Indications & Contraindications
Indications
- Postpartum hemorrhage refractory to uterotonics (oxytocin, methylergonovine / methergine, carboprost / hemabate) and uterine massage / tamponade (Bakri balloon)
- Massive PPH: defined as >1000 mL blood loss after delivery or any amount causing hemodynamic instability
- Primary PPH (within 24 h of delivery) or secondary PPH (>24 h post-delivery)
- Patient desire for future fertility — embolization as alternative to emergent hysterectomy
Contraindications
- Hemodynamic instability requiring emergent surgical intervention — patient too unstable for angiographic procedure; proceed to hysterectomy
- Anaphylaxis to iodinated contrast (relative — CO2 angiography may be used for uterine source)
- No absolute contraindications to embolization of PPH per literature
- Relative: unmanaged severe coagulopathy / DIC (known predictor of clinical failure)
Etiologies — The 4 Ts of PPH
| Etiology | Examples | Frequency |
|---|---|---|
| Tone | Uterine atony — macrosomia, polyhydramnios, prolonged labor, general anesthesia | Most common |
| Tissue | Retained products of conception, abnormal placentation (accreta spectrum) | Common |
| Trauma | Cervical / vaginal laceration, uterine rupture, uterine inversion | Moderate |
| Thrombin | DIC, coagulopathy (pre-existing or acquired), amniotic fluid embolism | Less common |
Pre-Procedure Planning
Resuscitation & Labs
- Massive transfusion protocol (MTP) — likely already activated; confirm with blood bank
- Type & screen / crossmatch (usually already done); if transfusion needed before crossmatch, use type-specific uncrossmatched blood
- Coag panel: PT/INR, fibrinogen, platelets — assess for DIC (DIC is a predictor of embolization failure)
- CBC, BMP (creatinine for contrast), blood gas analysis
- Hemodynamic stabilization: IV fluids wide open, pressors if needed, secure IV access ×2 (consider central line placement by IR)
Coordination & Imaging
- OB consultation: obtain obstetric history — mode of delivery (vaginal vs C-section), suspected bleeding source, prior interventions (uterotonics, Bakri balloon, O’Leary stitch, IIA ligation)
- Patient desire for future fertility — confirm directly or via OB team
- CTA pelvis if patient stable — identifies extravasation site, variant anatomy, ovarian collaterals
- Do NOT delay for CTA if patient is hemodynamically unstable — proceed directly to angiography
- Foley catheter placement (may already be in place)
Relevant Anatomy
Uterine Artery
- Arises from the anterior division of the internal iliac artery (IIA)
- Post-delivery: uterus dramatically enlarged with hypertrophied, tortuous uterine arteries — may extend well cephalad above the umbilicus
- Postpartum UA is typically large and easy to catheterize, but also prone to spasm
- Cervicovaginal branch — arises early from UA; can be a source of hemorrhage from cervical / vaginal laceration
- Other anterior division branches: superior vesical, middle rectal, inferior rectal, vaginal, internal pudendal arteries
Collateral Supply & Key Variants
- Ovarian artery — arises from aorta; provides significant collateral supply to gravid/postpartum uterus; must check if bleeding persists after bilateral UAE
- Round ligament artery — arises from inferior epigastric / external iliac artery; can supply vaginal and uterine arteries
- Bilateral embolization almost always required — rich cross-pelvic collateralization means unilateral embolization is rarely sufficient for diffuse uterine bleeding / atony
- Adjust imaging field of view: postpartum uterine arteries extend much more cephalad than typical pelvic arteriography
Angiographic Views
Contralateral anterior oblique (CAO, ~30 degrees) best shows the IIA origin. Steep ipsilateral oblique (30–45 degrees) differentiates anterior and posterior divisions and isolates the uterine artery. Use a fast filming rate (4–6 frames/sec) with injection rates of 7–10 mL/s for 2–4 seconds for pelvic arteriography. Road mapping or fluoro-fade technology is very helpful for rapid vessel selection in the setting of shock physiology.
Technique
Arterial Access
Pelvic Arteriography (Optional)
Contralateral IIA Selection
Selective Uterine Artery Catheterization
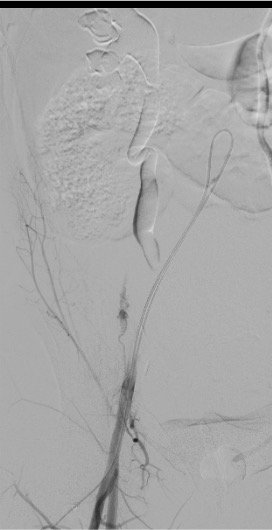
Embolization
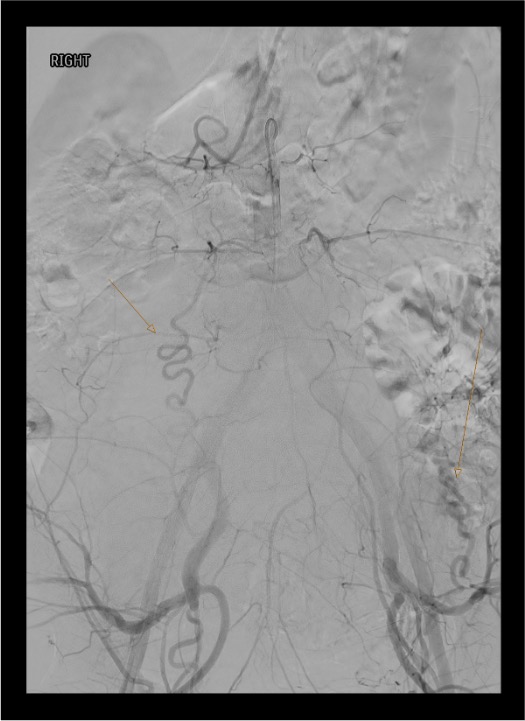
Ipsilateral IIA — Waltman Loop to Cross to Right Side
- Retract catheter to the distal aorta above the bifurcation, tip facing cephalad.
- Advance the catheter with gentle clockwise torque — the tip buckles against the opposite aortic wall, forming a reverse-curve loop in the aortic lumen.
- Continue advancing until the loop is stable and the tip points caudally toward the right iliac.
- Pull back slowly — traction in reverse-curve configuration paradoxically advances the tip into the ipsilateral IIA. Use fluoroscopy to monitor tip position.
- Confirm with contrast injection. If the catheter keeps slipping, advance a stiff wire deep into the right external iliac first to anchor, then advance the catheter over it.
Repeat selective right uterine artery catheterization with microcatheter past the cervicovaginal branch. Bilateral embolization is mandatory in diffuse atony — unilateral embolization has significantly higher failure rate in PPH.
Completion Angiography & Closure
Community Cards
Angiographic Landmarks
Uterine Artery Identification
- IIA angiogram in steep ipsilateral oblique (30–45 degrees) separates anterior and posterior divisions
- Uterine artery originates from the anterior division — typically the dominant vessel in postpartum patients
- Postpartum UA is markedly hypertrophied and tortuous with prominent spiral artery branches
- Road mapping / fluoro-fade essential for rapid selection, especially in shock physiology where vessels may appear “clamped down”
Key Findings
- Extravasation — frank contrast blush beyond the vessel lumen; indicates active arterial hemorrhage
- Pseudoaneurysm — contained contrast collection that persists and washes out with the arterial phase
- Enlarged postpartum vessels — uterine arteries extend well above the pelvis; widen field of view
- Posterior division landmark: identify superior gluteal artery origin — catheter tip must be positioned beyond this to prevent nontarget embolization of the gluteal supply
Troubleshooting
DIC Limiting Embolization Efficacy
DIC is a known predictor of embolization failure. Gelfoam and coils rely on in-situ thrombogenesis for vessel occlusion. Aggressively correct coagulopathy with cryoprecipitate (fibrinogen goal >150), FFP, and platelets. Consider n-BCA glue as embolic agent in coagulopathic patients — does not rely on the coagulation cascade for vessel occlusion. Coordinate ongoing resuscitation with anesthesia team throughout the case.
Continued Hemorrhage Despite Bilateral Uterine Artery Embolization
Check for ovarian artery supply — hypertrophied ovarian arteries from the aorta can provide extensive blood supply to the postpartum uterus. Perform nonselective aortography to identify collaterals. Investigate cervicovaginal branches, internal pudendal artery, obturator artery, and round ligament artery (from inferior epigastric / external iliac). Remove vaginal or uterine packing temporarily to unmask angiographic abnormality. If anterior division embolization of IIA may be required when uterine artery cannot be selectively catheterized.
Uterine Artery Spasm During Catheterization
Uterine arteries are prone to spasm. Use microcatheter for selection to minimize trauma. Avoid deep catheterization. Catheter-related vasospasm alone is unlikely to provide durable hemostasis — attempt to break or cross the spasm and proceed with distal embolization. If unable to cross: less selective anterior division embolization is an acceptable alternative. Avoid intra-arterial nitroglycerin in hemorrhaging patients (hypotensive side effects).
Hemodynamic Instability During Procedure
Patient may decompensate on the table. Ensure anesthesia team is at bedside managing resuscitation. Consider placing central venous catheter (IJ triple lumen) for volume resuscitation. If patient cannot be stabilized: embolize the entire anterior division of the IIA (faster, broader hemostasis) rather than attempting selective UA catheterization. Communicate continuously with OB — hysterectomy may become necessary if embolization cannot achieve hemostasis in a timely manner.
O’Leary Stitch or IIA Ligation by OB Team
Obtain detailed operative history from OB. Suspect ligation when usually apparent uterine or IIA arteries are not visualized. In a published series, 9 of 11 ligated IIAs demonstrated residual angiographic flow — crossing the ligated vessel remains viable. Empiric anterior division embolization can control collateral flow around a ligated uterine artery. Prior ligation has not been shown to impact embolization success rates.
Complications
Common / Expected
- Post-embolization syndrome — pelvic pain, low-grade fever, nausea; self-limited; manage with analgesics and antipyretics
- Access site hematoma — especially in coagulopathic patients; more common with femoral vs radial approach
- Rebleeding / re-embolization (~5–10%) — may require repeat angiography; CTA useful for workup of rebleeding
Serious Complications
- Uterine necrosis (rare with Gelfoam) — associated with smaller particles (<500 µm) and simultaneous ovarian + uterine artery embolization; requires hysterectomy
- Infection / endometritis / abscess — low incidence; prophylactic antibiotics recommended
- Asherman syndrome (intrauterine adhesions) — rare; may affect future fertility
- Ovarian failure / amenorrhea — extremely rare with Gelfoam; more concerning with permanent embolic agents or if ovarian arteries embolized
- Nontarget embolization — reflux into posterior division (superior gluteal artery); meticulous catheter positioning beyond posterior division origin is critical
Pearls & Pitfalls
References & Resources
Key Outcomes
- Technical success: approaching 100% in most modern series
- Clinical success: ~89% per meta-analysis (Sathe et al. 2016)
- Fertility preservation: multiple retrospective studies confirm successful conception and delivery post-UAE for PPH
Standards of Practice
- CIRSE SoP: GYN/Obstetric Hemorrhage → Rand T et al., CVIR Endovascular 2020 · DOI
Primary References
- Pelage JP, Le Dref O, Jacob D, et al. Selective arterial embolization of the uterine arteries in the management of intractable post-partum hemorrhage. Acta Obstet Gynecol Scand. 1999;78(8):698–703.
- Soncini E, Pelicelli A, Larini P, Marcato C, Monaco D, Grignaffini A. Uterine artery embolization in the treatment and prevention of postpartum hemorrhage. Int J Gynaecol Obstet. 2007;96(3):181–185.
- Newsome J, Martin JG, Bercu Z, Shah J, Shekhani H, Peters G. Postpartum hemorrhage. Tech Vasc Interv Radiol. 2017;20(4):266–273.
- Brown M, Hong M Jr, Lindquist J. Uterine artery embolization for primary postpartum hemorrhage. Tech Vasc Interv Radiol. 2021;24(4):100727.
- Heaston DK, Mineau DE, Brown BJ, et al. Transcatheter arterial embolization for control of persistent massive puerperal hemorrhage after bilateral surgical hypogastric artery ligation. AJR Am J Roentgenol. 1979;133(1):152–154.
- Lee HY, Shin JH, Kim J, et al. Primary postpartum hemorrhage: outcome of pelvic arterial embolization in 251 patients at a single institution. Radiology. 2012;264(3):903–909.