Indications & Contraindications
Acute Mesenteric Ischemia (AMI)
- Embolic (40–50%) — most common; cardiac source (atrial fibrillation, post-MI); abrupt onset severe pain out of proportion to exam; SMA occlusion typically ≥3 cm from origin
- Thrombotic (25%) — acute-on-chronic atherosclerotic disease; occlusion within 1–2 cm of SMA origin; often history of postprandial pain (intestinal angina); collaterals present
- Nonocclusive (NOMI) (20–30%) — prolonged hypotension, vasopressors, digitalis/dopamine use; diffuse mesenteric vasospasm at branch points; no focal occlusion
- Mesenteric venous thrombosis (<5%) — portal hypertension, hypercoagulable states, abdominal inflammation, oral contraceptives
- Aortic dissection (<5%) — flap extending into SMA; true/false lumen compromise
Chronic Mesenteric Ischemia (CMI)
- Postprandial pain — crampy, dull; begins shortly after eating, lasts 1–2 hours
- Weight loss — present in ~80% of patients
- “Food fear” / food aversion — patients avoid eating due to anticipated pain; pathognomonic for CMI
- Intestinal angina — classic description; insufficient splanchnic blood flow during periods of heightened demand
- Atherosclerotic narrowing of ≥2 of 3 mesenteric vessels (celiac, SMA, IMA) classically required before symptom onset
- Other etiologies: fibromuscular dysplasia, vasculitis, post-surgical intimal hyperplasia
- More common in women (more acute aortomesenteric angle)
Contraindications
- Peritonitis / frank bowel infarction — requires surgical exploration and bowel resection first; endovascular alone insufficient
- No suitable vascular access (severe aortoiliac occlusive disease without upper extremity alternative)
- Uncorrectable coagulopathy
- Severe contrast allergy without adequate premedication
- Relative: Median arcuate ligament compression — surgical decompression preferred over stenting (high stent fracture rate from extrinsic compression)
Pre-Procedure Planning
Imaging
- CTA abdomen/pelvis with arterial phase — initial diagnostic exam of choice for both AMI and CMI; identifies stenosis/occlusion location, collateral pathways, bowel wall changes
- Look for: pneumatosis, portal venous gas, bowel wall thickening/enhancement (AMI signs of infarction)
- Sagittal reconstructions: evaluate ostial disease of celiac, SMA, IMA
- MRA: alternative when CTA contraindicated (eGFR 30–60); limited in small vessel disease and bowel assessment
- Duplex ultrasound (CMI workup): fasting and postprandial states; PSV and EDV assessment; inexpensive screening tool
Labs & Patient Prep
- AMI labs: CBC, CMP with liver enzymes, lipase, serum lactate, coagulation panel (PT/INR, aPTT), D-dimer, type & screen
- CMI labs: CBC, BMP, coagulation panel, type & screen
- Elevated WBC, lactate, and liver enzymes consistent with AMI — markedly elevated lactate with peritoneal signs is an ominous combination
- NPO 6–8 hours (if elective CMI); AMI is emergent — do not delay for NPO status
- IV access ×2; arterial line for AMI cases
- Echocardiogram or cardiac CTA (AMI embolic): rule out cardiac chamber thrombus before thrombolysis
Relevant Anatomy
Celiac Trunk
- Origin: anterior aorta at T12–L1
- Branches: left gastric, splenic, common hepatic arteries
- Supplies: stomach, liver, spleen, proximal duodenum, pancreas
- May be compressed by median arcuate ligament
Superior Mesenteric Artery (SMA)
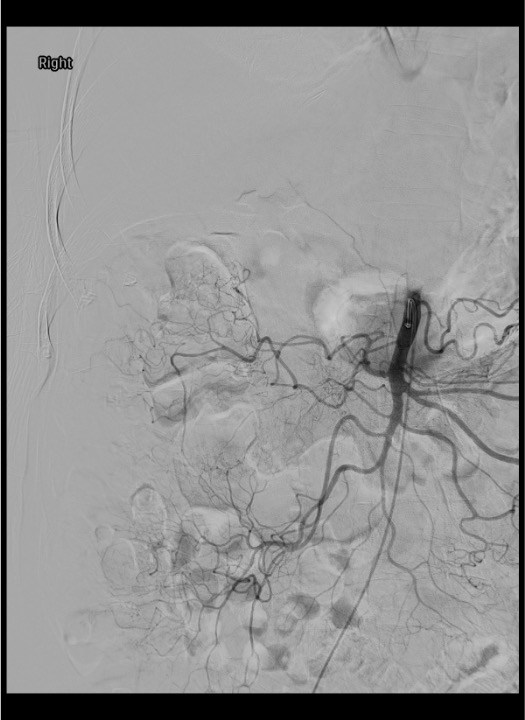
- Origin: anterior aorta at L1, ~1 cm below celiac trunk
- Branches: inferior pancreaticoduodenal, jejunal, ileal, ileocolic, right colic, middle colic arteries
- Supplies: distal duodenum to splenic flexure of colon
- Primary target vessel in both AMI and CMI interventions
Inferior Mesenteric Artery (IMA)
- Origin: anterior aorta at L3
- Branches: left colic, sigmoidal, superior rectal arteries
- Supplies: splenic flexure to proximal rectum
- Smallest of the three; may require 0.014–0.018″ systems
Critical Collateral Pathways
| Collateral | Connects | Clinical Significance |
|---|---|---|
| Pancreaticoduodenal arcade | Celiac (GDA) ↔ SMA (inferior pancreaticoduodenal) | Primary celiac–SMA collateral; dilated arcade on CTA suggests proximal stenosis |
| Arc of Riolan (meandering mesenteric artery) | SMA (middle colic) ↔ IMA (left colic) | Central anastomosis; critical when SMA or IMA occluded |
| Marginal artery of Drummond | SMA ↔ IMA along colonic mesenteric border | Runs along mesenteric border of colon; variable at splenic flexure (Griffith point) |
| Arc of Buhler | Celiac ↔ SMA (rare embryologic variant) | Direct communication; present in ~2% of patients |
Supplies & Technique
Access & Catheters
- Femoral access: 7F 45-cm Flexor Ansel guiding sheath (Cook)
- Radial access (preferred over brachial): 6F radial sheath (Terumo); 90–110 cm Destination or Flexor Raabe long sheath for reach
- Selective catheters: Cobra 2, SOS Omni 2, RC 1, Simmons 1
- 7F ENVOY guiding catheter (for aspiration thrombectomy)
- Hydrophilic steerable guidewire (0.035″)
- Stiff exchange wire (Amplatz, Rosen)
Intervention Devices
- Balloon-expandable stents: Express LD (Boston Scientific), Herculink (Abbott) — preferred for ostial lesions
- Covered stents: iCast (Atrium) — for soft plaque, vessels <6 mm, or in-stent restenosis
- Low-profile angioplasty balloons: 0.014–0.018″ compatible; 2–3 mm for predilation, 4–8 mm for definitive
- AngioJet rheolytic thrombectomy system (Bayer) — for AMI embolic/thrombotic
- Infusion catheter (tPA delivery for AMI): multi-sidehole
- Aspiration catheters / Penumbra system
Medications
- IV heparin 3000–5000 IU (target ACT >220s)
- tPA (alteplase) 4–12 mg for catheter-directed thrombolysis (AMI)
- IA nitroglycerin 100–200 mcg boluses for vasospasm
- IA papaverine 45–60 mg bolus, then 1.0 mg/min infusion (NOMI and post-revascularization)
- Clopidogrel 300 mg loading + aspirin 325 mg post-stent
- Maintenance: clopidogrel 75 mg/d × 3–6 months + aspirin 81–325 mg/d for life
AMI Technique
Arterial Access & Aortogram
Selective SMA Catheterization
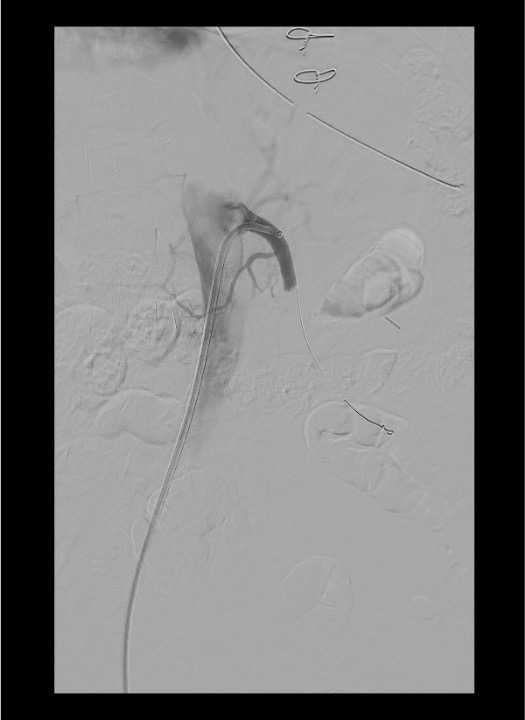
Thrombus Removal
Treat Underlying Lesion (Thrombotic AMI)
Completion Angiogram & Adjuncts
CMI Technique
Access & Diagnostic Aortogram
Selective Catheterization & Pressure Measurement
Lesion Crossing & Predilation
Stent Deployment
Completion Angiogram & Antiplatelet Loading
Community Cards
Landmarks & Imaging
Lateral Aortogram
AP (Flush) Aortogram
Selective SMA Angiogram
NOMI Findings
Troubleshooting
Cannot Engage the SMA Origin from Femoral Access
Try reverse-curve catheter (Simmons 1, SOS Omni 2) which may provide better purchase on downward-angled SMA. If acute angle persists, switch to radial access (right radial preferred; 6F sheath, 90–110 cm Destination or Flexor long sheath) — the coaxial approach from above provides a more favorable vector for acutely angled celiac/SMA origins. An 8F Morph steerable catheter (BioCardia) can navigate severe tortuosity and provide additional support for wire and device delivery.
Proximal Vessel Open but Distal Branches Remain Poorly Perfused
Administer IA papaverine 45–60 mg bolus directly into SMA (potent vasodilator; >90% first-pass hepatic clearance, low systemic risk). Consider additional thrombolysis for residual distal clot. If persistent malperfusion with peritoneal signs and rising lactate, proceed to exploratory laparotomy — do not delay surgical assessment.
Clot Fragments Migrate to Distal SMA Branches During Intervention
Aspirate through guiding catheter or sheath advanced beyond embolization site. Consider AngioJet or additional directed thrombolysis to affected branches. Distal protection devices remain controversial in mesenteric intervention but may be considered in cases with large thrombus burden. Adequate heparinization is critical to prevent propagation.
Catheter-Induced or Post-Intervention Vasospasm
IA nitroglycerin 100–200 mcg bolus directly into SMA. May repeat every 3–5 minutes as needed. For sustained vasospasm (particularly NOMI), initiate IA papaverine infusion at 1.0 mg/min via indwelling SMA catheter. Monitor for systemic hypotension (uncommon with IA papaverine due to hepatic first-pass effect). Avoid vasopressin and alpha-agonists — worsen mesenteric ischemia.
Cannot Cross Chronic Occlusion with Standard Wire
Use hydrophilic steerable guidewire with aggressive tip shape. Predilate with low-profile 2–3 mm balloon (0.014–0.018″ system) to create initial channel. Advance sheath over deflating balloon. If unable to cross from femoral approach, switch to radial access for a coaxial, downgoing approach — more favorable wire/sheath angulation from above the lesion. Consider 0.018″ or 0.014″ platform for heavily calcified lesions.
Complications
Major Complications
- Bowel infarction despite intervention — may occur if revascularization is delayed or incomplete; persistent peritoneal signs and rising lactate post-procedure require surgical exploration
- Reperfusion injury — occurs after re-establishing inline flow; oxygen-rich blood interacts with locally produced free radicals; can cause additional cell death beyond initial ischemic insult; may lead to SIRS and multiorgan failure
- Distal embolization — clot fragments to branch vessels during thrombectomy or catheter manipulation; can convert partial ischemia to complete infarction
- Vessel dissection — wire or catheter injury to SMA intima; can be flow-limiting; may require stenting
- Stent thrombosis — acute or subacute; requires aggressive antiplatelet therapy and surveillance
Other Complications
- Access site complications — hematoma, pseudoaneurysm (increased risk with thrombolysis/anticoagulation); if radial access used: check post-procedure radial pulse + Allen test; if brachial used (avoid if possible): risk of thrombosis and median nerve compression
- Contrast-induced nephropathy — dehydration common in AMI patients exacerbates risk; pre/post hydration essential
- Stent dislodgement — balloon-expandable stent not fully deployed; careful positioning before final inflation
- In-stent restenosis (CMI) — intimal hyperplasia; risk factors: occlusions, lesions >3 cm, stent <5 mm diameter; treated with re-angioplasty or covered stent
- SIRS / sepsis — bacterial translocation from ischemic bowel; broad-spectrum antibiotics indicated for AMI
Pearls & Pitfalls
References & Resources
Follow-up Protocol
- AMI: ICU admission; IV heparin; monitor lactate, WBC, hemodynamics; CTA at 1 month; duplex US 6–12 months; clopidogrel 75 mg/d × 3–6 months + aspirin 81–325 mg/d for life
- CMI: Outpatient clinic at 30 days (history, physical, duplex US); assess symptom relief, weight gain, food tolerance; repeat duplex at 6 months; yearly surveillance thereafter
- CTA or duplex for any recurrent symptoms; evaluate for in-stent restenosis
Standards of Practice
- CIRSE SoP: Mesenteric Ischemia → Loffroy R et al., CVIR 2025 · DOI
Primary References
- Stone JR, Wilkins LR. Acute mesenteric ischemia. Tech Vasc Interventional Rad. 2015;18:24–30.
- Wilkins LR, Stone JR. Chronic mesenteric ischemia. Tech Vasc Interventional Rad. 2015;18:31–37.
- Acosta S, Sonesson B, Resch T. Endovascular therapeutic approaches for acute superior mesenteric artery occlusion. Cardiovasc Intervent Radiol. 2009;32:896–905.
- Beaulieu RJ, Arnaoutakis KD, Abularrage CJ, et al. Comparison of open and endovascular treatment of acute mesenteric ischemia. J Vasc Surg. 2014;59:159–164.
- Oderich GS, Bower TC, Sullivan TM, et al. Open versus endovascular revascularization for chronic mesenteric ischemia: risk-stratified outcomes. J Vasc Surg. 2009;49:1472–1479.
- Oderich GS, Erdoes LS, Lesar C, et al. Comparison of covered stents versus bare metal stents for treatment of chronic atherosclerotic mesenteric arterial disease. J Vasc Surg. 2013;58:1316–1323.
- Hogendoorn W, Hunink MGM, Schlosser FJV, et al. A comparison of open and endovascular revascularization for chronic mesenteric ischemia in a clinical decision model. J Vasc Surg. 2014;60:715–725.