Indications & Treatment Methods
Indications for Treatment
- Post-catheterization pseudoaneurysm — iatrogenic is the most common etiology (0.6–7.7% of femoral catheterizations)
- Symptomatic — pain, expanding hematoma, pulsatile mass
- >2 cm diameter — typically requires intervention (PSA <2 cm spontaneously thromboses in ~85% of cases)
- Rapidly expanding or compromising overlying skin
- Untreated PSA: high risk of rupture, thromboembolism, skin necrosis
Contraindications
- Local infection at puncture site
- Thrombin allergy (prior bovine thrombin exposure — anaphylaxis risk)
- Concomitant AV fistula — thrombin injection is ineffective if significant AVF present; requires coil embolization or surgery
- Severe distal limb ischemia — thrombin injection requires caution (embolization risk)
- Relative: very wide neck PSA without ability to compress during injection
Treatment Method Decision Guide
| Method | Success Rate | Best For | Limitations |
|---|---|---|---|
| US-guided thrombin injection (UGTI) | 95–100% | First-line for most PSA; effective regardless of anticoagulation status | Risk of distal embolization; ineffective if AVF present |
| US-guided compression (UGCR) | 51–73% (anticoagulated); >90% (normal coag) | Second-line; no thrombin required | Painful; requires sedation; 4–6h supine; 20% recurrence |
| Covered stent | High | Thrombin failure, complex/multilobed PSA, wide-neck PSA | Requires arterial access; moderate bleeding risk |
| Surgical repair | High | Large PSA with pending rupture; significant hematoma requiring evacuation | Complication rate up to 21%; requires OR |
Pre-Procedure Planning
Imaging & Diagnosis
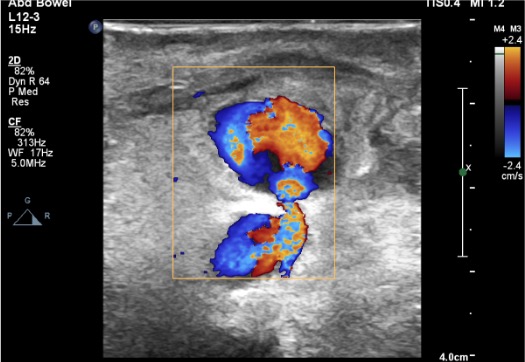
- Duplex US confirming PSA with characteristic to-and-fro flow (“yin-yang” sign) on color Doppler
- Measure neck dimensions (length, width) and sac dimensions
- Identify communication with CFA, SFA, or profunda femoris
- Evaluate for AV fistula component (critical — thrombin ineffective if AVF present)
- Distinguish from hematoma (no internal flow), true aneurysm, mycotic aneurysm
Patient Preparation
- Hold anticoagulation if possible (although UGTI is successful regardless of anticoagulation status)
- Labs: CBC, PT/INR, PTT if on heparin
- Pre- and post-procedural ankle-brachial indices and pulse assessment
- Local anesthesia only for thrombin injection (no sedation required)
- Informed consent: discuss risk of distal embolization, recurrence, potential need for escalation
Relevant Anatomy
Femoral Artery Anatomy
- Common femoral artery (CFA) — continuation of external iliac artery below inguinal ligament; most common site of catheterization and PSA formation
- Superficial femoral artery (SFA) — anterolateral continuation of CFA after bifurcation
- Profunda femoris artery — posterolateral branch of CFA; PSA can arise from any of these vessels
- PSA location relative to CFA bifurcation is critical for planning — influences neck access and compression approach
Pseudoaneurysm Characteristics
- Neck — communication between parent artery and PSA sac; narrow neck = lower embolization risk during thrombin injection; wide neck = higher risk, may need compression or balloon protection
- Sac — contained rupture of arterial wall; may be unilobed or multilobed
- To-and-fro flow — systolic inflow into sac, diastolic outflow back to artery; pathognomonic on Doppler
- “Yin-yang” sign — swirling bidirectional color flow within the sac on color Doppler
Differential Diagnosis
Hematoma: no internal flow on Doppler, no neck communication. AV fistula: continuous high-velocity turbulent flow between artery and vein, low-resistance arterial waveform. Mycotic (infected) aneurysm: true aneurysmal dilation of vessel wall with periarterial inflammation and gas; requires antibiotics and often surgical management. True aneurysm: involves all three layers of arterial wall (intima, media, adventitia); distinct from PSA where blood is contained only by extravascular tissue.
Technique
US Assessment & Planning
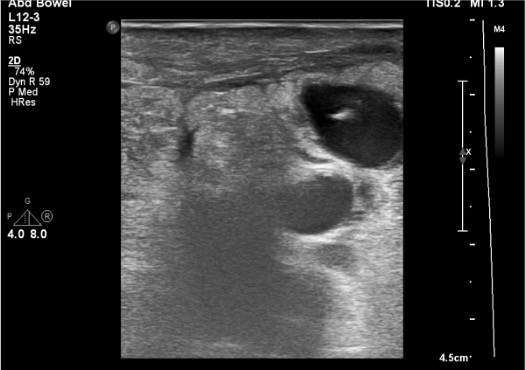
Needle Placement
Thrombin Injection
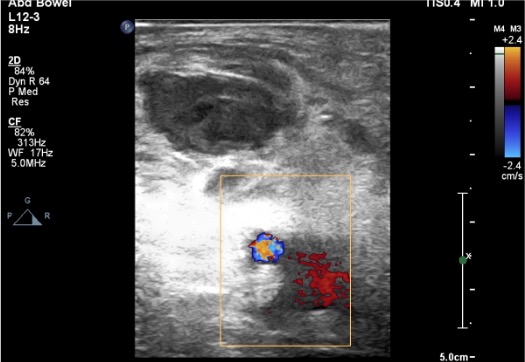
Confirmation & Post-Procedure Check
Alternative Techniques (If Thrombin Fails or Complex PSA)
- Covered stent placement: for multilobed PSA, wide-neck PSA, or thrombin injection failure. Arterial access via contralateral femoral approach; covered stent deployed across the PSA neck to exclude the sac. Moderate bleeding risk.
- US-guided compression repair (UGCR): second-line option. Compress PSA neck under US guidance until flow ceases; maintain 20–30 min. Painful; requires sedation. Lower success rate (51–73% in anticoagulated patients). Not effective for suprainguinal or post-operative PSA.
- Surgical repair: for large PSA with pending rupture, significant hematoma requiring evacuation, failed percutaneous treatment. Complication rate up to 21% (MI, death, bleeding, lymphocele, infection).
Community Cards
Ultrasound Landmarks
Key US Findings
- Gray-scale: hypoechoic/anechoic fluid collection adjacent to femoral artery; may see layered thrombus within sac
- Color Doppler “yin-yang” sign: swirling bidirectional color flow within the PSA sac — pathognomonic
- Spectral Doppler at neck: characteristic to-and-fro waveform — systolic inflow into sac, diastolic outflow back to artery
- Neck visualization: narrow channel connecting sac to parent artery; measure length and width
Needle Placement Landmarks
- Target needle tip at the apex (far wall) of the PSA sac, away from the neck
- Inject into the region where flow is directed away from the neck on color Doppler
- For multilobed PSA: inject the most superficial lobe first; if other lobes do not thrombose, inject each separately
- Compress the neck with the US probe during injection to prevent thrombin reflux into parent artery
Troubleshooting
Thrombin Into Parent Artery — Limb-Threatening
If thrombin enters the native femoral artery, distal thrombosis and limb ischemia can result. Prevention: always inject into the sac center/apex, never near the neck. Compress neck during injection. Use slow injection rate and stop immediately if thrombus is seen extending toward the neck. Treatment: emergent catheter-directed thrombolysis with TPA or surgical thrombectomy. Check distal pulses immediately after every injection.
Multiple Communicating Sac Lobes
Inject the most superficial lobe first under US guidance. If other lobes do not thrombose spontaneously, perform additional targeted injections into each lobe separately. Multilobed PSA may ultimately require covered stent if thrombin injection fails to thrombose all lobes.
Large Neck Communication Increases Embolization Risk
Compress neck firmly with US probe during thrombin injection. Consider placing an endovascular balloon via contralateral access to protect the native artery during injection. If wide neck prevents safe thrombin injection, proceed to covered stent placement.
PSA Recurs After Thrombin Injection
Recurrence rate after UGTI is approximately 5–10%. Repeat thrombin injection can be performed (note: increased embolization risk with re-treatment of persistent necks). ~66% of persistent necks regress spontaneously. If repeat thrombin injection fails, escalate to covered stent placement or surgical repair.
Concomitant Arteriovenous Fistula Detected
Thrombin injection is ineffective if a significant AV fistula is present (continuous high-velocity flow prevents thrombosis). Requires concomitant coil embolization of the fistulous tract, covered stent, or surgical repair. Always evaluate for AVF on duplex US before proceeding with thrombin injection.
Complications
Serious Complications
- Distal embolization — thrombin entering parent artery causing downstream thrombosis; rare but limb-threatening; treat with catheter-directed TPA or surgical thrombectomy
- Anaphylaxis to bovine thrombin — reported in patients with prior bovine thrombin exposure; avoid re-exposure; consider human thrombin alternative
- Nontarget arterial thrombosis — intraluminal clot propagation from PSA into femoral artery; may resolve spontaneously or require intervention
Minor Complications & Expected Outcomes
- Recurrence (5–10%) — PSA re-establishes flow after initial thrombosis; repeat UGTI or escalate
- Transient loss of distal pulse — may occur and typically returns within 24h; monitor closely
- Local infection / abscess — rare; one case report of abscess requiring I&D
- Soleal vein thrombosis — reported rarely; monitor for DVT symptoms
- Persistent hematoma — thrombosed PSA sac remains palpable; resolves over weeks
Pearls & Pitfalls
References & Resources
Primary References
- Krueger K, Zaehringer M, Strohe D, et al. Postcatheterization pseudoaneurysm: results of US-guided percutaneous thrombin injection in 240 patients. J Vasc Interv Radiol. 2005;16(1):57–62.
- Morgan R, Belli AM. Current treatment methods for postcatheterization pseudoaneurysms. J Vasc Interv Radiol. 2003;14(6):697–710.
- Padidar AM, Kee ST, Razavi MK. Treatment of femoral artery pseudoaneurysms using ultrasound-guided thrombin injection. Tech Vasc Interv Radiol. 2003;6(2):96–102.
- Kang SS, Labropoulos N, Mansour MA, et al. Percutaneous ultrasound guided thrombin injection: a new method for treating postcatheterization femoral pseudoaneurysms. J Vasc Surg. 1998;28:1120–1121.
- Paulson EK, Nelson RC, Mayes CE, et al. Sonographically guided thrombin injection of iatrogenic femoral pseudoaneurysms: further experience of a single institution. Am J Roentgenol. 2001;177:309–316.
- Pezzullo JA, Dupuy DE, Cronan JJ. Percutaneous injection of thrombin for the treatment of pseudoaneurysms after catheterization. Am J Roentgenol. 2000;175:1035–1040.