Indications & Patient Selection
Indications
- Primary lung cancer (Stage I NSCLC) — T1a–T1b N0M0; medically inoperable (poor PFTs, COPD, cardiac comorbidities) or patient refuses surgery; ≤3 cm preferred
- Pulmonary metastases — colorectal cancer, sarcoma, melanoma, RCC; oligometastatic (≤5 lesions); resection-refractory or patient preference
- Chest wall malignancy — primary (Askin, Ewing) or metastatic rib lesions with pain
Contraindications
- Central location (<1 cm from central airways or great vessels)
- Lesion abutting heart or pericardium
- Severe COPD (FEV1 <30% predicted)
- Bullous disease at entry site
- Single functioning lung
- Uncorrectable coagulopathy (INR >1.5, platelets <50K)
Best Candidates
- Peripheral tumors (outer one-third of lung parenchyma)
- Lesion size ≤3 cm; optimal ≤2 cm for highest local control rates
- ≥1 cm from bronchi and major vessels
- No endobronchial component
Required Workup
- CT chest — lesion characterization, size, location, vascular proximity
- PET-CT — staging, exclude occult metastatic disease
- PFTs (FEV1 / DLCO) — assess pulmonary reserve
- Bronchoscopy / biopsy if lesion uncharacterized
- INR, CBC with platelets
Pre-Procedure Checklist
Planning & Setup
- CT guidance required — interventional CT suite; CT fluoroscopy or step-and-shoot
- Modality selection: RFA (RITA, Covidien Cool-Tip), MWA (Neuwave 17G), or cryo (small peripheral tumors, near hilum)
- Positioning: prone or lateral decubitus; ipsilateral side down minimizes pleural bleeding and keeps ablated tissue dependent
- Chest tube kit available (20–24 Fr) — pneumothorax incidence 20–40%
- Hydrodissection: 5% dextrose (D5W) for chest wall and diaphragm protection
Anesthesia & Labs
- Local anesthesia + moderate sedation for parenchymal ablation
- General anesthesia for chest wall / rib lesions
- INR <1.5; platelets >50K; hold anticoagulation per SIR guidelines
- Concurrent biopsy if histology unconfirmed — coaxial technique preferred
Relevant Anatomy
Peripheral vs Central Zones
- Peripheral (outer 1/3 of lung) — safer for ablation; lower risk of airway injury; standard approach
- Central (inner 1/3, near hilum) — high-risk zone; thermal injury to bronchi causes bronchial stenosis, bronchopleural fistula, or massive hemoptysis; reserve for carefully selected cases
- Minimum clearance from left/right main bronchi and lobar bronchi: >1 cm required
Fissures & Vascular Structures
- Fissures: avoid transgressing major or minor fissures — significantly increases pneumothorax risk
- Heat sink effect: vessels >3 mm cause incomplete ablation at adjacent margins; probe repositioning and overlapping ablations needed near major vessels
- Diaphragm / pleural proximity: ablation within 1 cm of diaphragm or pleura requires D5W hydrodissection
Procedural Anatomy Summary
The lung parenchyma is divided into peripheral (outer one-third) and central (inner two-thirds near hilum) zones for ablation planning. Peripheral tumors are ideal: probe trajectories avoid crossing fissures, and no major airways or vessels are jeopardized. Central tumors near the hilar vessels and major bronchi require additional planning, often disqualifying them from ablation entirely. The diaphragm and chest wall serve as key hydrodissection targets when the lesion is within 1 cm — 5% dextrose (not saline) is used because it does not conduct radiofrequency current and provides electrical insulation. Pulmonary vessels >3 mm adjacent to the ablation zone act as heat sinks, cooling the tissue and producing incomplete margins; MWA has less susceptibility to heat sink than RFA due to higher energy deposition rates.
Step-by-Step Technique
Positioning & CT Planning Scan
Local Anesthesia & Skin Entry
Probe Placement
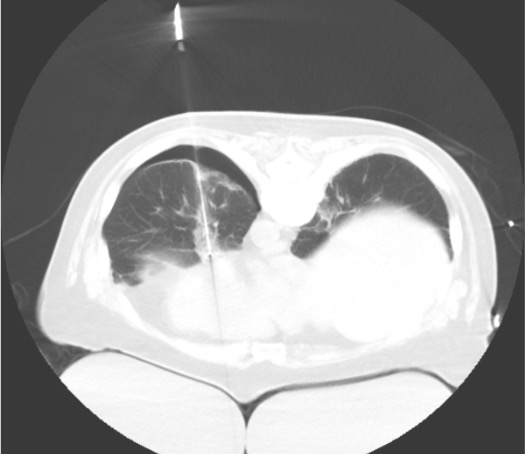
Radiofrequency Ablation (RFA)
Microwave Ablation (MWA)
Post-Ablation CT
Pneumothorax Management
Recovery & Discharge
Ablation Modality Comparison — Lung Tumors
| Feature | RFA | MWA | Cryoablation |
|---|---|---|---|
| Heat sink effect | Significant (major limitation near vessels) | Minimal (higher energy deposition) | Moderate (ice ball may be preserved) |
| Ablation zone | Smaller; variable; affected by impedance roll-off | Larger; more reproducible; sphere-shaped | Visible ice ball on CT; excellent real-time control |
| Real-time monitoring | Temperature / impedance; indirect | Indirect (CT confirmation) | Direct visualization of ice ball on CT |
| Chest wall safety | Risk of neuropathy / pain if probe near chest wall | Risk of thermal injury; D5W hydrodissection needed | Safest near chest wall; ice ball protective for nerves |
| Main lung limitation | Heat sink, roll-off in aerated lung | Charring at probe tip if power too high | Longer procedure; freeze-thaw cycles required; bronchopleural fistula risk near airways |
| Preferred for | Standard peripheral tumors ≤2 cm | Preferred modality; tumors up to 3 cm; near vessels | Small peripheral; near hilum; chest wall |
Community Cards
CT Landmarks
Immediate Post-Ablation Findings (Normal)
- Ground-glass opacity (GGO) halo — surrounds treated lesion; represents reactive inflammation and hemorrhage; a 1-cm GGO halo around the entire lesion = adequate margin; this is the primary adequacy endpoint
- Cavitation — seen with RFA; thin-walled cavity within ablation zone; normal post-ablation change; does NOT indicate recurrence if thin-walled and stable or regressing
- GGO progressively retracts on follow-up scans over 3–6 months; stable retraction = treatment success
Follow-up Findings (Concerning)
- "Target sign" on follow-up CT — central low density (necrosis) + peripheral rim enhancement = local recurrence; biopsy recommended to confirm before re-treatment
- Growing soft tissue nodule at ablation margin — local progression; compare to prior CT; PET-CT may help differentiate from post-ablation fibrosis
- New or enlarging enhancement at 3-month or 6-month follow-up CT → local recurrence until proven otherwise
Pneumothorax Assessment on CT
| Size | Definition | Management |
|---|---|---|
| Small | <15% of hemithorax (thin rim; <2 cm at apex) | Conservative; serial CXR at 1h and 2h; observe in recovery |
| Moderate | 15–33% of hemithorax; >1/3 of lateral chest wall | Small-bore chest tube (8–14 Fr) if symptomatic; consider tube if increasing |
| Large / Increasing | >33% of hemithorax or actively enlarging on serial imaging | Chest tube required; 20–24 Fr if large or tension physiology present |
Troubleshooting
Air Accumulates in Pleural Space While Ablation In Progress
If small and patient is stable: complete the ablation first, then treat the pneumothorax. If large or tension physiology develops (hypoxia, tracheal deviation, hemodynamic compromise): stop ablation immediately and place chest tube emergently. After treatment is complete, resume ablation if margins were inadequate and patient can tolerate continued procedure.
Rib, Scapula, or Bony Structure Obstructs Needle Path
Change access angle: use a more cranial or caudal intercostal space; angulate the needle craniocaudally within the same interspace. For posterior lesions obstructed by scapula: position arm above head to rotate scapula laterally. Consider an anterior or lateral approach. If all standard approaches fail, a transfissural or translobar trajectory may be necessary with increased pneumothorax counseling.
GGO Halo Does Not Achieve ≥1 cm Margin on Post-Ablation CT
Perform a second overlapping ablation: reposition the probe to the undertreated margin and ablate again. For MWA, multiple overlapping activations are well tolerated. For RFA, allow probe to cool to avoid impedance roll-off before repositioning. Document final coverage on CT and report the percentage of margin achieved. If margin still inadequate at a central/hilar location, SBRT referral is appropriate.
Ground-Glass Opacity or Inflammatory Change Obscures Target Lesion
Fuse the current procedural CT with the pre-procedure planning CT using CT workstation co-registration. Use tracked electromagnetic navigation (e.g., LungPoint) if available. Target the center of the GGO region that corresponds to the original solid nodule position. If completely invisible, abort ablation and reschedule after GGO resolves; do not ablate a target that cannot be confirmed.
Patient Coughs Blood During or After Probe Activation
Minor blood-streaking: expected and normal; reassure patient; continue ablation with observation. Significant hemoptysis (frank blood, >a few mL): stop ablation immediately; roll patient to affected side (bleeding side down); notify anesthesia. Major hemoptysis: secure airway; bronchoscopy urgently; interventional bronchoscopy or bronchial artery embolization if massive. Consider CT angiography to identify source.
Complications
Common Complications
- Pneumothorax (20–40% incidence) — most common complication; 10–15% require chest tube; risk factors: emphysema, bullous disease, traversed fissures, multiple probe passes, peripheral location
- Pleural effusion — reactive; common post-ablation; no intervention unless symptomatic (progressive dyspnea or large effusion); thoracentesis if needed
- Pain — pleuritic or chest wall; expected; NSAIDs and opioids PRN; severe chest wall pain with chest wall ablation may require nerve block
Serious Complications
- Pulmonary hemorrhage — usually self-limiting intrapulmonary; bronchovascular fistula with major hemoptysis rare (<1%); bronchial artery embolization or surgery if massive
- Bronchopleural fistula — rare; persistent air leak >5 days; chest tube; surgical consultation; risk higher with central tumors and prior radiation
- Thermal injury to adjacent structures — diaphragm, pericardium, intercostal nerve; hydrodissection with D5W reduces risk
- Local recurrence — 15–30% for lesions >2 cm at 1 year; re-ablation or SBRT referral
Local Control by Tumor Size — Summary
| Tumor Size | 2-Year Local Control Rate | Recommendation |
|---|---|---|
| ≤2 cm | >90% | Optimal candidate; ablation strongly preferred |
| 2–3 cm | 65–75% | Acceptable; discuss SBRT as alternative or sequential therapy |
| >3 cm | <50% | Poor local control; SBRT preferred unless ablation uniquely indicated |
Critical Pearls
References & Resources
Key Guidelines
- SIR / CIRSE Standards of Practice: Image-Guided Tumor Ablation of Lung Tumors — indications, technical standards, and quality improvement
- NCCN Clinical Practice Guidelines: Non-Small Cell Lung Cancer — ablation as option for medically inoperable stage I NSCLC
- ACR Appropriateness Criteria: Nonsurgical Treatment for Locally Advanced NSCLC
Primary References
- Faintuch S, Salazar GM, eds. Interventional Oncology. Thieme; 2016. Ch. 3: McGraw C et al. Lung Tumor Ablation.
- Lencioni R, Crocetti L, Cioni R, et al. Response to radiofrequency ablation of pulmonary tumours: a prospective, intention-to-treat, multicentre clinical trial (the RAPTURE study). Lancet Oncol. 2008;9(7):621–628.
- Lanuti M, Sharma A, Digumarthy SR, et al. Radiofrequency ablation for treatment of medically inoperable stage I non-small cell lung cancer. J Thorac Cardiovasc Surg. 2009;137(1):160–166.
- Hiraki T, Gobara H, Iishi T, et al. Percutaneous radiofrequency ablation for clinical stage I non-small-cell lung cancer: results in 20 nonsurgical candidates. J Thorac Cardiovasc Surg. 2007;134(5):1306–1312.
- Pusceddu C, Sotgia B, Fele RM, Melis L. CT-guided thin needles percutaneous cryoablation (CA) in patients with primary and secondary lung tumors. Eur J Radiol. 2013;82(5):e246–e253.