Indications & Contraindications
Indications
- BPH with moderate-to-severe LUTS refractory to medical therapy (alpha-blockers, 5-alpha reductase inhibitors)
- IPSS ≥13 (moderate–severe symptom score)
- Qmax <12 mL/s on uroflowmetry
- Prostate volume >40 mL (larger prostates tend to respond better)
- Alternative to TURP or surgical prostatectomy — especially for patients who are poor surgical candidates or decline surgery
- Patients with urinary retention secondary to BPH (Foley-dependent)
Contraindications
- Prostate cancer — must be excluded prior to PAE (MRI + PSA)
- Active urinary tract infection — treat before proceeding
- Uncorrectable coagulopathy
- Severe atherosclerosis of iliac arteries — may prevent catheter access to prostatic arteries
- Known allergy to iodinated contrast
- Relative: large bladder diverticula, neurogenic bladder (symptoms may not improve with PAE)
- Relative: very small prostate (<40 mL) — lower response rates
PAE vs. Surgical Options
| Feature | PAE | TURP / Surgery |
|---|---|---|
| Anesthesia | Local + moderate sedation | General / spinal |
| Hospital stay | Outpatient / same-day | 1–3 days |
| Sexual side effects | Low risk of retrograde ejaculation | 65–75% retrograde ejaculation (TURP) |
| Efficacy (IPSS reduction) | 40–60% improvement at 6 months | 70–80% improvement |
| Best for | Large prostates (>80 mL), poor surgical candidates, patient preference | Moderate-sized glands, failed PAE, median lobe predominant |
Pre-Procedure Planning
Imaging & Labs
- MRI prostate: evaluate prostate anatomy, zonal volumes, exclude prostate cancer (PI-RADS scoring)
- CTA pelvis: map prostatic artery anatomy and origin, assess iliac atherosclerosis and tortuosity, plan catheterization strategy
- IPSS questionnaire: baseline symptom severity score (document for follow-up comparison)
- Qmax uroflowmetry: baseline peak flow rate
- PSA: rule out prostate cancer (biopsy if elevated)
- UA / UCx: exclude active UTI before procedure
- CBC, BMP, PT/INR — standard pre-procedure labs
Patient Preparation
- Alpha-blocker — continue (or start) tamsulosin; helps reduce post-PAE urinary retention; maintain for 2–4 weeks post-procedure
- Prophylactic antibiotics: ciprofloxacin 400 mg IV or levofloxacin 500 mg PO pre-procedure; continue oral course 5–7 days post
- Proton-pump inhibitor and anti-inflammatory started 24h before procedure
- Foley catheter if patient in urinary retention; fill balloon with dilute contrast (10–20% iodinated) for intraprocedural bladder neck identification
- NPO 6h for moderate sedation
- IV access established; standard monitoring
Relevant Anatomy
Prostatic Artery Origin
- Most common: arises from the anterior trunk of the internal iliac artery, typically as a branch of the inferior vesical artery (Type I — common trunk with superior vesical artery, ~29%)
- Type II (~15%): origin from the anterior division of the IIA, inferior to the superior vesical artery
- Type III (~19%): origin from the obturator artery
- Type IV (~31%): origin from the internal pudendal artery (most common single origin)
- Type V (~6%): rare origins — accessory internal pudendal, aberrant obturator from external iliac/inferior epigastric
- Double vascularization (two prostatic arteries to one hemi-prostate) seen in ~8% of hemi-pelves
Key Anatomical Relationships
- Bilateral supply: each hemi-prostate receives its own prostatic artery — bilateral embolization preferred for optimal outcomes
- Intraprostatic branches: anteromedial branches supply the central/transition zone (BPH target); posterolateral branches supply the peripheral zone
- Dangerous anastomoses: prostatic artery communicates with bladder branches (superior vesical), rectal branches (middle rectal), and penile branches (internal pudendal) — non-target embolization risk
- PROVISO mnemonic for ipsilateral oblique angiography: internal Pudendal, middle Rectal, Obturator, Vesical Inferior, Superior under Oblique view
- Cone-beam CT (CBCT) is critical for identifying prostatic artery origins and mapping anastomoses before embolization
Vascular Anatomy Planning
Pre-procedural CTA provides identification of prostatic artery origins in up to 97% of cases. However, intraprocedural cone-beam CT from the internal iliac artery remains essential for real-time confirmation of catheter position, identification of accessory prostatic branches, and assessment of dangerous anastomoses to bladder, rectum, and penile territories. The best angiographic projection to identify pelvic branches is the 20–50 degree ipsilateral oblique view with 10–20 degree caudal angulation.
Technique
Arterial Access
Internal Iliac Artery Catheterization
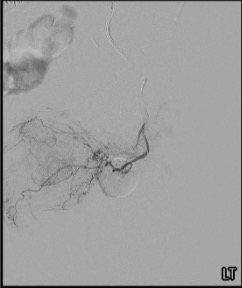
Selective DSA & Prostatic Artery Identification
Microcatheter Advancement
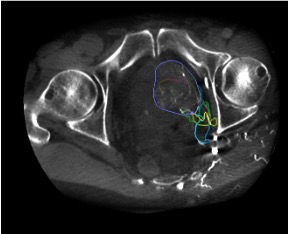
CBCT Confirmation
Embolization
Control Angiogram & Contralateral Embolization
Access Site Closure
Community Cards
Landmarks & Imaging Confirmation
CBCT Confirmation Landmarks
- Prostatic blush: diffuse parenchymal enhancement of the hemi-prostate on CBCT confirms correct catheter position — the defining landmark before embolization
- Central gland vs. peripheral zone: anteromedial branches supply the transition zone (BPH target); posterolateral branches supply the peripheral zone
- Foley catheter balloon (contrast-filled) helps identify bladder neck and prostatic urethra on CBCT
- 3D reconstruction models from proximal CBCT datasets allow advanced guidance for microcatheterization path planning
Non-Target Vessel Assessment
- Bladder branches: identified on CBCT as enhancement extending cephalad beyond the prostatic gland into the bladder wall. Caudal 10–20 degree view on DSA helps identify bladder branches
- Rectal branches: enhancement posterior to the prostate extending toward rectal wall — if identified, reposition microcatheter distal to branch origin or perform protective coiling
- Penile/pudendal branches: high-flow anastomoses to pudendal territory — protective embolization with coils or gelatin sponge may be needed
- If non-target supply cannot be avoided, consider using larger embolic particles (300–500 µm) to reduce risk of distal non-target migration
Angiographic Anatomy on Pelvic Arteriogram
On ipsilateral oblique selective IIA arteriography, the pelvic arterial branches are best identified using the PROVISO system. The prostatic artery arises most commonly from the anterior division, courses inferomedially toward the prostate, and gives rise to anteromedial and posterolateral intraprostatic branches. Ipsilateral oblique views (20–50 degrees) separate overlapping vessels, while caudal angulation (10–20 degrees) helps delineate bladder branches from prostatic feeders. Venous phase on the prostatic arteriogram confirms complete parenchymal perfusion.
Troubleshooting
Prostatic Artery Not Seen on Selective IIA Arteriogram
Perform cone-beam CT from the IIA with pressurized contrast injection (2 mL/sec, 4–6 sec delay) to identify small or variant prostatic branches not visible on DSA. If still not identified, perform CBCT from the external iliac artery to search for aberrant obturator artery origin (from external iliac or inferior epigastric, seen in ~2% of cases). Check both anterior and posterior divisions of the IIA. Previous embolization or atherosclerotic occlusion of native prostatic artery may result in revascularization from accessory branches.
Vasospasm During Microcatheterization of Prostatic Artery
Inject 100–200 mcg nitroglycerin diluted in saline directly through the microcatheter. Wait 2–3 minutes for vasodilation. Avoid excessive catheter manipulation. Nitroglycerin should be administered prophylactically once the prostatic artery is catheterized to increase arterial diameter and facilitate distal advancement.
Unable to Catheterize or Embolize Contralateral Prostatic Artery
Unilateral PAE is still effective but associated with lower clinical success compared to bilateral embolization. Document which side was embolized. Consider second-session attempt for the contralateral side from alternative access (ipsilateral femoral, contralateral femoral, or radial). If due to atherosclerotic iliac disease, alternative access or long sheath may help.
Severe Iliac Tortuosity or Atherosclerosis Limiting Catheter Advancement
Use longer sheaths (45 cm) and extra-support guidewires to straighten tortuous segments. Conform preshaped 5F catheters or use catheter loops in branch arteries for additional support. Consider contralateral femoral, radial, or ulnar access to avoid the problematic iliac segment. Iliac tortuosity increases procedure time by ~43%, fluoroscopy time by ~47%, and contrast volume by ~25 mL.
Early Proximal Occlusion During Embolization Preventing Adequate Particle Delivery
If microcatheter position is proximal within the prostatic artery trunk, premature stasis with reflux frequently occurs. Advance microcatheter distally into the intraprostatic vessels and perform wedged injection (PErFecTED technique: Proximal Embolization First, Then Embolize Distal). This allows additional embolic volume delivery without free-flow reflux and has been associated with lower 1-year recurrence rates.
Complications
Common / Expected Side Effects
- Post-embolization syndrome (20–30%) — dysuria, pelvic discomfort, low-grade fever, urinary frequency; self-limited 3–7 days; manage with NSAIDs, alpha-blockers, anti-emetics
- Transient hematuria — common in first 24–48h; usually self-limited
- Transient urinary retention — due to prostatic edema; Foley catheter if needed; alpha-blocker continuation
- UTI (<5%) — prophylactic antibiotics reduce incidence; treat promptly if develops post-procedure
- Access site complications — hematoma, pseudoaneurysm; standard femoral access management
Serious Complications
- Rectal ischemia — non-target embolization to middle rectal artery branches; CBCT mandatory to assess for rectal enhancement before embolizing; if suspected post-procedure: rectal pain, bloody stools → proctoscopy
- Bladder ischemia — non-target embolization to superior vesical / bladder branches; presents as severe suprapubic pain, hematuria; CBCT pre-embolization identifies at-risk branches
- Seminal vesicle ischemia — rare; may cause hematospermia
- Penile/urethral ischemia — rare but serious; from non-target embolization via pudendal anastomoses; protective coiling when anastomoses identified
- Radiation injury — prolonged fluoroscopy time in complex cases; minimize with CBCT guidance and dose reduction techniques
Pearls & Pitfalls
References & Resources
Primary References
- Pisco JM, Bilhim T, Pinheiro LC, et al. Medium- and long-term outcome of prostate artery embolization for patients with benign prostatic hyperplasia: results in 630 patients. J Vasc Interv Radiol. 2016;27(8):1115–1122.
- Abt D, Hechelhammer L, Müllhaupt G, et al. Comparison of prostatic artery embolisation (PAE) versus transurethral resection of the prostate (TURP) for benign prostatic hyperplasia: randomised, open label, non-inferiority trial (WATER). BMJ. 2018;361:k2338.
- Ray AF, Powell J, Wallis C, et al. Efficacy and safety of prostate artery embolization for benign prostatic hyperplasia: an observational study and propensity-matched comparison with transurethral resection of the prostate (UK-ROPE). Cardiovasc Intervent Radiol. 2018;41(8):1138–1148.
- Carnevale FC, Antunes AA. Prostatic artery embolization for enlarged prostates due to benign prostatic hyperplasia: How I do it. Cardiovasc Intervent Radiol. 2014;37:1602–1605.
- de Assis AM, Moreira AM, de Paula Rodrigues VC, et al. Pelvic arterial anatomy relevant to prostatic artery embolisation and proposal for angiographic classification. Cardiovasc Intervent Radiol. 2015;38:855–861.
- McWilliams JP, Bilhim TA, Carnevale FC, et al. Society of Interventional Radiology Multisociety Consensus Position Statement on Prostatic Artery Embolization for Treatment of LUTS Attributed to BPH. J Vasc Interv Radiol. 2019;30:627–637.
- Carnevale FC, Moreira AM, Harward SH, et al. Recurrence of lower urinary tract symptoms following prostate artery embolization for benign hyperplasia: single center experience comparing two techniques. Cardiovasc Intervent Radiol. 2017;40:366–374.