Indications & Patient Selection
Classic Presentation
- Nocturnal pain relieved by aspirin or NSAIDs — highly characteristic; driven by elevated prostaglandins (PGE2/PGI2) in the nidus
- Age 5–30 years; male predominance (~4:1 male-to-female ratio)
- Locations: long bones (femur/tibia ~50%), spine (posterior elements), small bones of hands and feet; ischium, patella, skull are atypical
- Painful scoliosis in adolescents — consider spinal OO
Imaging Workup
- CT (thin-slice, ≤1 mm): lucent nidus (<2 cm) surrounded by dense reactive cortical sclerosis — "halo sign"; central mineralized focus may be present
- Bone scan (Tc-99m MDP): near 100% sensitivity; "double density" sign with focal uptake in nidus + surrounding halo
- MRI: less sensitive for nidus but demonstrates perilesional marrow edema (STIR hyperintensity) and joint effusion; useful for spinal lesions
- Radiographs: may be normal or show only reactive thickening; not reliable for nidus identification
Indications for Ablation
- Failed conservative therapy (NSAIDs) ≥6 months — up to 75% respond to NSAIDs alone; ablation reserved for those who do not
- Intolerable pain significantly impairing daily activity or sleep
- Spinal lesions with neurologic risk or scoliosis progression
- Patient preference for definitive treatment over long-term NSAID use
Contraindications
- Nidus >2 cm — consider osteoblastoma or other diagnosis; surgical consultation preferred
- Proximity to bowel or major neurovascular structures without thermal protection strategy
- Pregnancy
- Active infection at access site or suspected osteomyelitis
- Uncorrectable coagulopathy
- Articular cartilage within ablation zone (relative; consider cryo)
Pre-Procedure Checklist
Relevant Anatomy
Nidus Structure
- Inner nidus: central vascularized fibrous stroma containing osteoid trabeculae with osteoblasts — highly metabolically active; contains prostaglandins 100–1000× normal bone levels
- Outer reactive zone: thickened woven and lamellar bone — the dense sclerotic rim seen on CT; this is NOT the target
- Size: nidus characteristically <2 cm (usually 5–15 mm); lesions >2 cm raise concern for osteoblastoma
- Nidus on CT: rounded or ovoid lucency ± dense central mineralized focus; high T2/STIR signal on MRI with surrounding edema
Location-Specific Anatomy
- Cortical (intracortical): most common; within bony cortex of diaphysis or meta-diaphysis — dense surrounding sclerosis makes drilling challenging
- Cancellous: medullary location; easier needle passage but nidus may be less conspicuous
- Subperiosteal: beneath periosteum, no cortex to drill; highest risk of skin burn from superficial location
- Intra-articular: near articular cartilage — risk of chondral thermal injury; cryo preferred
- Spinal: posterior elements (pedicle, lamina, facet joint); proximity to spinal canal and nerve roots is the key hazard
Proximity Hazards by Location
- Proximal femur: femoral vessels and nerve; periarticular lesions risk femoral head avascular necrosis
- Spine: spinal cord, nerve roots, dural sac; epidural vessels
- Intra-articular (hip, knee, ankle): articular cartilage — limit thermal spread
- Hand/foot small bones: digital nerves and tendons in close proximity
Periosteum
- Highly innervated — primary source of pain during needle placement and drilling
- Generous local anesthesia required from skin to periosteum regardless of general anesthesia
- Subperiosteal lesions: periosteum elevation can spread ablation heat superficially → skin burn risk → hydrodissection with saline or D5W recommended
Technique
Default RadCall approach · share your own below
Supplies
Steps
Position and CT planning scan
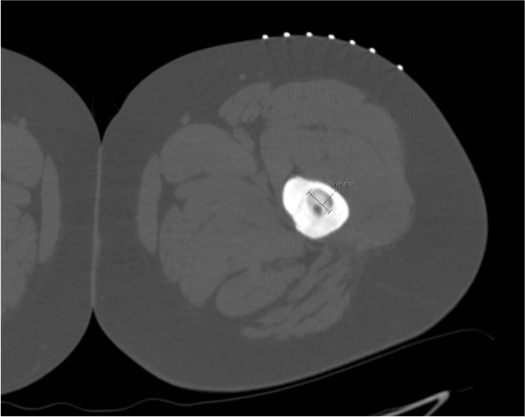
Skin prep and local anesthesia
Advance bone access needle to nidus
Coaxial access and biopsy (optional)
Insert RFA probe
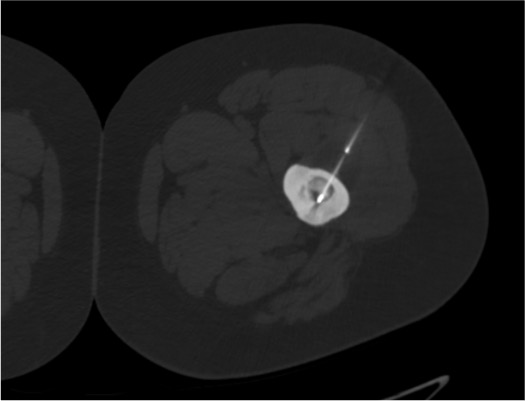
Ablation
Probe removal and hemostasis
Post-procedure CT
Recovery and discharge
Ablation Modality Comparison
| Modality | Mechanism | Advantages | Considerations |
|---|---|---|---|
| RFA (standard) | Alternating current → ionic oscillation → heat | Most evidence base; predictable 90–95°C zone; cooled probes reduce charring | Cannot use near implanted metal; limited by carbonization at high temps |
| Microwave (MWA) | 2.45 GHz EM field → water molecule oscillation → heat | Higher temperatures more rapidly; less affected by heat sink; promising early data | Less literature for OO specifically; requires shorter ablation cycles (30W × 3 min) |
| Cryoablation | Pressurized gas expansion → lethal freezing | Preferred near articular cartilage and neural structures; visible iceball on CT | Requires larger access needle; ice ball less predictable in cortical bone; limited OO-specific series |
| LITT (Laser) | Laser energy → photothermal tissue destruction | MRI-compatible; very precise small zone; useful in difficult locations | Requires MRI guidance suite; less commonly available; less OO-specific data |
Fluoroscopy / CT Landmarks
Key CT Findings
- "Halo sign": dense reactive sclerosis surrounding the central lucent nidus — the sclerosis is the conspicuous finding; the nidus is within it
- Nidus: rounded or ovoid lucency; may contain a dense central calcified focus (mineralized osteoid). Average diameter 5–15 mm
- Central mineralized focus within the lucency should not be confused with the reactive sclerosis — the probe tip targets the lucent nidus around it
- MRI correlation: STIR hyperintensity extending beyond nidus indicates perilesional edema — confirms active lesion
Probe Positioning Targets
- Probe tip within 1 mm of nidus center — required for adequate ablation; minor displacement significantly reduces efficacy
- Active tip length must match nidus size: 1-cm tip for nidus ≤1 cm; 2-cm tip for larger nidus; overlap two 1-cm burns for nidus >1 cm if 2-cm probe unavailable
- Spinal lesions: confirm probe tip is ≥1 cm from posterior spinal canal on axial CT; ≥5 mm from nerve root foramina
- Articular lesions: confirm distance from articular cartilage surface; <1 cm warrants reduced power or cryoablation
- Post-ablation CT: gas foci within the nidus confirm adequate heating; lack of gas may indicate incomplete ablation
Troubleshooting
Sclerotic bone prevents needle or drill advance
Likely cause: Dense reactive perilesional sclerosis in cortical intracortical OO — common in long-standing lesions.
Next step: Switch to diamond-tip drill bit attachment on coaxial system. Apply firm steady pressure with rotational motion — avoid excessive torque that can deviate trajectory. Consider manual bone drill as backup. If biopsy is indicated, perform concurrent drill biopsy of nidus material. Confirm trajectory on CT before fully advancing.
Cannot identify or confirm nidus on CT
Likely cause: Nidus very small (<5 mm), heavily calcified, or reactive sclerosis is overwhelming. Suboptimal CT technique (thick slices).
Next step: Correlate with bone scan — site of maximum uptake corresponds to nidus. Consider nuclear medicine probe (gamma probe) guidance in the procedure suite. Request MRI-guided ablation if available (LITT). If still uncertain after correlation, target the area of maximum bone scan activity within the sclerotic zone.
Probe tip near articular cartilage
Likely cause: Intra-articular or periarticular OO at hip, knee, or ankle — relatively common; up to 13% of OO are intra-articular.
Next step: Reduce power setting and shorten ablation cycle duration to minimize thermal spread. Inject saline or D5W into the joint to create a heat-sink buffer. If ≤1 cm from cartilage, strongly consider cryoablation — iceball is visible on CT and cryo preserves cartilage better than heat-based modalities.
Spinal lesion near nerve root or spinal canal
Likely cause: Posterior element OO (pedicle, lamina, facet) — proximity to epidural space or neural foramen is the critical risk.
Next step: Place temperature monitoring probe adjacent to neural structures; abort if >45°C reached. Perform D5W epidural cooling via separately placed epidural needle (D5W does not stimulate neural tissue at low temperatures). CO2 pneumodissection can physically separate the nidus from the neural structures. Consider cryoablation — cryo is better characterized near neural structures and the iceball is visible.
Hemodynamic response during ablation (hypertension / tachycardia)
Likely cause: Expected — osteoid osteomas contain prostaglandins at 100–1,000× normal levels; ablation releases these, causing transient cardiovascular stimulation.
Next step: Warn anesthesia team before ablation begins. This is a recognized phenomenon — manage with short-acting antihypertensives or beta-blockers as needed. Symptoms resolve when ablation is turned off. Document and reassure.
Complications
Periprocedural / Early
- Post-procedure pain flare (expected, not a complication per se) — typically 1–3 days of increased pain; managed with NSAIDs + short course of acetaminophen; warn patient before discharge
- Skin burn — risk with subperiosteal lesions; prevent with hydrodissection (saline or D5W) between nidus and skin; treat with standard wound care
- Intraosseous bleeding / hematoma — usually self-limited; hemostatic plug at access tract reduces risk
- Hemodynamic instability (transient hypertension/tachycardia) — prostaglandin-mediated; resolves with ablation cessation; coordinate with anesthesia
Delayed / Major
- Incomplete ablation / Recurrence (~5–10%) — persistent or recurrent symptoms after initial apparent success; repeat RFA is usually successful; correlate with repeat CT and bone scan at 3–6 months
- Infection / Osteomyelitis — rare (<1%); cefazolin prophylaxis is standard; fever + worsening pain post-procedure → MRI + ID consultation
- Pathologic fracture — risk if >50% cortical involvement; restrict weight-bearing 4–6 weeks for high-risk lesions; prophylactic fixation rarely required
- Neurologic injury — <1% with appropriate thermal protection for spinal lesions; presents as new radiculopathy or weakness; MRI workup; usually resolves with conservative management
- Growth plate injury — pediatric patients; avoid ablation zone crossing open physis; confirm safe distance on pre-procedure CT
Critical Pearls
References
Citations
- Prologo JD, Ray CE Jr., eds. Advanced Pain Management in Interventional Radiology: A Case-Based Approach. Thieme; 2024. Chapters 37–38 (Macha V, Devane AM, Gunn AJ). DOI: 10.1055/b000000387
- Rosenthal DI, Hornicek FJ, Wolfe MW, Jennings LC, Gebhardt MC, Mankin HJ. Percutaneous radiofrequency coagulation of osteoid osteoma compared with operative treatment. J Bone Joint Surg Am. 1998;80(6):815–821.
- Rosenthal DI, Hornicek FJ, Torriani M, Gebhardt MC, Mankin HJ. Osteoid osteoma: percutaneous treatment with radiofrequency energy. Radiology. 2003;229(1):171–175.
- Kransdorf MJ, Stull MA, Gilkey FW, Moser RP Jr. Osteoid osteoma. Radiographics. 1991;11(4):671–696.
- Chai JW, Hong SH, Choi JY, et al. Radiologic diagnosis of osteoid osteoma: from simple to challenging findings. Radiographics. 2010;30(3):737–749.
- Lindner NJ, Ozaki T, Roedl R, Gosheger G, Winkelmann W, Wörtler K. Percutaneous radiofrequency ablation in osteoid osteoma. J Bone Joint Surg Br. 2001;83(3):391–396.
- Coupal TM, Mallinson PI, Munk PL, et al. CT-guided percutaneous cryoablation for osteoid osteoma: initial experience in adults. AJR Am J Roentgenol. 2014;202(5):1136–1139.
- Vanderschueren GM, Taminiau AH, Obermann WR, Bloem JL. Osteoid osteoma: clinical results with thermocoagulation. Radiology. 2002;224(1):82–86.
References & Resources
Key Guidelines
- SIR Standards of Practice for Thermal Ablation
- CIRSE Standards of Practice for Bone Lesion Ablation
Primary References
- Rosenthal DI et al. Osteoid osteoma: percutaneous radio-frequency ablation. Radiology. 1998;209(3):794-798.
- Gangi A et al. Percutaneous laser photocoagulation of spinal osteoid osteomas under CT guidance. AJNR Am J Neuroradiol. 1998;19(10):1955-1958.
- Vanderschueren GM et al. The natural history of osteoid osteoma. J Bone Joint Surg Am. 2005;87(8):1795-1800.