Indications
| Criterion | Threshold | Notes |
|---|---|---|
| IPSS symptom score | ≥13 (moderate–severe) | Score 0–35; PAE typically for scores ≥13 refractory to medical therapy |
| Uroflowmetry (Qmax) | <12 mL/s | Peak flow rate reduced by bladder outlet obstruction |
| Prostate volume | >40 mL | Larger prostates (>80 mL) respond best; very small glands (<40 mL) have lower success rates |
| Medical therapy failure | Alpha-blockers ± 5-ARI | Must have failed adequate medical therapy trial |
| Urinary retention | Foley-dependent | BPH-related retention; high success rate for catheter liberation |
| Surgical poor candidate | High anesthetic risk | PAE performed under local + moderate sedation; no general anesthesia required |
Contraindications
| Type | Contraindication |
|---|---|
| Absolute | Prostate cancer (must be excluded with MRI + PSA; biopsy if elevated) · Active UTI (treat first) · Uncorrectable coagulopathy · Known contrast allergy |
| Relative | Severe atherosclerotic iliac disease limiting catheter access · Neurogenic bladder · Large bladder diverticula · Prostate <40 mL (lower response rate) · Median lobe predominant BPH (TURP preferred) |
PAE vs. TURP Comparison
| Feature | PAE | TURP / Surgery |
|---|---|---|
| Anesthesia | Local + moderate sedation | General / spinal anesthesia |
| Hospital stay | Outpatient / same-day | 1–3 days inpatient |
| Retrograde ejaculation | <5% | 65–75% (TURP) |
| IPSS improvement at 6 months | 40–60% | 70–80% |
| Prostate volume reduction | ~25–30% | Tissue resected directly |
| Best for | Large prostates (>80 mL), poor surgical candidates, patient preference for preserving ejaculation | Moderate-sized glands, median lobe predominant, failed PAE |
| SIR position | Endorsed by multi-society consensus (SIR, AUA, EAU) | Standard of care for BPH |
Multi-society consensus (SIR 2019): PAE is an appropriate, evidence-based treatment option for LUTS/BPH in properly selected patients. The WATER trial (BMJ 2018) and UK-ROPE study both demonstrated non-inferiority of PAE to TURP for symptom relief with significantly better sexual function preservation.
Relevant Anatomy
Prostatic Artery Origin
The prostatic artery most commonly arises from the anterior division of the internal iliac artery (IIA), typically in close association with the inferior vesical artery. Variants are common — the PROVISO mnemonic covers five key pelvic branches plus the optimal angiographic projection to identify them. VI counts as a single paired entry (Inferior Vesical); the final O is the technique reminder:
| PROVISO Branch | Full Name | Relevance |
|---|---|---|
| P | Internal Pudendal Artery | Most common single origin for prostatic artery (~31%); anastomoses to penile territory — non-target supply requiring CBCT exclusion |
| R | Middle Rectal Artery | Non-target — rectal ischemia if embolized; must be excluded on CBCT before embolizing |
| O | Obturator Artery | Prostatic artery arises from obturator in ~19% of cases; identify origin to avoid missing target supply |
| VI | Inferior Vesical Artery and its prostatic branches | Shares common trunk with prostatic artery in ~29%; bladder branch = non-target; prostatic branches = target |
| S | Superior Vesical Artery | Common trunk origin — must distinguish superior vesical (bladder) from prostatic branches on CBCT |
| O | Oblique perspective | 20–50° ipsilateral oblique + 10–20° caudal angulation — the optimal projection to separate all pelvic branches and identify the prostatic artery origin |
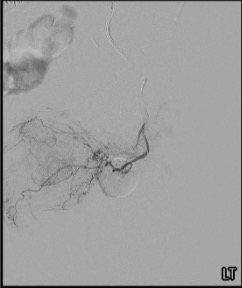
Intraprostatic Supply
Within the prostate, the prostatic artery divides into anteromedial branches (supplying the transition zone — the BPH target) and posterolateral branches (peripheral zone). Dangerous anastomoses to the bladder (superior vesical), rectum (middle rectal), and penis (internal pudendal) require identification on CBCT before embolization. The best angiographic projection is the 20–50° ipsilateral oblique with 10–20° caudal angulation.
Pre-Procedure Checklist
- MRI prostate (PI-RADS): Exclude cancer (PI-RADS ≤2 required); measure prostate volume; assess zonal anatomy and transition zone; confirm >40 mL
- CTA pelvis: Map prostatic artery origins bilaterally; assess iliac tortuosity and atherosclerosis; plan catheterization strategy pre-procedure
- IPSS questionnaire: Document baseline score ≥13
- Qmax uroflowmetry: Document baseline Qmax <12 mL/s
- PSA: If elevated → biopsy required and cancer must be excluded before scheduling PAE
- UA/UCx: Confirm no active UTI; reschedule and treat if positive
- Labs: CBC, BMP, PT/INR
- Antibiotics: Ciprofloxacin 400 mg IV or levofloxacin 500 mg PO pre-procedure; oral course for 5–7 days post
- Alpha-blocker: Continue or start tamsulosin; reduce post-procedure urinary retention; maintain 2–4 weeks post-PAE
- Foley catheter: If patient in retention — fill balloon with dilute contrast (10–20% iodinated) for intraprocedural bladder neck identification on CBCT
Procedure Overview
The following is a high-level summary. Full step-by-step technique, catheter and microcatheter selection, CBCT acquisition protocols, and troubleshooting are available in RadCall Pro.
Access and Catheterization
Unilateral common femoral artery access. Cross the aortic bifurcation to catheterize the contralateral internal iliac artery. Perform selective DSA of the IIA in the 20–50° ipsilateral oblique view with 10–20° caudal angulation to map PROVISO branches.
CBCT and Microcatheter
Perform cone-beam CT from the IIA to identify the prostatic artery origin and all pelvic branches. Advance a microcatheter into the prostatic artery and obtain a prostatic arteriogram — confirm characteristic hemi-prostatic parenchymal blush.
CBCT Confirmation (Mandatory)
Perform CBCT with the microcatheter in position before embolizing. Confirm:
- Prostatic parenchymal enhancement (hemi-prostate blush)
- No enhancement extending to bladder wall (cephalad)
- No enhancement posterior to prostate toward rectum
- No penile/pudendal territory enhancement
If dangerous anastomoses are identified: advance the microcatheter distal to the anastomosis origin, or place protective coils/gelatin sponge before particle embolization.
Embolization
Inject microspheres (100–300 µm or 300–500 µm) in high dilution at a slow, controlled rate under fluoroscopy. Rapid injection causes proximal stasis and incomplete embolization. Endpoint: total stasis with venous phase visualization.
PErFecTED technique: When premature proximal stasis occurs, advance the microcatheter distally into intraprostatic vessels and perform wedged injection. This achieves more complete gland ischemia and is associated with lower 1-year recurrence rates (5% vs. 22% for standard technique).
Bilateral Embolization
Repeat the entire process on the contralateral side and confirm no remaining prostatic blood supply bilaterally. Bilateral embolization is essential — unilateral PAE is associated with significantly higher clinical failure rates and should only be accepted when the contralateral side is truly inaccessible.
Complications
| Complication | Rate | Management |
|---|---|---|
| Post-embolization syndrome | 20–30% | Dysuria, pelvic discomfort, low-grade fever — self-limited 3–7 days; NSAIDs, alpha-blockers, anti-emetics |
| Transient urinary retention | Common | Prostatic edema; Foley catheter if needed; alpha-blocker continuation |
| Transient hematuria | Common | Usually self-limited 24–48h |
| UTI | <5% | Prophylactic antibiotics reduce incidence; treat promptly |
| Rectal ischemia | Rare with CBCT | Non-target embolization to middle rectal branches — CBCT mandatory to exclude before embolizing; proctoscopy if suspected post-procedure |
| Bladder ischemia | Rare with CBCT | Non-target embolization to superior vesical branches; severe suprapubic pain + hematuria; CBCT pre-embolization identifies at-risk branches |
| Access site | Standard | Hematoma, pseudoaneurysm — standard femoral management |
Post-Procedure Care and Follow-up
- Discharge: Same-day outpatient; continue alpha-blocker (tamsulosin) for 2–4 weeks; complete oral antibiotic course
- Post-embolization syndrome: NSAIDs scheduled for 5–7 days; patient education that dysuria and low-grade fever are expected
- Follow-up schedule: IPSS + Qmax at 1, 3, 6, and 12 months post-procedure
- MRI prostate at 6 months: Assess prostate volume reduction (target ~25–30% reduction) and treatment zone
- Expected timeline: Maximum IPSS improvement typically takes 3–6 months to fully manifest; prostate volume reduction continues over this period
- Recurrence: If symptoms return after initial success → repeat CTA/MRI; repeat PAE for recanalization or new collateral supply
Evidence Summary: PAE vs. MISTs
No head-to-head RCTs compare PAE directly to Rezum or UroLift. Evidence comes from network meta-analyses (NMAs) and indirect comparisons. Key findings from the largest available NMAs and RCTs:
PAE vs. TURP (5 RCTs, n=352 — Cochrane 2022)
- IPSS improvement: Similar short-term (MD 1.72 points favoring TURP; low-certainty evidence); at 2 years, TURP significantly superior (9.21 vs. 12.09 points; p=0.047)
- Qmax improvement: Significantly favors TURP (e.g., 5.19 vs. 15.34 mL/s at 12 weeks)
- Retreatment rate: Higher with PAE (RR 3.20; 95% CI: 1.41–7.27); ~21% of PAE patients required TURP within 2 years in one trial
- Adverse events: Fewer total adverse events with PAE (36 vs. 70 in one trial; p=0.003)
- Ejaculatory dysfunction: Less with PAE (RR 0.67; 95% CI: 0.45–0.98)
- Urinary incontinence: Lower with PAE (RR 0.13; 95% CI: 0.02–0.70)
Sham-controlled trial (Pisco et al., 2020; n=80): IPSS improvement favored PAE by −13.2 points (95% CI: −16.2 to −10.2; p<0.001) vs. sham at 6 months. Prostate volume reduced by −17.6 g with PAE vs. −0.1 g with sham — establishing that PAE effect is not placebo.
PAE vs. Rezum vs. UroLift — NMA Comparison
| Feature | PAE | Rezum (WVTT) | UroLift (PUL) |
|---|---|---|---|
| Prostate volume range | No upper limit | 30–80 cc (FDA-approved) | 30–80 cc (FDA-approved) |
| IPSS improvement (12 mo) | −9 to −12 pts | ~−11 pts | −7 to −11 pts |
| Qmax improvement | Moderate | Moderate | Modest |
| Erectile function | Improved (IIEF-5) — only MIST with significant improvement; continues 6→12 months | Preserved | Preserved (best IIEF ranking in one NMA) |
| Ejaculatory function | Better than TURP | Best preserved | Best preserved |
| Durability (12 mo) | IPSS continues to improve 6→12 months | Stable | Worsens 6→12 months |
| Retreatment rate | ~10–21% | ~4–7% | ~6–14% |
| Setting | IR suite | Office / OR | Office / OR |
Bottom line: PAE, Rezum, and UroLift offer similar short-term symptom improvement. PAE is uniquely advantaged for very large prostates (>80 mL), patients on anticoagulation, and those prioritizing erectile function improvement. Rezum and UroLift are preferred when ejaculatory preservation is paramount. All three carry higher retreatment rates than TURP. PAE is technically demanding and should only be performed by operators with specific training.
Technical Success Factors (n=551, single-center series)
- Bilateral embolization required for optimal outcomes — unilateral is associated with higher failure
- Particle size: 100–300 µm microspheres used in 80% of cases
- Learning curve: Fluoroscopy time decreased 25.2% and procedural time 26.1% over the study period
- Embolic volume: Outcomes positively correlated with absolute amount of embolic material injected (P-Easy PLUS, 2025)
- Technical success rate: 86–100% across studies; experienced centers achieve near-100%
Guideline Recommendations
| Indication | Guideline | Strength |
|---|---|---|
| Moderate–severe LUTS/BPH refractory to medical therapy | AUA 2023 / SIR Multisociety | AUA: Conditional (Grade C) · SIR: Level B, strong |
| Very large prostate (>80 cm³) — no upper size limit | SIR Multisociety | Level C, moderate |
| Urinary retention — catheter liberation goal | SIR Multisociety | Level C, moderate |
| Preservation of erectile/ejaculatory function | SIR Multisociety | Level C, weak |
| Prostatic hematuria (historical first use) | SIR Multisociety | Level D, strong |
| Poor surgical candidates (advanced age, comorbidities, anticoagulation) | SIR Multisociety | Level E, moderate |
PAE was endorsed by multi-society consensus (SIR, AUA, EAU, CIRSE) in 2019 and affirmed in the AUA BPH Guideline Amendment 2023. The 2024 P-Easy ADVANCE trial demonstrated superiority of PAE over combined medical therapy (tamsulosin + dutasteride) in treatment-naïve patients — raising the potential for PAE as an earlier intervention, though further validation is needed.
References
- Sandhu JS, Bixler BR, Dahm P, et al. Management of Lower Urinary Tract Symptoms Attributed to Benign Prostatic Hyperplasia (BPH): AUA Guideline Amendment 2023. J Urol. 2024;211(1):11–19.
- McWilliams JP, Bilhim TA, Carnevale FC, et al. Society of Interventional Radiology Multisociety Consensus Position Statement on Prostatic Artery Embolization for Treatment of LUTS Attributed to BPH. J Vasc Interv Radiol. 2019;30:627–637.
- Jung JH, McCutcheon KA, Borofsky M, et al. Prostatic Arterial Embolization for the Treatment of Lower Urinary Tract Symptoms in Men With Benign Prostatic Hyperplasia. Cochrane Database Syst Rev. 2022;3:CD012867.
- Abt D, Hechelhammer L, Müllhaupt G, et al. Comparison of prostatic artery embolisation (PAE) versus transurethral resection of the prostate (TURP): randomised, open label, non-inferiority trial (WATER). BMJ. 2018;361:k2338.
- Abt D, Müllhaupt G, Hechelhammer L, et al. Prostatic Artery Embolisation Versus Transurethral Resection of the Prostate: 2-Year Outcomes of a Randomised Trial. Eur Urol. 2021;80(1):34–42.
- Pisco JM, Bilhim T, Costa NV, et al. Randomised Clinical Trial of Prostatic Artery Embolisation Versus a Sham Procedure for Benign Prostatic Hyperplasia. Eur Urol. 2020;77(3):354–362.
- Sajan A, Mehta T, Desai P, Isaacson A, Bagla S. Minimally Invasive Treatments for Benign Prostatic Hyperplasia: Systematic Review and Network Meta-Analysis. J Vasc Interv Radiol. 2022;33(4):359–367.
- LaRussa S, Pantuck M, Wilcox Vanden Berg R, et al. Symptomatic Improvement of LUTS/BPH: A Comparative Systematic Review and Meta-Analysis of 4 Minimally Invasive Therapies. J Vasc Interv Radiol. 2021;32(9):1328–1340.
- Lucas-Cava V, Sánchez-Margallo FM, Insausti-Gorbea I, Sun F. Comparative Efficacy and Safety of Prostatic Urethral Lift vs Prostatic Artery Embolization: A Systematic Review and Network Meta-Analysis. BJU Int. 2023;131(2):139–152.
- Alizadeh LS, Radek D, Booz C, et al. Prostatic Artery Embolization: Lessons From 551 Procedures at a Single Center. Acad Radiol. 2024;31(11):4519–4527.
- Brown N, Firouzmand S, Kiosoglous A, et al. P-Easy PLUS. BJU Int. 2025. doi:10.1111/bju.16808.
- Brown N, Kiosoglous A, Castree S, et al. P-Easy ADVANCE RCT: PAE vs. Medical Therapy in Treatment-Naïve Men. BJU Int. 2024;134 Suppl 2:38–46.
- Ray AF, Powell J, Wallis C, et al. Efficacy and safety of prostate artery embolization for BPH: UK-ROPE study. Cardiovasc Intervent Radiol. 2018;41(8):1138–1148.
- de Assis AM, Moreira AM, de Paula Rodrigues VC, et al. Pelvic arterial anatomy relevant to prostatic artery embolisation and proposal for angiographic classification. Cardiovasc Intervent Radiol. 2015;38:855–861.
- Carnevale FC, Moreira AM, Harward SH, et al. Recurrence of LUTS following PAE: comparing PErFecTED vs. standard technique. Cardiovasc Intervent Radiol. 2017;40:366–374.