Indications & Contraindications
Indications
- Pancreatic cancer pain — most common indication; 70–90% response rate with neurolysis
- Chronic pancreatitis — block preferred over neurolysis (repeat injections); repeated as needed
- Other upper abdominal malignancies (gastric, hepatic, biliary)
- Block (anesthetic only): diagnostic — confirms plexus is pain generator; therapeutic short-term relief; repeatable
- Neurolysis (alcohol 50–100% or phenol 6–10%): intended permanent destruction — reserve for cancer pain with life expectancy <12 months
Contraindications
- Coagulopathy (INR >1.5, platelets <50K)
- Tumor involvement / encasement of celiac axis — distorted anatomy increases risk
- Bowel interposition in needle path
- Patient refusal
- Relative: prior aortic surgery (altered anatomy)
- Relative: chronic pancreatitis with long life expectancy — do NOT perform neurolysis
Block vs. Neurolysis Decision Guide
| Feature | Block (Anesthetic) | Neurolysis (Alcohol/Phenol) |
|---|---|---|
| Agent | Bupivacaine 0.25% | Absolute ethanol 50–100% or phenol 6–10% |
| Intent | Diagnostic / short-term relief | Permanent nerve destruction |
| Duration | 4–6 weeks | Months (durable in cancer patients) |
| Best for | Chronic pancreatitis, diagnostic | Pancreatic/upper abdominal cancer, life expectancy <12 months |
| Repeatable | Yes | Can be repeated; cryoneurolysis alternative |
Pre-Procedure Planning
Imaging & Labs
- Review recent CT abdomen/pelvis: identify celiac trunk origin, aorta, bowel proximity, degree of tumor infiltration in retroperitoneum
- Assess bilateral posterior access window for paravertebral approach — confirm no bulky nodal disease blocking needle path
- Labs: CBC, PT/INR (INR ≤1.5 required), platelets (≥50K)
- Type & screen not required for routine cases
Patient Preparation
- Well hydrated pre-procedure — 1 L NS IV before start (orthostatic hypotension prevention)
- Patient should empty bowels (or tap-water enema) morning of procedure
- NPO 6h before (moderate sedation / MAC)
- IV access ×2; blood pressure monitoring throughout
Relevant Anatomy
Celiac Plexus
- Network of ganglia located anterolateral to the aorta, between origins of the celiac trunk and superior mesenteric artery — typically at T12–L1
- Receives preganglionic sympathetic input from: greater (T5–T9), lesser (T10–T11), and least (T12) splanchnic nerves
- Relays sympathetic efferents + visceral sensory afferents from upper abdominal organs (liver, gallbladder, pancreas, spleen, adrenal glands, kidneys, stomach, proximal colon)
- Right celiac ganglion: between IVC and right diaphragmatic crus, anteromedial to medial limb of right adrenal gland
- Left celiac ganglion: medial and inferior to medial limb of left adrenal gland, lateral to left diaphragmatic crus
Approach Spaces
- Retrocrural space: posterior to diaphragmatic crura, more cephalad; needle tip just posterior to crura; spreads to surround celiac plexus via cephalad migration
- Anterocrural space: anterior to diaphragmatic crura, directly at celiac plexus; preferred for proven cancer pain; higher risk if anatomy distorted by tumor
- Posterior (dorsal) bilateral approach: standard; bilateral 22G Chiba needles, prone positioning; CT for precise needle tip placement and test injection spread visualization
- Anterior (transgastric): rarely used; traverses stomach; one-needle technique; acceptable tolerability
CT Appearance of Celiac Ganglia
The celiac ganglia appear as discoid or lobulated soft tissue structures on axial CT, often resembling the limbs of the adjacent adrenal gland or diaphragmatic crus. The right ganglion is located just posterior to the angle formed by the left renal vein entering the IVC. Bulky retroperitoneal adenopathy from malignancy can obscure the plexus and alter needle access — in such cases, cryoneurolysis may be preferred over chemical neurolysis, as ice ball distribution is more predictable than fluid spread through nodal disease.
Supplies & Setup
Needles & Access
- 22G Chiba needles ×2 (15 cm length standard)
- Extension tubing ×2
- 10 mL syringes ×4
- Dilute iodinated contrast (30 mL saline : 1 mL iodinated contrast) for test injection
- Standard procedural tray: sterile drapes, 25G/22G needles, lidocaine 1%
Neurolytic / Anesthetic Agents
- Block: Bupivacaine 0.25% — 5–10 mL per side (10–20 mL total)
- Neurolysis — ethanol: Absolute ethanol 100% — 20 mL per side (40 mL total per chapter 14 technique)
- Neurolysis — phenol: Phenol 6–10% in water or glycerol — 5–10 mL per side
- Saline flush 3 mL per side (post-ethanol injection)
Medications (Peri-procedure)
- IV fentanyl 50–100 mcg immediately before ethanol injection (burning pain prevention)
- Midazolam 1–2 mg IV for moderate sedation
- IV fluid: 1 L NS pre-procedure; continue at 100–125 mL/h during case
- Loperamide (Imodium) 2 mg PO PRN — discharge prescription
- Vasopressors at bedside (phenylephrine) for severe hypotension
Procedure Steps
CT Planning
Prone Positioning & Skin Prep
Bilateral 22G Chiba Needle Placement
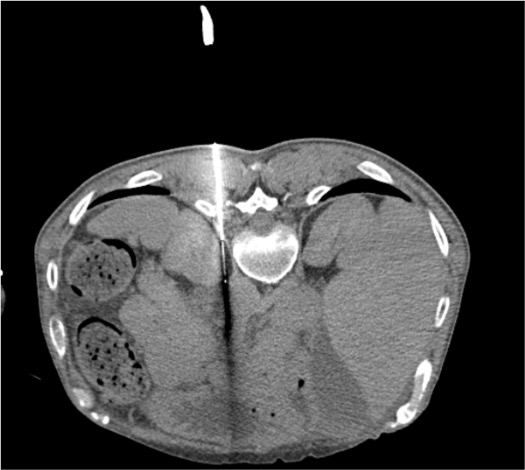
Aspiration Check
Test Injection with Dilute Contrast
Injection of Therapeutic Agent
Saline Flush (Neurolysis Only)
Post-injection CT & Needle Removal
Community Cards
Troubleshooting
Blood On Aspiration or Contrast Demonstrates Vascular Opacification
Stop injection immediately. Withdraw needle to subcutaneous fat. Do NOT inject neurolytic agent. Allow time for any intravascular contrast to clear. Reposition needle under CT guidance to periaortic soft tissue plane. Repeat aspiration check and contrast test before proceeding.
Needle Traverses Aorta
Withdraw needle slowly while monitoring for pulsatile blood. Apply firm manual pressure over puncture entry site. Monitor BP closely. Aortic needle puncture with 22G is usually self-limited and resolves without intervention. Continue to monitor closely in recovery — if BP drops significantly, obtain emergent CT to assess for retroperitoneal hematoma.
Contrast Does Not Spread Around Celiac Plexus
Inadequate spread in retrocrural position may indicate the needle tip is too posterior. Advance needle tip slightly anterior. If retrocrural spread still inadequate, consider repositioning to anterocrural space for direct plexus access. Bulky tumor/adenopathy can impede chemical spread — consider cryoneurolysis as alternative.
Patient Reports Intense Burning or Back Pain During Injection
Expected with ethanol neurolysis. If not pre-treated: pause injection, administer IV fentanyl 50 mcg, wait 2–3 min, then resume slowly. Pre-treating with opioid before injection is strongly preferred. Slow injection rate reduces intensity.
BP Drop >20 mmHg Systolic During or After Procedure
Expected autonomic effect of celiac plexus neurolysis (sympathectomy). IV fluid bolus 500 mL NS. Trendelenburg position. If persistent: phenylephrine 100 mcg IV bolus, repeat as needed. Maintain monitoring for 2h post-procedure minimum. Ensure patient does not ambulate until BP stable.
Complications
Common / Expected Side Effects
- Orthostatic hypotension (20–40%) — most common; due to splanchnic vasodilation from sympathectomy; usually self-limited 24–48h; IV fluid, fall precautions
- Diarrhea — celiac denervation releases parasympathetic tone; usually transient 2–3 days; can be prolonged (2–3 weeks as in case report); loperamide PRN
- Back pain post-injection — common, transient, from needle trauma and local inflammation; NSAIDs/Tylenol
Serious Complications
- Paralysis (<1%) — anterior spinal artery injury or intrathecal injection; most feared complication; bilateral lower extremity weakness post-procedure = emergency imaging and neurology
- Pneumothorax — upper needle approach crossing pleural space; monitor post-procedure CT
- Retroperitoneal hematoma — aortic or vessel injury; usually self-limited with 22G; monitor BP
- Infection / abscess — rare; sterile technique mandatory
- Nontarget neurolytic spread — somatic nerve injury from ethanol outside plexus
Post-Procedure Care
Recovery (0–2 Hours)
- BP monitoring every 15 min × 2h minimum
- IV fluid continued at 100 mL/h × 2h post-procedure
- Keep supine until BP stable; assist with first ambulation (fall precautions)
- Pain assessment: expect improvement beginning within hours; full effect 24–48h
- If neurolysis performed: patient may be admitted overnight for BP monitoring and pain management optimization
Discharge Instructions
- Rise slowly from lying/sitting — orthostatic hypotension risk up to 1 week
- Loperamide 2 mg PO PRN for diarrhea
- Increase oral fluid intake
- Expect back soreness at puncture sites — Tylenol PRN
- Return precautions: new weakness in legs, bowel/bladder dysfunction → ER immediately
- Pain reassessment at 2 weeks (follow-up call or clinic visit)
- Provide patient with pain diary for tracking daily pain levels before follow-up appointment.
Follow-up & Repeat Procedures
- Block (anesthetic): expect 4–6 weeks of pain relief; can be repeated; if good response, consider neurolysis for cancer patients
- Neurolysis (ethanol): durable relief for cancer patients; can be repeated if pain recurs; cryoneurolysis considered if bulky adenopathy impeded ethanol spread or prior chemical neurolysis failed
- Narcotic weaning: success allows reduction in opioid dose — coordinate with palliative care / primary team
- Pain score at 2 weeks: 70–90% of pancreatic cancer patients report significant improvement; document VAS/NRS score
References & Resources
Cryoneurolysis Alternative
- CT-guided cryoneurolysis of the celiac plexus: first reported by Mortell et al (J Vasc Interv Radiol 2014)
- Preferred when: bulky retroperitoneal adenopathy (chemical spread impeded), prior ethanol neurolysis failure, operator preference
- Technique: bilateral cryoprobes into celiac plexus; two 10-min freeze cycles separated by 5-min passive thaw (per Chapter 14 technique)
Primary References
- Prologo JD, Ray CE Jr., eds. Advanced Pain Management in Interventional Radiology: A Case-Based Approach. Thieme; 2024. DOI: 10.1055/b000000387
- Bhatnagar S, Gupta M. Evidence-based clinical practice guidelines for interventional pain management in cancer pain. Indian J Palliat Care. 2015;21(2):137–147.
- Kambadakone A, Thabet A, Gervais DA, Mueller PR, Arellano RS. CT-guided celiac plexus neurolysis: a review of anatomy, indications, technique, and tips for successful treatment. Radiographics. 2011;31(6):1599–1621.
- Wong GY, Schroeder DR, Carns PE, et al. Effect of neurolytic celiac plexus block on pain relief, quality of life, and survival in patients with unresectable pancreatic cancer: a randomized controlled trial. JAMA. 2004;291(9):1092–1099.
- Bittman RW, Peters GL, Newsome JM, et al. Percutaneous image-guided cryoneurolysis. AJR Am J Roentgenol. 2018;210(2):454–465.