Indications
| Indication | Details |
|---|---|
| Acute acalculous cholecystitis (AAC) | Critically ill ICU patients (trauma, post-op, TPN-dependent, burns) — #1 indication; mortality >50% without treatment |
| Acute calculous cholecystitis | High-risk surgical candidates (ASA 3–4); bridge to cholecystectomy — stabilize first, then plan elective surgery |
| Bridge to cholecystectomy | Stabilize acutely ill patient; elective cholecystectomy 6–8 weeks after resolution |
| Gallbladder perforation | With bile leak or pericholecystic collection |
| Malignant GB obstruction | Non-surgical palliation of biliary obstruction |
| Transcholecystic CBD access | Portal for biliary interventions when ERCP and PTBD not feasible |
Transhepatic vs. Transperitoneal Approach
| Feature | Transhepatic (Preferred) | Transperitoneal |
|---|---|---|
| Hepatic parenchymal traversal | Yes — acts as catheter seal | No |
| Bile peritonitis risk if catheter dislodges | Low — liver seals the tract | Higher — no protective seal |
| Hemorrhagic risk | Slightly higher (parenchymal crossing) | Lower |
| Use when | Standard approach; bowel interposition; large ascites; most routine cases | Coagulopathy; cirrhosis/portal hypertension; hepatomegaly; pregnancy |
| Requirement | Confirm no severe coagulopathy before crossing liver parenchyma | Confirm no bowel interposition on real-time US before proceeding |
Pre-Procedure Checklist
- US gallbladder: Wall thickening (>3.5 mm), pericholecystic fluid, sonographic Murphy's sign, stones or sludge. Confirm GB is distended — non-distended GB is less likely to be the source and a harder target.
- CT for complex anatomy: Bowel interposition on planned access route; ascites; hepatomegaly; pericholecystic abscess
- HIDA if diagnosis uncertain: No GB visualization at 60 min after morphine augmentation = acalculous cholecystitis (99% sensitivity)
- Labs (SIR Category 2–3): CBC, LFTs, BMP, coagulation panel. INR <1.5, platelets >50K for transhepatic. Bilirubin elevation suggests CBD involvement — may need PTBD.
- Antibiotics: Piperacillin-tazobactam 3.375g IV pre-procedure; continue 24–48h post. Covers gram-negatives and anaerobes expected in biliary infections.
- Consent: Bile peritonitis (~1%), hemorrhage (<1%), vasovagal during procedure, catheter dislodgement, need for surgery (~10%). Emphasize drain must NOT be removed early.
Technique
The following is a high-level summary. Full access technique, trocar vs. Seldinger decision-making, catheter sizing, drain management algorithms, and removal criteria are available in RadCall Pro.
Access: Trocar vs. Seldinger
Trocar method (preferred in critically ill): Combined catheter/cannula/stylet advanced directly into the GB lumen in one motion under real-time US. Faster — GB is entered and secured before it can decompress. Use for: distended GB, critically ill, urgent cases.
Seldinger method: Needle → aspirate bile confirmation → wire → dilate → catheter. Allows small-needle confirmation before large catheter placement. Use for: deep GB or uncertain access.
Procedure Steps
- US survey and planning: Identify GB fundus and body; plan transhepatic vs. transperitoneal; confirm no bowel interposition; measure depth from skin to GB lumen
- Sterile prep + local anesthesia: Infiltrate from skin to GB capsule — do not puncture the GB lumen with the anesthesia needle
- Real-time US-guided access: Trocar method preferred — advance directly into GB lumen. Confirm bile return to verify intraluminal position
- Bile aspiration and culture: Aspirate for gram stain + aerobic and anaerobic culture. Note: purulent = infected; bloody = hemorrhagic; mucoid = mucocele
- Catheter placement: Advance locking pigtail catheter; release locking mechanism to form pigtail within GB. Confirm position on US
- Secure and connect: Suture catheter to skin; connect to external drainage bag; record initial output volume, color, and character
Complications
| Complication | Rate | Management |
|---|---|---|
| Bile peritonitis | 1–4% | Most feared — catheter dislodgement or inadequate hepatic seal; transhepatic approach strongly mitigates this risk |
| Hemorrhage | Usually minor | Minor tract hemostasis; significant if hepatic vessel injured (transhepatic); transfusion if Hgb drops significantly |
| Vasovagal reaction | Uncommon | Monitor during and after; have atropine available |
| Sepsis | Procedure-related | Infected bile manipulation — pre-procedure antibiotics essential |
| Catheter dislodgement | Most common delayed | Patient education critical; never remove early; reinsertion may be possible via existing tract if caught within hours |
| Failed decompression requiring surgery | ~10% | Gangrenous cholecystitis, emphysematous cholecystitis, perforation — surgical consult when no clinical improvement at 48–72h |
Drain Management and Removal Criteria
| Parameter | Target / Normal | Notes |
|---|---|---|
| Clinical response | Fever resolves, WBC trends down within 24–72h | No improvement at 48–72h → CT reassess; surgical consult if gangrenous cholecystitis |
| Output monitoring | 50–500 mL/day | High output in first 24–48h is expected as tensely distended GB decompresses |
| Daily flushing | 5–10 mL sterile saline q12h | Nursing order required; prevents sludge accumulation and catheter occlusion |
| Imaging confirmation before removal | Cholecystocholangiogram at 4 weeks | Inject dilute contrast through catheter under fluoroscopy; confirm cystic duct patent and CBD anatomy |
| Removal criteria (all must be met) | Mature tract ≥4 weeks + clinical resolution + patent cystic duct on imaging | Never remove early — if all three not met, continue drainage and reassess |
| Long-term exchange | Every 3 months if permanent drainage needed | Non-surgical candidates; scheduled IR follow-up with cholangiogram at exchange |
| Cholecystectomy timing | 6–8 weeks after acute resolution | For surgical candidates; HIDA prior to surgery if laparoscopic approach planned |
Post-Procedure Care
- Clinical response expected within 24–72 hours: Fever resolves, WBC trends down, RUQ pain improves — if not, CT to reassess for gangrenous cholecystitis, GB perforation, or drain malposition
- Surgical consult if no improvement — approximately 10% require urgent cholecystectomy
- For non-surgical candidates: long-term catheter management with scheduled 3-month exchanges; annual reassessment of surgical candidacy
- Patient education is critical: Catheter must not be removed at home or by non-IR providers until removal criteria are confirmed. Discharge instructions must clearly state this.
Evidence Summary: PC vs. Cholecystectomy
Percutaneous cholecystostomy occupies a specific and narrow niche in the management of acute cholecystitis. The evidence consistently supports laparoscopic cholecystectomy as superior when surgically feasible — even in high-risk patients.
CHOCOLATE Trial (2018) — Landmark RCT
The only RCT comparing laparoscopic cholecystectomy to PC in high-risk patients (APACHE II ≥7, n=142) was stopped early due to clear superiority of surgery:
| Outcome | Laparoscopic Cholecystectomy | Percutaneous Drainage | P-value |
|---|---|---|---|
| Mortality at 1 year | 3% | 9% | 0.27 |
| Major complications | 12% | 65% | 0.001 |
| Reintervention rate | 12% | 66% | 0.001 |
| Recurrent biliary disease | 5% | 53% | 0.001 |
| Median hospital stay | 5 days | 9 days | 0.001 |
Bottom line: Even in APACHE II ≥7 patients, laparoscopic cholecystectomy had dramatically lower major complications (12% vs. 65%) and reintervention rates (12% vs. 66%). PC should not be chosen over surgery simply because a patient appears "high risk" — surgical risk must be truly prohibitive.
Meta-Analysis (2023) — PC as Definitive Treatment
Systematic review of 17 studies (783,672 patients) comparing PC as definitive treatment vs. emergency cholecystectomy:[7]
- Unadjusted mortality: PC 10.1% vs. cholecystectomy 0.8% vs. no intervention 5.2%
- After adjusting for baseline risk, both PC (OR 0.78) and cholecystectomy (OR 0.42) reduced mortality vs. no intervention — but cholecystectomy was superior to PC
- PC patients were significantly older (mean 70.6 vs. 53.8 years) with higher comorbidity burden, reflecting appropriate patient selection
Guideline Recommendations
| Guideline | Recommendation |
|---|---|
| Tokyo Guidelines 2018 | Poor surgical candidates (CCI ≥4, ASA-PS ≥3, neurologic or respiratory dysfunction) → PC or antibiotics before cholecystectomy |
| 2025 International Delphi Consensus | Moderate AC + CCI ≥6 + ASA ≥3 failing conservative therapy → laparoscopic cholecystectomy first-line. Severe AC at high surgical risk → PC within 24–48h. Interval cholecystectomy ≥6 weeks after PC if patient becomes surgical candidate. |
| WSES/SICG 2017 (Elderly) | AC in patients >65 with ASA III/IV, PS 3–4, or septic shock deemed unfit for surgery → PC (LoE 2, GoR B) |
| 2025 Multisociety Expert Panel | Never-surgical candidates → EUS-GBD with LAMS preferred over long-term PC if institutional expertise and patient can tolerate monitored anesthesia |
Technical Success and Adverse Events
- Clinical success rate: ~90% resolution of acute cholecystitis[9]
- Adverse events: Up to 25% — catheter dislodgement (peritonitis risk if within first weeks), tube discomfort, quality of life impact
- Timing: PC within 24h of diagnosis associated with improved outcomes; PC performed >7 days after diagnosis associated with increased mortality[3]
Management Algorithm
The following algorithm (Sadaka et al., JAMA Surgery 2025) synthesizes current evidence-based management of acute cholecystitis — stratifying by operative candidacy, sepsis status, and disease severity:
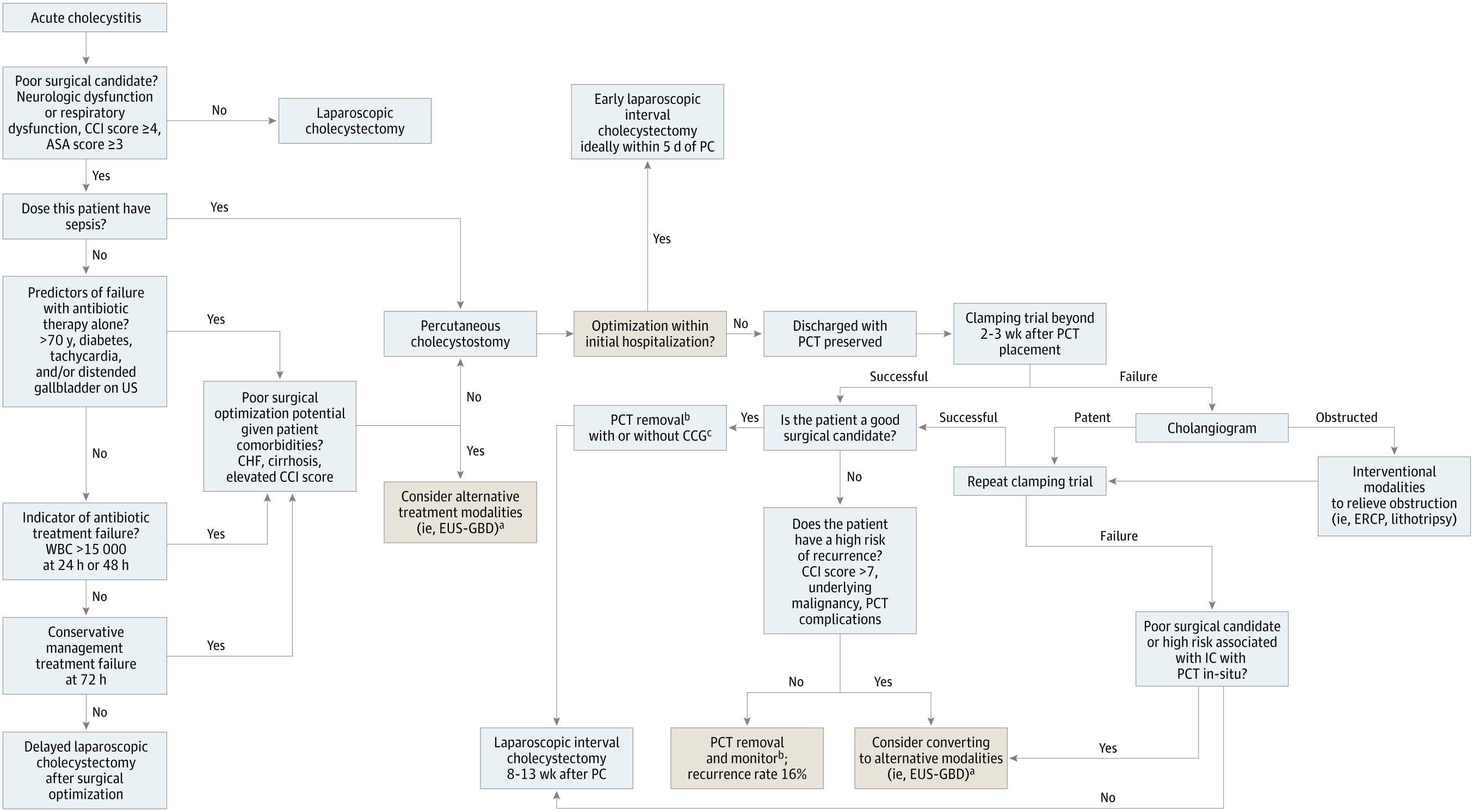
Key Clinical Principles
- PC is primarily a bridge to interval cholecystectomy — not definitive treatment in surgically eligible patients
- Reserve PC for: poor surgical candidates with sepsis, antibiotic treatment failure, or reversible surgical contraindications (recent MI, severe malnutrition)
- Tube management: Clamping trial superior to cholangiography alone for determining readiness for tube removal
- Risk stratification: CHF and chronic liver disease predict patients unlikely to become surgical candidates — consider EUS-GBD early in these patients
- Elderly (>65 years): Laparoscopic cholecystectomy associated with lower 2-year mortality (15.2%) vs. nonoperative management (29.3%)
- Acalculous cholecystitis: PC reserved for severely ill patients at diagnosis; all others → laparoscopic cholecystectomy
References
- Gallaher JR, Charles A. Acute Cholecystitis: A Review. JAMA. 2022;327(10):965–975.
- Loozen CS, van Santvoort HC, van Duijvendijk P, et al. Laparoscopic Cholecystectomy Versus Percutaneous Catheter Drainage for Acute Cholecystitis in High Risk Patients (CHOCOLATE): Multicentre Randomised Clinical Trial. BMJ. 2018;363:k3965.
- Sadaka AH, Tseng JF, Itani KMF. Indications for and Optimal Management of Percutaneous Cholecystectomy Drainage. JAMA Surgery. 2025;160(12):1368–1377.
- Pesce A, Ramírez-Giraldo C, Arkoudis NA, et al. Management of High-Surgical-Risk Patients With Acute Cholecystitis Following Percutaneous Cholecystostomy: Results of an International Delphi Consensus Study. Int J Surg. 2025;111(5):3185–3192.
- Pisano M, Ceresoli M, Cimbanassi S, et al. 2017 WSES and SICG Guidelines on Acute Calcolous Cholecystitis in Elderly Population. World J Emerg Surg. 2019;14:10.
- Baron TH, Jorge I, Husnain A, et al. Comprehensive Review of the Management of Patients With Acute Cholecystitis Who Are Ineligible for Surgery. Ann Surg. 2025.
- Cirocchi R, Amato L, Ungania S, et al. Management of Acute Cholecystitis in High-Risk Patients: PC as Definitive Treatment vs. Emergency Cholecystectomy — Systematic Review and Meta-Analysis. J Clin Med. 2023;12(15):4903.
- Wadhwa V, Trivedi PS, Makary MS, et al. Utilization and Outcomes of Cholecystostomy and Cholecystectomy in Patients Admitted With Acute Cholecystitis: A Nationwide Analysis. AJR. 2021;216(6):1558–1565.
- Baron TH, Grimm IS, Swanstrom LL. Interventional Approaches to Gallbladder Disease. N Engl J Med. 2015;373(4):357–365.
- Latif J, Kushairi A, Thurley P, Bhatti I, Awan A. Laparoscopic Cholecystectomy Versus Percutaneous Cholecystostomy: Suitability of APACHE-II Score, ASA Grade, and Tokyo Guidelines 18 Grade as Predictors of Outcome in Acute Cholecystitis. Surg Laparosc Endosc Percutan Tech. 2022;32(3):342–349.
- Yokoe M, Hata J, Takada T, et al. Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholecystitis. J Hepatobiliary Pancreat Sci. 2018;25(1):41–54.
- ACR-SIR Practice Parameter for Cholecystostomy and Drainage.