- Subxiphoid or subcostal skin entry
- Target segment II or III ducts — more peripheral, predictably accessible
- More horizontal angle than right approach — better alignment to cross obstruction into duodenum
- Lower pneumothorax risk — does not traverse intercostal space or pleura
- Preferred for hilar cholangiocarcinoma and pancreatic head obstruction
Right Hepatic Approach
- Right anterior axillary line, 8th–10th intercostal space entry (stay below the 10th rib and anterior to the midaxillary line to minimize the chance of traversing the pleural space). Pleural reflections are at the 8th rib anteriorly, 10th rib laterally, and 12th rib posteriorly.
- AP + oblique fluoroscopic views for targeting
- Pneumothorax risk if above 8th rib — confirm rib level carefully
- Used for isolated right-system obstruction or when left approach fails/not feasible
- Gallbladder and hepatic flexure of colon are nearby — avoid
Danger Structures
- Right approach: Pleura (above 10th rib), gallbladder, hepatic flexure of colon
- Left approach: Stomach (left lobe is anterior to stomach — confirm on US/fluoro), hepatic artery, portal vein branches
- Both approaches: Hepatic artery and portal vein — injuries cause hemobilia and pseudoaneurysm. Recognize hepatic artery access on fluoroscopy by pulsatile contrast flow.
- Fluoroscopic access recognition: Bile duct = slow central contrast column; portal vein = rapid central dilution; hepatic artery = pulsatile flow
Technique
Default RadCall approach · share your own below
Supplies
Internal-External vs. External-Only Drainage
Internal-External — sideholes above AND below obstruction, tip in duodenum
- Drains both to bag AND internally into duodenum
- Bile enters GI tract → physiologically superior
- Requires crossing the obstruction
- Can be capped once patient tolerating oral intake
External Only — all bile drains to external bag
- Used when obstruction cannot be crossed
- All bile to bag — requires bile replacement orally
- Acceptable initial step; reattempt crossing at 48-72h after decompression
Steps
Review and plan
Fluoroscopic setup
Initial needle access
Confirm bile duct access
Cholangiogram
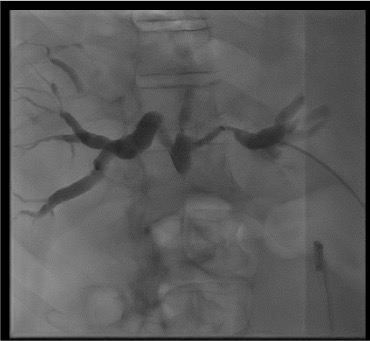
Wire and dilator
Cross the obstruction
Catheter positioning
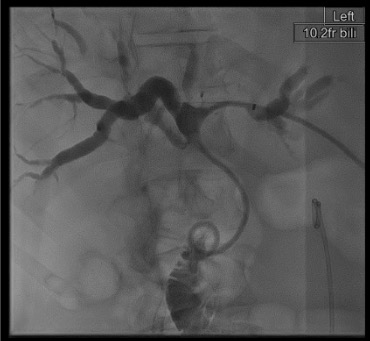
Secure and complete
Troubleshooting
Cannot access a dilated duct
Likely cause: Needle not in duct, or ducts not adequately dilated at the puncture site. Peripheral ducts are smaller and easier targets than central ducts.
Next step: Use ultrasound guidance for initial needle targeting — real-time US dramatically improves first-pass success rate. Target more peripheral ducts. Confirm breath-hold technique with patient.
Cannot cross the obstruction
Likely cause: Tight or complete malignant stricture, wrong angle of approach, or distended upstream system making navigation difficult.
Next step: Consider balloon dilation of the duct and aggressive wire work. External drainage only is an acceptable initial step — decompress the system for 48-72 hours, then return. A decompressed system is significantly easier to navigate. Changing approach (left ↔ right) may improve angle.
Hemobilia (blood in drainage bag)
Likely cause: Hepatic artery branch injury during access. Small amounts common. Bright red pulsatile output is concerning for pseudoaneurysm.
Next step: Small amounts usually resolve with drainage catheter in place (tamponades the tract). If persistent bright-red pulsatile output → urgent CT angiography → hepatic artery embolization. Never remove the drain prematurely if hemobilia is present.
Cholangitis / Sepsis post-procedure
Likely cause: Bile contamination into bloodstream from overdistension during cholangiogram or from underlying cholangitis at the time of drainage.
Next step: Blood cultures, broaden antibiotics, ensure drain is patent and draining freely. Vigorous IV fluid resuscitation. ICU if hemodynamically unstable. Confirm drain position and patency with cholangiogram once patient stable.
Bilateral obstruction — Klatskin tumor
Likely cause: Bismuth III-IV hilar cholangiocarcinoma obstructing both right and left hepatic duct systems independently.
Next step: May need two separate drains (left + right system). Ideally placed same session if patient tolerates; otherwise staged 48 hours apart. Limit to draining the two ducts providing the most functional liver volume. Bismuth IV bilateral drainage rarely achieves meaningful benefit — limit attempts.
Complications
Immediate / Periprocedural
- Sepsis / cholangitis (2-10%) — leading cause of procedural death; prophylaxis is critical; do not overdistend the biliary system
- Hemorrhage (1-3% major) — hemobilia; hepatic artery pseudoaneurysm → coil embolization
- Bile peritonitis / leak (<1%) — catheter malposition or inadequate seal
- Pleural complication — hemothorax or biliothorax, especially right intercostal approach
- Vasovagal — monitor during and immediately after
Delayed
- Drain occlusion — most common delayed complication; scheduled exchange every 3 months
- Recurrent cholangitis episodes — common with indwelling drain; requires periodic exchange
- Tumor progression — drain dysfunction from tumor ingrowth into metal stent or external compression
- Catheter dislodgement — patient education on drain care is essential
- Mortality — overall 0.1-0.8%; higher in cholangitis and poor performance status
Post-Procedure Care
Immediate Monitoring
- Vitals q30 min × 2h; biliary drainage output monitoring
- Initial drainage: bile may be thick/sludgy — normal. Dark green-brown = good bile flow
- Watch for fever, rigors, hypotension in first 4h — signs of procedure-induced cholangitis
- Day 2 cholangiogram: confirm catheter position and function
Bile Replacement
- If external drainage only: replace up to 500 mL/day orally (this is not nice to do to patients)
- Bile salts important for fat absorption and production of fat-soluble clotting factors (II, VII, IX, X)
- Patients on external-only drainage are at risk for progressive coagulopathy without bile replacement
- Track daily output volume — expect 200-800 mL/day
Metallic Stent Considerations
- For selected malignant cases: life expectancy >3 months, crossing achieved, patient prefers freedom from exchanges
- Uncovered self-expanding metal stent (SEMS): longer patency (~6-12 months); tumor ingrowth possible; not removable
- Covered SEMS: reduces ingrowth; potentially removable; higher migration risk
- Metal stent placement removes need for scheduled exchange but requires return for re-intervention if dysfunctional
- Oncology coordination: Some chemotherapy regimens require bilirubin normalization — track labs closely after drainage
Critical Pearls
Biliary Drain Management
| Parameter | Normal / Target | Action / Notes |
|---|---|---|
| Drainage color | Light yellow-green to dark green-brown | Dark = concentrated (normal early); bloody = concerning; frankly bloody pulsatile = urgent CT angio |
| Output volume | 200-800 mL/day expected | Less than 100 mL = possible occlusion (flush, check position); more than 1000 mL = high-output (replace electrolytes) |
| Flushing | 10 mL sterile saline twice daily | Nursing order required; maintains catheter patency; prevents sludge occlusion |
| Capping (internal drainage) | Once adequate oral intake established | Close external drainage valve to promote physiologic bile flow into duodenum; monitor for pain/fever suggesting obstruction |
| Dressing | Every 3-5 days or when soiled | Inspect entry site for erythema, bile leak, or signs of skin breakdown |
| Drain exchange | Every 3 months (sooner if occluded or infected) | Scheduled IR follow-up; cholangiogram at exchange to reassess anatomy |
| Signs of dysfunction | — | Abdominal pain, fever, returning jaundice, change in output volume/color, drain site erythema or leak |
| Lab follow-up | Bilirubin trending down over 1-2 weeks | For chemotherapy eligibility: target bilirubin normalization; coordinate with oncology |
References & Resources
Key Guidelines
- SIR Standards of Practice for Biliary Interventions
- ESGAR Guidelines for Biliary Drainage
- CIRSE SoP: Biliary Drainage & Stenting → CIRSE Standards of Practice Committee
Primary References
- Becker CD et al. Percutaneous palliation of malignant obstructive jaundice. AJR Am J Roentgenol. 1989;152(2):249-256.
- Covey AM, Brown KT. Percutaneous transhepatic biliary drainage. Tech Vasc Interv Radiol. 2008;11(1):14-20.
- Murata S et al. Decompression of malignant biliary obstruction. J Vasc Interv Radiol. 2011;22(7):946-953.