Indications & Classification
The 6 P's of Acute Limb Ischemia
- Pain — acute onset, often severe; rate on numeric scale and compare to contralateral limb
- Pallor — skin pallor with demarcation line at level of occlusion; compare temperature and color
- Pulselessness — absent or diminished pulses; Doppler evaluation to supplement exam; ABI calculation
- Poikilothermia — cool extremity below occlusion with discernible temperature difference
- Paresthesia — numbness and sensory deficit; test light touch bilaterally with eyes closed
- Paralysis — motor loss; test dorsi/plantar flexion, knee flex/extension; late finding indicating severe ischemia
Time-Critical Window
- <6 hours is the optimal window for revascularization to achieve limb salvage
- Biotoxins accumulate distal to occlusion causing tissue edema and progression to compartment syndrome
- Affects 15–26 per 100,000 annually; 20× more common in lower extremities
- 1-year mortality >40%; 30-day amputation-free survival ~74.5%
- Multidisciplinary approach: IR and vascular surgery collaboration essential
Embolic Etiology (~15%)
- Abrupt onset, no prior claudication history
- Cardiac source in >90% (atrial fibrillation, acute MI, valvular disease, endocarditis)
- Lodges at arterial bifurcations (aortic, CFA, popliteal)
- Absent collateral vessels (no time for development)
- May be bilateral; may affect renal/mesenteric vasculature
Thrombotic Etiology (~85%)
- Gradual onset; history of claudication and atherosclerotic risk factors
- Occurs at pre-existing stenosis or in thrombosed bypass grafts
- Collateral vessels typically present (less severe presentation)
- Thrombosed bypass graft is the most frequent cause overall
- Popliteal artery aneurysm thrombosis or distal embolization
Pre-Procedure Planning
Imaging
- CTA abdomen/pelvis with lower extremity runoff — preferred initial study; defines etiology, extent, and outflow
- Review prior imaging: vascular US, prior CTA/MRA; look for existing grafts, stents, endografts
- Abdominal radiograph: rule out aortic endograft or kissing iliac stents that alter access strategy
- Echocardiography — evaluate for cardiac embolic source (thrombus, endocarditis, atrial fibrillation)
- MRA is alternative but rarely obtained emergently (time constraints, patient unable to remain still)
Labs & Anticoagulation
- CBC, hemoglobin ≥10 g/dL target (transfuse if below)
- BMP with K+, creatinine, CK (baseline for rhabdomyolysis monitoring)
- Lactate — marker of tissue ischemia severity
- PT/INR, fibrinogen (baseline before lysis), type & screen
- Heparin bolus per hospital protocol — start immediately upon diagnosis; maintain therapeutic anticoagulation until crossing the lesion
- Compartment pressure measurement if clinical concern (threshold >30 mmHg)
Contraindications to Thrombolysis
Absolute
- Active clinically significant bleeding
- Intracranial hemorrhage
- Established compartment syndrome
- Absolute contraindication to anticoagulation
Relative
- Stroke or neurosurgery within 6 months
- GI bleeding or hemoptysis within 3 months
- Any surgery within 14 days
- Intracranial metastases (confirm ruled out with malignancy history)
- Severe thrombocytopenia, pregnancy, bacterial endocarditis
Relevant Anatomy
Common Sites of Embolic Lodgment
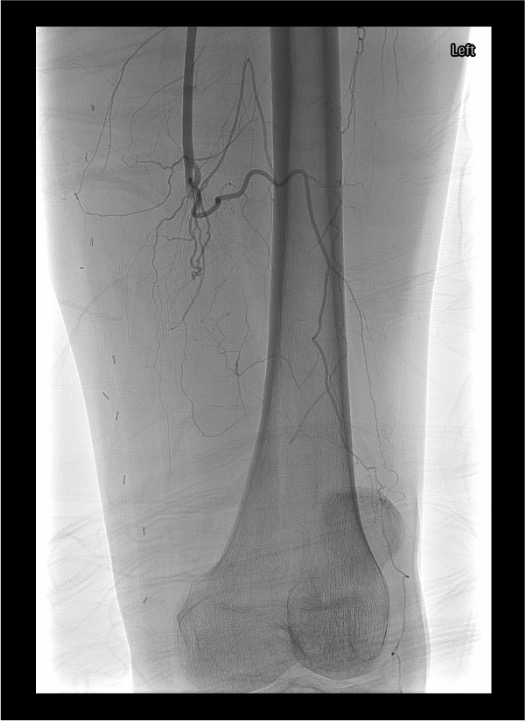
- Common femoral artery bifurcation — most common site; embolus lodges at SFA/profunda femoris bifurcation
- Aortic bifurcation — saddle embolus; bilateral symptoms; can also involve renal/mesenteric vessels
- Distal popliteal artery — trifurcation into anterior tibial, posterior tibial, and peroneal arteries
- Superior mesenteric artery — concurrent mesenteric ischemia must be excluded with abdominal emboli
- Emboli may be bilateral and involve multiple levels
In-Situ Thrombosis Sites
- Superficial femoral artery at adductor hiatus — common location of chronic atherosclerotic stenosis
- Bypass grafts (most frequent cause of ALI) — thrombosis at inflow or outflow anastomosis; identify underlying stenosis
- Popliteal artery aneurysm — degenerative in ~90%; bilateral in 60–70%; thrombosis or distal embolization far more common than rupture
- Any pre-existing stenotic segment; often superimposed acute-on-chronic disease
Collateral Pathways
- Profunda femoris artery — critical collateral for SFA occlusion; genicular branches reconstitute popliteal
- Genicular network — around the knee; connects SFA territory to tibial vessels
- Peroneal artery — serves as collateral pathway to anterior tibial and posterior tibial territories via communicating branches at the ankle
- Absence of collaterals on angiography suggests embolic (acute) rather than thrombotic (chronic) etiology
- At least one identifiable runoff vessel portends a favorable outcome with catheter-directed thrombolysis
Technique
Catheter-Directed Thrombolysis (CDT)
Pharmacomechanical Thrombectomy (PMT)
Aspiration Thrombectomy
Surgical Embolectomy
Treat Underlying Lesion
Community Cards
Landmarks & Access
Access Strategy
- Contralateral common femoral artery — typical access site; US-guided micropuncture (21G) over the femoral head, ≥1 cm above bifurcation
- Single-wall puncture technique preferred (minimizes hemorrhagic complications during subsequent lysis)
- Serial dilation to 6 Fr Ansel 2 sheath (Cook) with tip placed in ipsilateral common iliac artery
- Ipsilateral antegrade access if contralateral approach not feasible (aortic endograft, steep bifurcation, kissing iliac stents)
- Radial artery access — viable when femoral approach not feasible (bilateral groin issues, aortoiliac occlusion, kissing iliac stents); right radial preferred; use 6F sheath with 90–110 cm Flexor or Destination long sheath to reach ipsilateral iliac; suitable for both diagnostic and CDT delivery catheter placement
Up-and-Over Technique
- 5 Fr Omniflush catheter into pelvic aorta for initial angiography (10 mL/s × 25 mL if no recent pelvic imaging)
- 0.035″ Terumo angled Glidewire through Omniflush — gain significant wire access to contralateral limb before seating catheter
- Exchange for Rosen or Amplatz Super Stiff wire; advance Ansel sheath up and over bifurcation as far as anatomy allows
- Alternative catheters for steep bifurcation: Cobra, Sos, Simmons 1, Roberts Uterine Catheter
- Body floss technique for kissing iliac stents: snare wire from contralateral side with Amplatz Gooseneck or EN Snare
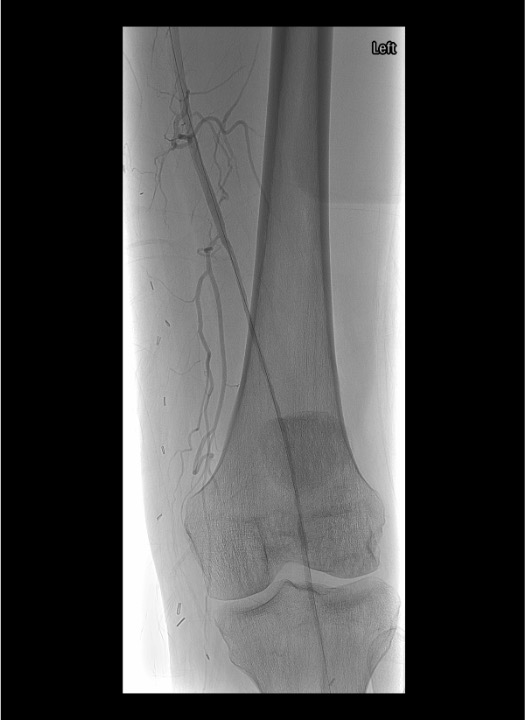
Angiographic Assessment & Endpoints
- Runoff angiography through sheath: 4 mL/s × 8 mL for outflow vessels; increase to 16–20 mL for lower leg and foot imaging
- Document baseline outflow to foot before initiating any intervention
- After crossing occlusion, gentle test injection to confirm intraluminal position and distal flow
- Select infusion catheter (Unifuse) to span entire lesion length; available in 10 cm increments (10–50 cm)
- Angiographic endpoints: restoration of inline flow, resolution of filling defects, treatment of underlying lesion
- If >50 cm occlusion, position 50 cm catheter with proximal marker at proximal extent; lytic medication flows distally as flow re-establishes
Troubleshooting
Persistent Thrombus After 12–24 Hours of CDT
Reassess clinical status and Rutherford classification. If no significant improvement at 12–24 hours, consider surgical revascularization. Dripping tPA just proximal to the occlusion for several hours may soften the fibrous platelet plug and allow subsequent wire crossing. If motor function deteriorating, call surgeon immediately for emergent surgical embolectomy.
Tense Compartments, Pain Out of Proportion, Pain on Passive Stretch
Emergent fasciotomy. Compartment pressure >30 mmHg is threshold for fasciotomy. Can be worsened by abrupt reperfusion of acutely ischemic limb. Involve surgery immediately. Do NOT continue lysis if compartment syndrome develops — this is an absolute contraindication to further thrombolysis. Monitor CK and K+ closely.
New Filling Defects Distal to Original Occlusion
Common during lysis as thrombus fragments. Continued tPA administration will usually improve or resolve distal emboli. For calcified plaque emboli that will not lyse, use catheter aspiration or Indigo system suction embolectomy. Avoid forcing wire or catheter if not progressing easily — risk of vessel dissection.
Difficulty Obtaining Contralateral Access or Crossing Occlusion
For scarred groins from prior surgery: use stiff 21G micropuncture set or 18G needle with stiff wire. For steep bifurcation or aortic endograft: ipsilateral antegrade puncture below inguinal ligament. Change imaging obliquity and correlate with CTA landmarks. Try different catheter shapes (Cobra, Sos, reverse curve) to alter angle of approach. Popliteal puncture or direct puncture of thrombosed vessel may rarely be necessary.
Headache, Altered Mental Status, or New Focal Deficit
Stop tPA immediately. Obtain emergent non-contrast head CT. Intracranial hemorrhage is the most feared complication (0–2.5%). Reverse anticoagulation, administer cryoprecipitate if fibrinogen depleted. Neurologic checks q2h by ICU nursing throughout lysis; any deterioration triggers immediate cessation and imaging.
Complications
Procedural / Hemorrhagic
- Intracranial hemorrhage (0–2.5%) — most serious complication of lysis; may be fatal; suggested threshold <2%
- Major bleed requiring transfusion or surgery (1–20%) — suggested threshold <10%; target Hgb ≥10 pre-procedure
- Distal embolization (1–5%) — not corrected by thrombolysis in some cases; mechanical embolization 1.8%
- Access site hematoma — minimize with single-wall puncture; observe closely during lysis
- Vessel dissection or perforation — gentle wire technique; lytic medication running post-perforation is devastating
Reperfusion & Systemic
- Reperfusion injury — tissue edema, acidosis, electrolyte derangement upon restoration of flow
- Compartment syndrome (1–10%) — worsened by abrupt reperfusion; suggested threshold <4%; requires fasciotomy
- Hyperkalemia — potassium release from ischemic/necrotic muscle; can cause cardiac arrhythmia; monitor q6h
- Myoglobinuria / acute renal failure — rhabdomyolysis from muscle necrosis; aggressive IV hydration; monitor CK q6h
- Amputation — may be necessary for irreversible ischemia (Rutherford III); attempting revascularization in Rutherford III may worsen clinical status
Pearls & Pitfalls
References & Resources
Landmark Trials
- STILE Trial — Surgery vs Thrombolysis for Ischemia of the Lower Extremity; prospective randomized; demonstrated equivalent outcomes for limb salvage between CDT and surgery in acute (<14 day) occlusions
- TOPAS Trial — Thrombolysis or Peripheral Arterial Surgery; recombinant urokinase vs surgery; similar amputation-free survival at 1 year; lysis associated with reduced surgical intervention complexity
Primary References
- Ouriel K, Veith FJ, Sasahara AA. A comparison of recombinant urokinase with vascular surgery as initial treatment for acute arterial occlusion of the legs (STILE trial). N Engl J Med. 1998;338(16):1105–1111.
- Comerota AJ, Weaver FA, Hosking JD, et al. Results of a prospective, randomized trial of surgery versus thrombolysis for occluded lower extremity bypass grafts (TOPAS). J Vasc Surg. 1996;23(1):64–73.
- Rutherford RB, Baker JD, Ernst C, et al. Recommended standards for reports dealing with lower extremity ischemia: revised version. J Vasc Surg. 1997;26(3):517–538.
- Gilliland CA, Shah J, Martin JG, Miller MJ Jr. Acute limb ischemia. Tech Vasc Interventional Rad. 2017;20:274–280.
- Walker TG. Acute limb ischemia. Tech Vasc Interventional Rad. 2009;12:117–129.
- Patel NH, Krishnamurthy VN, Kim S, et al. Quality improvement guidelines for percutaneous management of acute lower-extremity ischemia. J Vasc Interv Radiol. 2013;24:3–15.
- Creager MA, Kaufman JA, Conte MS. Clinical practice: acute limb ischemia. N Engl J Med. 2012;366:2198–2206.
- Berridge DC, Kessel DO, Robertson I. Surgery versus thrombolysis for initial management of acute limb ischaemia. Cochrane Database Syst Rev. 2013;6:CD002784.