Indications / Contraindications
⚠ Sentinel Bleed — Do Not Ignore
Any small bleed from drain output, hematemesis, hematochezia, or unexplained falling Hgb within 2–4 weeks of Whipple = GDA stump problem until proven otherwise. Do NOT wait for massive hemorrhage — the sentinel bleed is your intervention window.
Background and Indications
- Post-pancreaticoduodenectomy (Whipple) hemorrhage: rare (3–8%) but potentially fatal (10–40% mortality)
- GDA stump eroded by pancreatic juice (fistula), infection, or pseudoaneurysm formation
- Delayed hemorrhage (>24h post-op) — endovascular first line; IR preferred over re-operation
- Hemodynamically unstable post-surgical patient
- Sentinel bleed: IR evaluation — do not wait for massive hemorrhage
- Other post-Whipple sites: right hepatic artery, proper hepatic artery, SMA branches, hepaticojejunostomy
Contraindications
- Early hemorrhage (<24h): Surgery is often preferred — anastomotic or technical bleeding more amenable to operative repair
- Hemodynamic instability not responding to resuscitation: Emergent OR — cannot wait for IR setup
- Variant hepatic anatomy not adequately characterized before stenting — review CTA first
- Active infection of planned stent zone — relative contraindication; treat underlying infection while proceeding with embolization
Pre-Procedure Checklist
Relevant Anatomy
GDA Anatomy and Post-Whipple Changes
- Normal: Celiac trunk → common hepatic artery (CHA) → GDA arises at the "genu" of CHA, at the junction with the proper hepatic artery
- After Whipple (pancreaticoduodenectomy): GDA is ligated/stapled at its origin from CHA. The stump is exposed to duodenal/pancreatic fluid.
- Pseudoaneurysm formation: Activated pancreatic enzymes erode the GDA stump suture line → weak point → pseudoaneurysm → blowout
- CHA → proper hepatic artery → right and left hepatic arteries. Blood flow to liver maintained after Whipple via CHA → proper hepatic (GDA branch no longer present).
Critical Hepatic Artery Considerations
- Covered stent principle: Stent is placed in CHA spanning the GDA stump origin, maintaining hepatic perfusion while excluding the bleeding stump. Hepatic flow is preserved — liver does not lose its blood supply.
- Variant anatomy (CRITICAL): Replaced right hepatic artery from SMA in ~20% of population. Must identify on CTA before any intervention. Covering a replaced hepatic = hepatic ischemia.
- If coil embolization used instead: Risk of hepatic ischemia if CHA/proper hepatic artery supply is compromised. Check collateral development first.
Technique
Default RadCall approach · share your own below
Supplies
Steps
Celiac angiogram
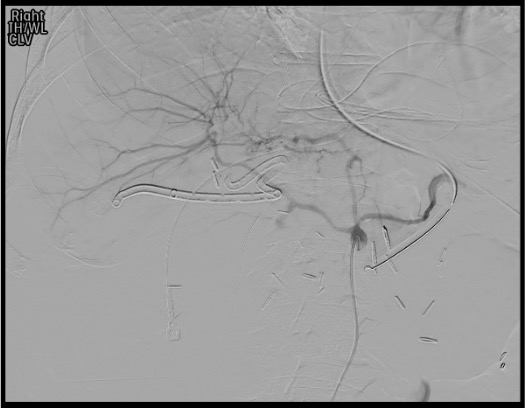
Selective CHA angiogram and Exchange
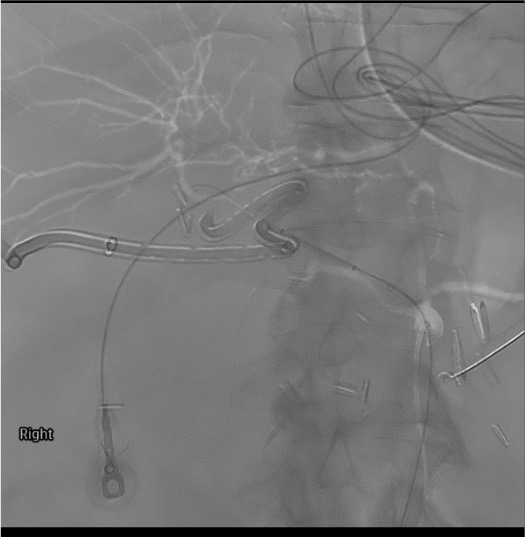
Treatment decision: covered stent vs coil embolization
Covered stent deployment
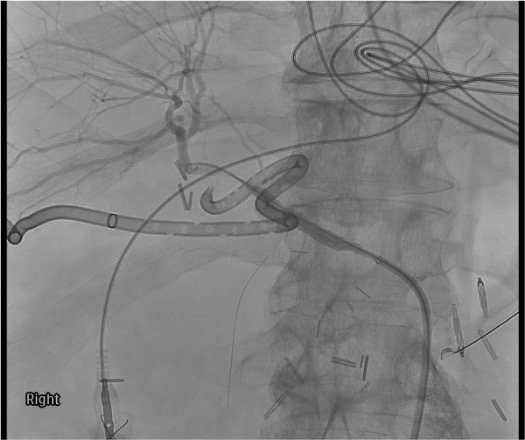
Completion angiogram
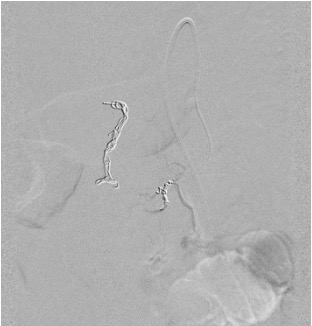
Troubleshooting
Cannot access GDA stump — stump too short or vessel too tortuous
Likely cause: Short surgical stump, post-operative adhesions, vessel tortuosity at celiac origin.
Next step: Coaxial microcatheter for additional support. Consider right radial access — provides excellent anterior approach to celiac axis, often with superior curve. Direct sac embolization with coils from the CHA side as last resort.
Variant hepatic artery anatomy (replaced right or left hepatic)
Likely cause: Replaced right hepatic from SMA (~20%); replaced left hepatic from left gastric (~10%). Must be identified on pre-procedure CTA — not discovered for the first time on the table.
Next step: Stop — review CTA anatomy before proceeding with any intervention. If replaced right hepatic from SMA is present and CHA gets occluded, hepatic supply is unaffected (right liver gets flow from SMA). Adjust treatment strategy accordingly. Never proceed without understanding the hepatic anatomy.
Covered stent thrombosis on follow-up CT
Likely cause: Very common (~50% long-term) — stent in an infected field with sluggish hepatic flow or suboptimal stent sizing.
Next step: Do NOT panic. Hepatic collaterals typically develop after Whipple, making stent thrombosis usually asymptomatic. Treat only if symptomatic (rising LFTs, hepatic infarcts). Expectant management in most cases. Anticoagulation in selected symptomatic cases.
Recurrent bleed after stent placement
Likely cause: Bleeding from jejunal branches, hepaticojejunostomy site, or other post-operative vessels — not from GDA stump itself.
Next step: Repeat angiography — complete celiac + SMA survey. Identify new bleeding source. Targeted superselective embolization of new vessel. Involve surgery early if repeat failure.
Complications
Primary Risks
- Hepatic ischemia/infarction — most feared; risk reduced significantly with covered stent over embolization alone
- Stent thrombosis — ~50% long-term; usually benign due to hepatic collateral development; treat only if symptomatic
- Rebleeding — from non-GDA vessel (jejunal branches, hepaticojejunostomy); requires complete re-survey
Secondary Risks
- Stent migration — particularly in infected field; monitor on CT at 24–48h
- Stent infection — peri-anastomotic contamination from pancreatic fistula; antibiotics and source control
- Biliary ischemia — if hepatic artery compromise affects bile duct supply
- Access site hematoma — standard complication; manual compression or closure device
- Contrast nephropathy — minimize contrast in post-operative patients with possible pre-renal impairment
Post-Procedure Care
Monitoring
- ICU: vital signs continuous monitoring, drain output hourly
- LFTs daily for 48–72h: rising AST/ALT = hepatic ischemia concern → CT hepatic perfusion assessment
- CT at 24–48h: confirm stent position, hepatic perfusion, resolution of pseudoaneurysm, exclude hepatic infarct
- Drain management: If abscess/fistula present, ensure drainage is optimized — treat underlying cause of GDA erosion
Medical Management
- Antiplatelet therapy: Aspirin 81 mg daily for covered stents — reduce thrombosis risk. Continue for 6 months.
- Antibiotics: Broad-spectrum coverage continued; tailor per cultures from drain/abscess
- Continued surgical consultation — close surgical team involvement required throughout recovery
- Recurrent bleed: urgent repeat CTA → repeat angiography (complete celiac + SMA) → surgery if repeat endovascular failure
Critical Pearls
Post-Whipple Hemorrhage Classification
ISGPS Classification of Post-Pancreatectomy Hemorrhage (PPH)
| Grade | Timing | Location | Severity | Management |
|---|---|---|---|---|
| A | Early (<24h) | Intra- or extra-luminal | Mild | Conservative (observation, transfusion if needed) |
| B | Early or Late | Intra- or extra-luminal | Moderate | Transfusion; IR embolization or re-laparotomy |
| C | Late (>24h) | Intra- or extra-luminal | Severe | IR (covered stent / embolization) or emergent surgery |
Common Vessels Involved Post-Whipple
- GDA stump — #1 most common source; pseudoaneurysm + blowout
- Right hepatic artery — branch erosion from peri-anastomotic infection
- Superior mesenteric artery branches — erosion by pancreatic fistula
- Replaced hepatic artery branches — particularly in variant anatomy patients
- Hepaticojejunostomy site — anastomotic hemorrhage (early) or pseudoaneurysm (late)
References & Resources
Key Guidelines
- SIR Standards of Practice
- CIRSE Standards of Practice for Hepatic Arterial Embolization
Primary References
- Yoon W et al. Gastrointestinal bleeding: diagnosis and treatment. Korean J Radiol. 2016;17(4):505-515.
- Tessier DJ et al. Management of vascular complications after pancreaticoduodenectomy. Arch Surg. 2006;141(8):816-823.
- Puppala S et al. Vascular complications after pancreaticoduodenectomy: radiology and treatment. Cardiovasc Intervent Radiol. 2011;34(5):1142-1153.