Indications & Patient Selection
Indications
- Refractory visceral pelvic pain from pelvic organs — cervical, endometrial, or ovarian cancer; bladder cancer; rectal cancer; locally advanced prostate cancer
- Non-oncologic pelvic pain — endometriosis, interstitial cystitis, chronic prostatitis, uterine fibroids (peri-embolization)
- Superior hypogastric plexus (SHP) mediates afferent pain from pelvic viscera below the peritoneal reflection; blockade interrupts this pathway
- Diagnostic / therapeutic block: lidocaine or bupivacaine ± steroid → assess response; if ≥50% relief → proceed to neurolysis
- Neurolysis: 6–10% absolute ethanol or 6–10% phenol for durable relief in cancer pain (≥3–6 months)
- Caudal block (sacral hiatus approach): perineal pain, tailbone pain, S2–S5 radiculopathy; useful in diffuse sacral metastatic disease
- Ganglion impar block: perianal, perineal burning/sitting-induced pain — coccydynia or anorectal cancer pain
Contraindications & Workup
- Absolute contraindications: active pelvic infection, uncorrectable coagulopathy, allergy to contrast or phenol
- Relative: bowel loops in planned needle trajectory (CT mandatory to identify and avoid), anatomic aberrancy precluding safe access
- Pre-procedure workup: characterize pain (visceral burning/pressure vs. somatic/radicular), VAS score baseline, prior pelvic CT or MRI to review iliac vessel course and bowel position
- Pain quality guiding target: diffuse visceral pelvic pain → SHP; perineal/perianal burning → ganglion impar; sacral radiculopathy → caudal epidural
- Diagnostic block first: always confirm ≥50% relief with local anesthetic block before committing to irreversible neurolysis
Pre-Procedure Checklist
Relevant Anatomy
Superior Hypogastric Plexus (SHP)
- Location: Retroperitoneal sympathetic plexus at the L5–S1 level, anterior to the L5-S1 intervertebral disc and the bifurcation of the common iliac vessels
- Lies between the common iliac arteries at their bifurcation into external and internal iliac arteries; ureters run just lateral
- Afferent pain signals from: uterus, cervix, upper vagina, bladder, prostate, sigmoid colon, rectum — all viscera below the peritoneal reflection
- Does not carry pain from ovaries and fallopian tubes (celiac/inferior mesenteric plexus via T10–L2)
- Receives fibers from celiac and inferior mesenteric plexuses, arising from L4 to S1
- Key surgical landmark: the aortic bifurcation is at L4; the common iliac bifurcation into external/internal iliacs is at L5-S1 — this is the SHP target
Related Structures: Ganglion Impar & Caudal Canal
- Ganglion impar: most caudal sympathetic ganglion; single midline structure located in the retroperitoneum at the sacrococcygeal junction, anterior to the sacrum, posterior to the rectum
- Carries pain from perineum, anus, distal rectum, distal urethra, vulva — burning, sitting-induced perianal pain is the hallmark
- Blockade via single midline needle through the sacrococcygeal ligament; contrast shows comma-shaped presacral spread
- Sacral canal (caudal epidural space): extends from the sacral hiatus (midline gap at caudal sacrum between the sacral cornua) cephalad; contains S1–S5 nerve roots, dural sac ending at S2
- Caudal epidural injection covers S2–S5 nerve roots — useful for perineal pain, sacral metastatic disease, pudendal neuralgia
- Inferior hypogastric plexus: mixed sympathetic/parasympathetic
▶ CT-guided inferior hypogastric nerve block; innervates bladder, urethra, corpora cavernosa; not purely sympathetic — transsacral blockade described for refractory pelvic pain
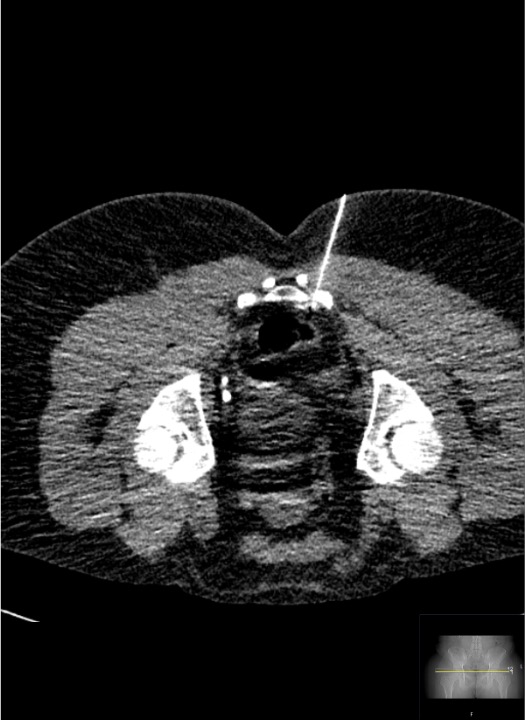 Inferior hypogastric plexus block: CT-guided needle in parametrial/pararectal space — inferior to the bifurcation of common iliac vessels, targeting pelvic visceral afferents.
Inferior hypogastric plexus block: CT-guided needle in parametrial/pararectal space — inferior to the bifurcation of common iliac vessels, targeting pelvic visceral afferents.
Technique
CT-guided bilateral posterior paravertebral approach + community cards
Supplies
Steps
Position & scout CT
Bilateral skin entry — L4-L5 level, lateral to L5 transverse process
Advance Chiba needles anteromedially
CT confirmation of retroperitoneal tip position
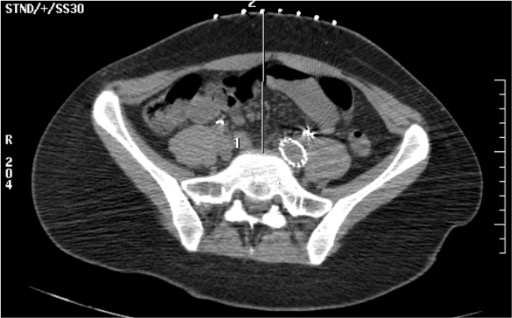
Aspirate — no blood or bowel content
Test injection — iohexol contrast
Inject block solution
Neurolysis (if indicated)
Remove needles & monitor
Steps
Prone position; identify L5 junction
Oblique fluoroscopy needle advance
AP + lateral confirmation; contrast spread
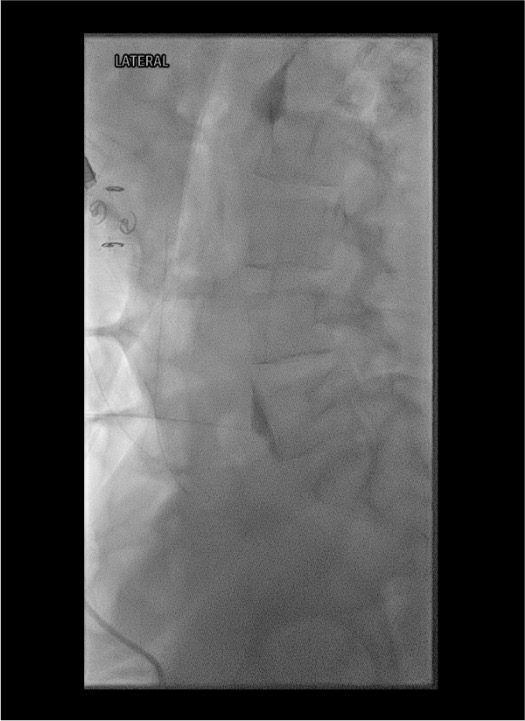
Inject therapeutic solution
Prone position with pillow under iliac crest
Advance needle through L5-S1 disc midline
Contrast confirmation
Inject and withdraw with intradiscal cefazolin
CT Landmarks & Caudal Block Technique
SHP Target — CT Anatomy
- Primary target: retroperitoneal fat space directly anterior to the L5-S1 intervertebral disc, medial to the common iliac artery bifurcation
- Bilateral needle configuration: two Chiba needles form a "V" with tips converging at the L5-S1 anterior disc/junction
- Correct contrast spread: diffuse retroperitoneal cloud anterior to L5-S1 disc, spreading laterally between the common iliac arteries — no bowel opacification, no intravascular linear flow
- Aortic bifurcation vs. iliac bifurcation: aorta bifurcates at L4; the common iliacs bifurcate into external/internal iliacs at L5-S1 — the SHP lies at this lower (L5-S1) bifurcation
- Depth guide: on axial CT, tips should be in the retroperitoneal fat immediately anterior to the psoas and vertebral body, posterior to the iliac vessels
- Avoid: iliac vein (posterior to artery), ureter (lateral to plexus), bowel loops (replan if any are in path)
Ganglion Impar — CT/Fluoroscopic Landmarks
- Target: retroperitoneal space at the sacrococcygeal junction anteriorly — between the anterior sacrum/coccyx and the posterior rectum
- Needle approach: single midline needle through the sacrococcygeal ligament/junction, just anterior to the sacrococcygeal disc — on lateral fluoroscopy, tip just beyond the ventral cortex of the bone
- Correct contrast pattern: comma-shaped spread in the presacral retroperitoneum on lateral projection
- Indication: perianal and perineal burning pain, sitting-induced pain, coccydynia, anorectal cancer pain — distinct from visceral pelvic organ pain treated by SHP block
- Injection: 3–5 mL 0.5% bupivacaine + 1 mL dexamethasone 4 mg for diagnostic/therapeutic; absolute ethanol 3–5 mL for neurolysis
Caudal Block Technique (Sacral Hiatus Approach)
Position & identify sacral hiatus
Needle insertion
Confirm epidural position
Inject therapeutic solution
Troubleshooting
Blood return on aspiration — needle in iliac vessel
Likely cause: Needle tip has traversed the iliac vein (which lies posterior to the artery) or the common iliac artery. The iliac vein is particularly susceptible as it lies immediately posterior and lateral to the artery.
Next step: Withdraw the needle without injecting. Obtain CT to re-plan needle trajectory — adjust the angle to pass more medially or at a different craniocaudal level to avoid the vessel. Reassess for any expanding retroperitoneal hematoma before proceeding. CT guidance essential; intravascular injection of ethanol is catastrophic.
Bowel loop in needle path on CT planning
Likely cause: Sigmoid colon, small bowel, or redundant descending colon overlies the planned bilateral paravertebral trajectory — more common on the left. Mobile bowel can shift with positioning.
Next step: Adjust needle trajectory angle on CT planning — try a more lateral entry or slightly different craniocaudal level to find a bowel-free window. Consider repositioning the patient slightly oblique. If the anterior transabdominal approach is used, bowel preparation prior to procedure and Trendelenburg positioning can displace bowel. Reserve transrectal approaches for rare cases with no posterior access.
Inadequate retroperitoneal contrast spread on test injection
Likely cause: Needle tip not sufficiently anterior — still in muscle or posterior to anterior vertebral cortex; or tip has exited the retroperitoneal fat plane.
Next step: Re-confirm CT position. Advance needle slightly more anteriorly until tip is clearly in retroperitoneal fat anterior to the disc. Increase test injection volume to 3–4 mL. If spread is still inadequate or loculated, consider slightly redirecting the needle tip medially or laterally to find less-resistive tissue.
No pain relief after neurolysis
Likely cause: Pain may have a somatic or radicular component not mediated by the SHP (e.g., bony metastases, sacral nerve root involvement, leptomeningeal disease); or the pain generator is the perineum/anus (ganglion impar territory rather than SHP territory).
Next step: Reassess pain quality — is there a burning, sitting-induced, perianal component? If yes, consider ganglion impar block/neurolysis at sacrococcygeal junction. If pain has radicular features (radiation down leg, dermatomal distribution), reassess for nerve root involvement with MRI and consider targeted epidural steroid or nerve root block. Caudal block if diffuse sacral nerve root pain.
Contrast flows intravascularly on test injection
Likely cause: Needle tip in epidural venous plexus or peri-iliac vascular plexus — venous structures in this region are thin-walled and easily entered without blood return on aspiration.
Next step: Withdraw needle 2–3 mm, reaspirate, and retest with 1 mL contrast under real-time CT or fluoroscopy. Never inject therapeutic solution — and especially never inject ethanol — with any intravascular contrast flow. Reposition needle until clean retroperitoneal spread is confirmed.
Complications
Immediate / Periprocedural
- Vascular injury (common iliac artery or vein): most serious immediate risk; avoided with CT guidance and aspiration before every injection. If significant hemorrhage suspected post-procedure, obtain CT and manage per vascular surgery protocol.
- Intravascular injection of ethanol or phenol: catastrophic — causes acute vessel wall injury, thrombosis, or embolic events. Mandatory: aspirate + contrast confirmation before ANY neurolytic injection.
- Bowel perforation: rare with CT guidance; if suspected (fever, peritoneal signs post-procedure), obtain CT abdomen/pelvis with contrast. Antibiotic prophylaxis if bowel entry suspected. Usually managed conservatively if single needle pass and no bowel contents aspirated.
- Bladder dysfunction (temporary urinary retention or urgency): from sympathetic blockade of the bladder; usually resolves within 24–48 hours. Monitor voiding before discharge.
Delayed
- Ethanol neuritis: burning pain in the pelvis/lower back for 1–7 days post-neurolysis — warn patient before discharge. Treat with NSAIDs, short gabapentin course (300–600 mg TID for 5–7 days), or brief opioid taper if severe.
- Lower extremity weakness or numbness: rare; from spread of neurolytic agent to lumbar plexus or L4-L5 nerve roots. Use minimum effective volume (5–8 mL per side) to reduce risk. Usually transient if from local anesthetic; potentially prolonged if from ethanol. Neurology consultation if persistent.
- Retroperitoneal abscess / infection: very rare with posterior CT-guided approach. More common with transdiscal approach if intradiscal cefazolin is omitted. Presents with fever, back pain, elevated inflammatory markers 1–2 weeks post-procedure. CT-guided drainage if fluid collection identified.
- Discitis (transdiscal approach): prevented by intradiscal cefazolin 50 mg on needle withdrawal. Risk of diskitis is the primary reason CT-guided posterior paravertebral approach is preferred over transdiscal.
- Sexual dysfunction: rare; from sympathetic blockade — usually transient with block, may be more lasting with neurolysis. Counsel patients with preserved sexual function before neurolysis.
Critical Pearls
References
Approach Comparison
| Approach | Guidance | Needles | Key Advantage | Key Risk |
|---|---|---|---|---|
| Posterior paravertebral (bilateral) | CT (preferred) or fluoro | Two 22G Chiba | Complete bilateral coverage; avoids disc | Vascular injury; bowel in path |
| Transdiscal | Fluoroscopy or CT | One 22–25G needle | Single needle; shorter procedure time | Discitis risk — give intradiscal cefazolin |
| Anterior transabdominal | CT or ultrasound | 22G needle infraumbilical | Less technically demanding; useful if posterior access blocked | Bowel/mesenteric injury; prep bowel first |
| Ganglion impar (sacrococcygeal) | Fluoroscopy or CT | One 22G spinal needle midline | Targets perineal/perianal pain specifically | Rectal perforation (rare) |
| Caudal block (sacral hiatus) | Fluoroscopy or CT | 18–20G angiocath or Tuohy | Covers S2–S5; useful for sacral met disease | Intrathecal injection if dura punctured |
Citations
- Prologo JD, Ray CE Jr., eds. Advanced Pain Management in Interventional Radiology: A Case-Based Approach. Thieme; 2024. Ch. 44–46 (Williams R, Murphy T; Gilyard S, Nezami N, Kokabi N).
- Plancarte R, Amescua C, Patt RB, Aldrete JA. Superior hypogastric plexus block for pelvic cancer pain. Anesthesiology. 1990;73(2):236–239.
- Plancarte R, de Leon-Casasola OA, El-Helaly M, Allende S, Lema MJ. Neurolytic superior hypogastric plexus block for chronic pelvic pain associated with cancer. Reg Anesth. 1997;22(6):562–568.
- Gamal G, Helaly M, Labib YM. Superior hypogastric block: transdiscal versus classic posterior approach in pelvic cancer pain. Clin J Pain. 2006;22(6):544–547.
- Pereira K, Morel-Ovalle LM, Taghipour M, et al. Superior hypogastric nerve block (SHNB) for pain control after uterine fibroid embolization (UFE): technique and troubleshooting. CVIR Endovasc. 2020;3(1):50.
- Pereira K, Morel-Ovalle LM, Wiemken TL, et al. Intraprocedural superior hypogastric nerve block allows same-day discharge following uterine artery embolization. J Vasc Interv Radiol. 2020;31(3):388–392.
- Kanazi GE, Perkins FM, Thakur R, Dotson E. New technique for superior hypogastric plexus block. Reg Anesth Pain Med. 1999;24(5):473–476.
- Erdine S, Yucel A, Celik M, Talu GK. Transdiscal approach for hypogastric plexus block. Reg Anesth Pain Med. 2003;28(4):304–308.
- Yang X, You J, Tao S, et al. Computed tomography-guided superior hypogastric plexus block for secondary dysmenorrhea in perimenopausal women. Med Sci Monit. 2018;24:5132–5138.
- Choi JW, Kim WH, Lee CJ, et al. The optimal approach for a superior hypogastric plexus block. Pain Pract. 2018;18(3):314–321.
References & Resources
Key Guidelines
- ISIS Practice Guidelines for Hypogastric Plexus Block
- ASRA Guidelines for Sympathetic Nerve Blocks
Primary References
- Plancarte R et al. Superior hypogastric plexus block for pelvic cancer pain. Anesthesiology. 1990;73(2):236-239.
- Prologo JD, Ray CE Jr., eds. Advanced Pain Management in Interventional Radiology. Thieme; 2024. Ch. 44–46: Hypogastric and Pelvic Neurolytic Blocks.
- de Leon-Casasola OA et al. Neurolytic superior hypogastric plexus block for chronic pelvic pain associated with cancer. Pain. 1993;54(2):145-151.