BI-RADS Assessment Categories
ACR BI-RADS 2025 — complete category definitions with management
| Feature | BI-RADS 5th Ed (2013) | BI-RADS 2025 | Clinical Impact |
|---|---|---|---|
| General | |||
| Category 0 | Single category: "Incomplete — Need Additional Imaging Evaluation and/or Prior Imaging for Comparison" | Split into two: 0A — Need Additional Imaging Evaluation 0B (NEW) — Need Prior Mammograms for Comparison Separated to reflect 2024 FDA MQSA amendments. 0B: must reach final assessment within 30 days. |
0B only when priors are genuinely required to render Cat 1 or 2 and can feasibly be obtained. Do NOT use 0A to defer biopsy on a suspicious finding — assign Cat 4/5. Do NOT use 0A/0B to recommend supplemental MRI — give a final category instead. |
| Category 6 | Management: "Surgical excision when clinically appropriate" | Management: "Clinical follow-up with surgeon and/or oncologist, and definitive local therapy (usually surgery) when clinically appropriate." Revised to recognize emerging non-surgical definitive therapies. NEW: Cat 6 may also be used for separate additional close findings (ACFs) within 2cm of biopsy-proven malignancy, if they do not increase total extent >2cm and would not change management. |
Ablation and other non-surgical therapies are now acknowledged. The ACF provision for Cat 6 avoids over-classifying small adjacent suspicious findings as Cat 4/5 when they fall within the surgical field. |
| Report organization | Variable by modality; mammography: 1. Indication 2. Breast composition 3. Findings 4. Comparison 5. Assessment 6. Management | Standardized across all modalities: 1. Indication 2. Comparison to prior exams 3. Technique 4. Breast density/composition 5. Findings 6. Assessment 7. Management recommendations. Structured Exam Indication verbiage standardized. | Technique is now a required report section. Comparison to priors moves up to section 2. Breast density becomes a mandatory standalone section (all modalities). |
| Mammography | |||
| DBT & Synthetic 2D | No more line drawings; all clinical images on digital equipment. DBT addressed but limited examples. | Standard DM, DBT, and synthetic mammogram (SM) examples all included. Mass definition updated: may be apparent on single projection when imaged on DBT (5th ed required 2 views). SM formally recognized: lower spatial resolution than true 2D, can produce pseudocalcifications (artifact not seen on tomo slices). | A mass seen on only one DBT view is valid. For SM: calcifications seen on SM but not tomo slices → likely pseudocalcification artifact. Confirm with true 2D or magnification. |
| Masses — Margin: Microlobulated | Microlobulated listed as a distinct margin descriptor (intermediate suspicion) | REMOVED as a margin descriptor. A microlobulated margin should now be described as indistinct. Reason: to avoid confusion with the shape term "lobulated." | Do not use "microlobulated margin" — use "indistinct margin" instead. This is one of the most impactful lexicon changes for daily reporting. |
| Masses — Shape: Lobulated | Masses/shape/lobular eliminated from 5th edition (to prevent confusion with Masses/Margin/Microlobulated) | RETURNED to lexicon as "lobulated" (not "lobular") — term changed to avoid confusion with the lobular subtype of breast cancer. Now valid in mammography, US, MRI, and CEM. | Lobulated shape is back. Use "lobulated" (not "lobular"). Applicable across all modalities for describing gently undulating, rounded mass contours. |
| Calcification terms removed | Popcorn-like, dystrophic, punctate (as parenthetical subset of round), and milk of calcium listed as distinct types | Popcorn-like → coarse. Dystrophic → coarse. Reason: simplify reporting and move away from food-related descriptors and histopathology-based terms. Punctate parenthetical removed from "round" (both refer to <0.5mm round particles; not practical to distinguish). Milk of calcium → layering (emphasizes morphologic appearance — sedimentation — rather than physiologic make-up). |
Use "coarse" for former popcorn/dystrophic. Use "layering" for former milk of calcium (smudgy on CC, layers on lateral view). "Round" now subsumes punctate. |
| Vascular calcifications | Listed as typically benign | Retained as typically benign; NOW noted to be associated with increased risk of cardiovascular disease per recent literature (referenced). Reporting encouraged when present. | Mention vascular calcifications in the report — they carry clinical significance beyond breast imaging (cardiovascular disease risk marker). |
| Calcification distribution table | Table 3: Likelihood of Malignancy as Function of BI-RADS Descriptors of Calcification Distribution | REMOVED. Table 3 (calcification morphology PPV) updated with more recent literature. Distribution likelihood table eliminated. | Morphology remains the primary driver for biopsy decision. Distribution (grouped, regional, segmental, etc.) provides context but no longer has its own PPV reference table. |
| "Developing asymmetry" | Listed as a distinct asymmetry subtype indicating interval change | REMOVED as a descriptor. Delineation of change over time no longer embedded in descriptor terminology. Now described as: an asymmetry (focal or global) that is "enlarging, becoming denser, or more conspicuous." | Do not use "developing asymmetry." Describe the finding type (focal/global asymmetry) and characterize the change in the report narrative. |
| Lymph nodes | Intramammary lymph node and axillary lymph nodes addressed as separate findings | Combined into a single Lymph Nodes section with sub-findings of intramammary and axillary. Multiple dilated ducts added as a NEW finding — considered typically benign. | Report intramammary and axillary nodes under unified "Lymph Nodes" heading. Multiple dilated ducts ≠ suspicious; solitary dilated duct requires more nuanced assessment (see below). |
| Solitary dilated duct | Considered suspicious unless benign etiology demonstrated; generally BI-RADS 4A | When not associated with suspicious imaging features (mass, architectural distortion, microcalcifications) and occurring in asymptomatic individuals, can be considered benign. If present on a baseline exam in a symptomatic woman or associated with other suspicious findings → additional imaging evaluation leading to possible tissue diagnosis should be considered. | Asymptomatic, isolated, no associated suspicious features → may be benign. No longer automatically 4A. Symptomatic or associated with suspicious findings → still warrants workup. |
| Associated features → Secondary findings | Axillary adenopathy, architectural distortion, and calcifications listed as associated features that could stand alone when no other abnormality present | Concept of "secondary findings" introduced: additional abnormalities present in association with a primary finding. Axillary adenopathy → moved to Lymph Nodes section. Architectural distortion → recategorized as a secondary finding. Calcifications → recategorized as a secondary finding. | Architectural distortion and calcifications are no longer "associated features" — they are secondary findings or can be primary findings in their own right. |
| Depth on MLO view | Determined by imaginary divisions based on vertically oriented lines | Determined by imaginary divisions that parallel the angle of the pectoralis major muscle. | MLO depth stratification (anterior/mid/posterior) now aligns with pec major angle, not vertical lines. |
| Special cases | Did not include gynecomastia, implants, or mastectomy as mammography special cases | Special cases reintroduced to include Gynecomastia, Implants and other forms of augmentation, and Mastectomy. Hormone-induced breast tissue in transfeminine patients should NOT be characterized as gynecomastia. | Gynecomastia, augmentation, and post-mastectomy imaging now have dedicated guidance in the mammography lexicon. |
| Ultrasound | |||
| Non-mass lesion | Not in previous version | NEW finding introduced: a discrete finding distinctly different from normal tissue, seen in 3 dimensions, but lacking the discrete margination of a mass and unable to be assigned a specific shape. Often subtle; may be detected only because background tissue is disrupted. | Non-mass lesion is now a reportable US finding — analogous to NME on MRI. Report location, size, and associated features. |
| Tissue composition — GTC | Tissue composition (single descriptor) | Expanded to: Tissue composition/tissue pattern + Tissue composition/glandular tissue component (GTC). GTC defined with supporting evidence. | GTC is a new sub-finding of tissue composition. Report pattern and GTC separately when applicable. |
| Posterior features — combined pattern | Combined pattern listed as a posterior feature option | Combined pattern removed. If a mass shows any shadowing, it should be characterized as shadowing. | Do not use "combined pattern." If shadowing is present at all → call it shadowing. |
| Echo pattern | Complex cystic and solid | Mixed solid and cystic (terminology revision) | Use "mixed solid and cystic" — "complex cystic and solid" is retired. |
| Associated features — new descriptors | Architectural distortion, duct changes, skin changes, edema, vascularity, elasticity assessment | Echogenic pseudocapsule and echogenic rind added as sub-findings to define tissue directly surrounding a finding. Vascularity terminology: "absent" → avascular; "vessels in rim" → peripheral vascularity. | Echogenic pseudocapsule and echogenic rind are new reportable features. Update vascularity terminology accordingly. |
| Special cases — new entries | Did not include foreign body, abscess, or post-traumatic (non-surgical) changes as explicit special cases | Added: foreign body, implants (as sub-finding), postsurgical changes including fluid collections, fat necrosis, post-traumatic (non-surgical) changes, abscess. Lymph nodes expanded to include intramammary, axillary, internal mammary, and supraclavicular staging. | Abscess and post-traumatic changes are now explicit US special case categories. Lymph node reporting expanded to include internal mammary and supraclavicular nodes. |
| MRI | |||
| Focus | Focus listed as a distinct finding (<5mm enhancing dot) | ELIMINATED as a finding. Focus has been removed from the MRI lexicon entirely. | Do not report "focus" on MRI. Describe small enhancing dots within mass or NME descriptors, or note as too small to characterize. This is a significant change from 5th edition practice. |
| BPE — minimal | Background parenchymal enhancement: minimal/mild/moderate/marked | BPE/minimal now explicitly includes no enhancement (terminology revision). Four-tier scale retained. | "No enhancement" and "minimal enhancement" are now the same BPE category. Do not create a separate "none" category. |
| Masses — Margin: Irregular | Masses/Margin/Not circumscribed/Irregular listed as a margin sub-descriptor | "Irregular" replaced by "indistinct" under non-circumscribed margins — to avoid duplication with "irregular" as a shape descriptor, and to harmonize with margin descriptors across other modalities. | Use "indistinct" not "irregular" for non-circumscribed MRI masses. Applies to CEM as well. Harmonizes with the mammography change (microlobulated → indistinct). |
| T2 signal intensity | Not in previous version as a mass sub-finding | T2 signal intensity added as a mass sub-finding: hyperintense / not hyperintense. | Report T2 signal of masses: hyperintense (e.g., fibroadenoma, cyst, mucinous carcinoma) vs. not hyperintense. Adds important characterization context. |
| NME — distribution: multiple regions | Non-mass enhancement/distribution/multiple regions listed as a distribution descriptor | REMOVED as a descriptor. | Do not use "multiple regions" as NME distribution. Describe each region separately or use the appropriate distribution term (diffuse, regional, etc.). |
| Enhancement kinetics | Kinetic curve assessment / initial phase | Kinetic curve assessment → Enhancement kinetics. Initial phase → Early phase (terminology revision). | Use "enhancement kinetics" and "early phase" in reports. "Kinetic curve" and "initial phase" are retired terms. |
| Internal enhancement — rim | Rim enhancement | Thick rim enhancement (terminology revision — "thick" added for precision) | Use "thick rim enhancement." Plain "rim enhancement" is retired. |
| Associated features: "invasion" → "involvement" | Skin invasion, nipple invasion, pectoralis muscle invasion, chest wall invasion | "Invasion" changed to "involvement" across all associated features: skin involvement, nipple involvement, pectoralis muscle involvement, chest wall involvement. Peritumoral edema added as a new descriptor. Architectural distortion removed from associated features. | Do not use "invasion" in MRI reports — use "involvement." Peritumoral edema is now a reportable MRI associated feature. |
| MRI Report organization | 1. Indication 2. MRI technique 3. Breast composition 4. Findings 5. Comparison 6. Composite reports 7. Assessment 8. Management | 1. Indication 2. Comparison to prior 3. Acquisition parameters 4. Amount of fibroglandular tissue (FGT) 5. Level of BPE 6. Findings 7. Assessment 8. Management recommendations. BPE added as mandatory standalone section. | BPE must be explicitly reported. "Breast composition" → FGT. Acquisition parameters (including abbreviated protocol or DWI use) is now a required section. |
| Audit & Outcomes | |||
| Cancer definition for audit | Tissue diagnosis of DCIS or any type of primary (not metastatic) breast cancer | Updated: tissue diagnosis of DCIS, pleomorphic or florid lobular carcinoma in situ, or any type of primary (not metastatic) invasive breast carcinoma. | PLCIS and FLCIS now count as cancer for audit purposes — important for tracking upgrade rates from high-risk lesions. |
| Category 3 outcomes in audit | Not included in basic clinically relevant audit | Outcomes for initial BI-RADS Category 3 assessments now included as part of the basic clinically relevant audit (new to v2025). | Track and report Category 3 outcomes (cancer found at follow-up) as part of standard quality metrics — not just Category 4/5. |
Use when additional imaging evaluation is needed (spot compression, magnification, ultrasound). Do NOT use Category 0 to recommend MRI for already-suspicious findings — use Category 4 or 5.
NEW per MQSA amendments effective Sept 10, 2024. Prior mammograms required before final assessment can be rendered.
No mammographic evidence of malignancy. No findings described (differs from Category 2 where a benign finding is explicitly noted).
Normal assessment with ≥1 described benign finding. Examples: calcified fibroadenoma, lipoma, oil cyst, skin calcification, vascular calcification.
Short-interval follow-up recommended. NOT recommended if finding has substantially increased or patient has known cancer. Examples: non-calcified circumscribed solid mass, focal asymmetry, cluster of round/punctate calcifications.
Low suspicion for malignancy. Examples: palpable partially circumscribed solid mass, new asymmetry.
Moderate suspicion for malignancy. Examples: indistinct margin mass, grouped amorphous calcifications.
High suspicion but not classic malignancy appearance. Examples: irregular spiculated mass, fine pleomorphic calcifications.
Classic appearance of malignancy. Examples: spiculated high-density mass, linear/branching calcifications, spiculated mass + pleomorphic calcifications.
Awaiting definitive therapy. Used for imaging prior to surgical excision or during neoadjuvant therapy monitoring.
Breast Density
ACR BI-RADS 2025 — FDA MQSA notification requirements effective Sept 10, 2024
Sensitivity of mammography highest. Cancer not likely obscured.
Some areas may obscure small masses.
May obscure small masses. Supplemental screening may be beneficial.
Lowest mammographic sensitivity (~50%). Supplemental screening recommended.
Supplemental Screening Options
| Density | Modality | Recommendation | Notes |
|---|---|---|---|
| C/D | Ultrasound | Consider (average risk) | Detects additional 2–4/1000; increased false positives |
| C/D | MRI | Recommended (high risk ≥20% lifetime) | Most sensitive; requires contrast |
| C/D | Contrast-Enhanced Mammography (CEM) | Emerging option | Similar sensitivity to MRI; radiation + contrast |
| D | ABUS (automated US) | Consider | FDA-approved supplemental screening |
Screening Recommendations
ACR 2024 guidelines — risk-stratified approach
| Age | Recommendation | Notes |
|---|---|---|
| <40 | No routine screening | Unless risk factors present |
| 40–44 | Annual mammography offered (optional) | Patient choice; discuss benefits/risks |
| 45+ | Annual screening mammography | ACR/SBI recommend starting at 40 |
| 40+ | 2D vs DBT | DBT preferred; higher CDR, lower recall rate |
| Any | US supplemental | Consider if dense breasts (C/D) + average risk |
| Modality | Timing | Indication |
|---|---|---|
| Annual screening MRI | Starting age 25–30 | BRCA1/2, TP53, PTEN, STK11/CDH1 mutations |
| Annual mammography | Starting age 30 (or 10 yrs before youngest family dx) | High-risk by Tyrer-Cuzick or other models |
| Annual MRI + mammo | Alternating every 6 months | BRCA carriers per NCCN |
| Clinical breast exam | Every 6–12 months | All high-risk patients |
| Modality | Recommendation |
|---|---|
| Annual mammography | Standard |
| Consider MRI supplemental | Individualized; discuss with patient |
| Annual clinical breast exam | Recommended |
Mammography — Masses
ACR BI-RADS 5th Ed. mammography lexicon
| Term | Description | Suspicion |
|---|---|---|
| CircumscribedRA | Well-defined; sharp transition | Benign — requires ≥75% circumscribed |
| Obscured | Hidden by superimposed tissue; ≥25% obscured | Indeterminate — needs additional views |
| MicrolobulatedRA | Short-cycle undulations | Intermediate |
| IndistinctRA | No clear demarcation from surrounding tissue | Suspicious |
| SpiculatedRA | Lines radiating from mass | Highly Suspicious |
| Term | HU Equivalent | Notes |
|---|---|---|
| High density | Denser than equal volume of fibroglandular tissue | Suspicious — possible malignancy |
| Equal density | Same as fibroglandular tissue | Indeterminate |
| Low density | Less dense than equal volume of tissue | Typically benign (lipid-containing) |
| Fat-containing | Contains fat (lucent areas) | Benign — lipoma, oil cyst, hamartoma, LN |
Mammography — Calcifications
Morphology and distribution — the most important section in mammography BI-RADS
| Term | Appearance | Assessment | |
|---|---|---|---|
| Typically Benign | |||
| Skin (dermal) | 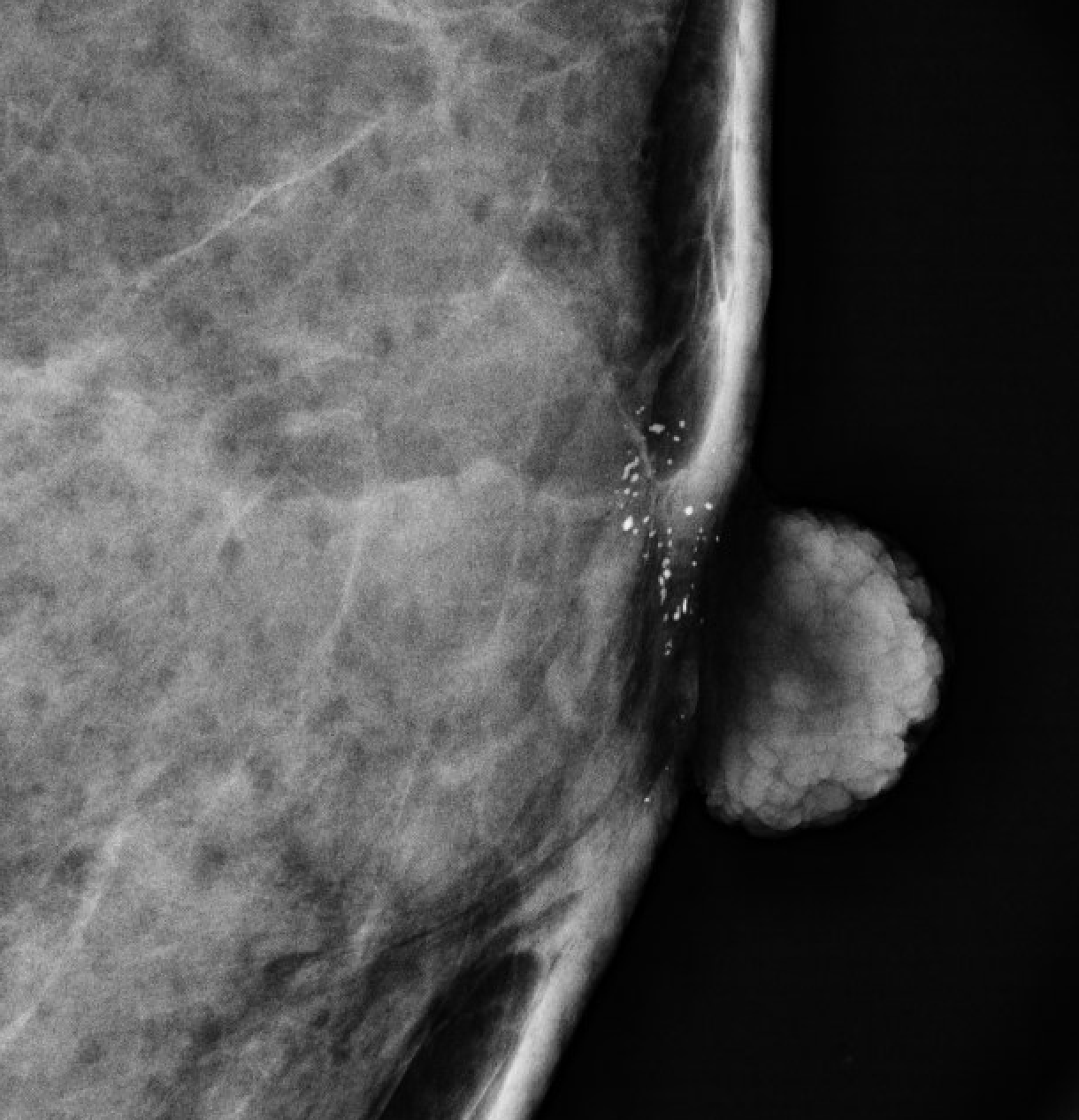 | Lucent center; confirm with tangential view | BI-RADS 2 |
| Vascular | 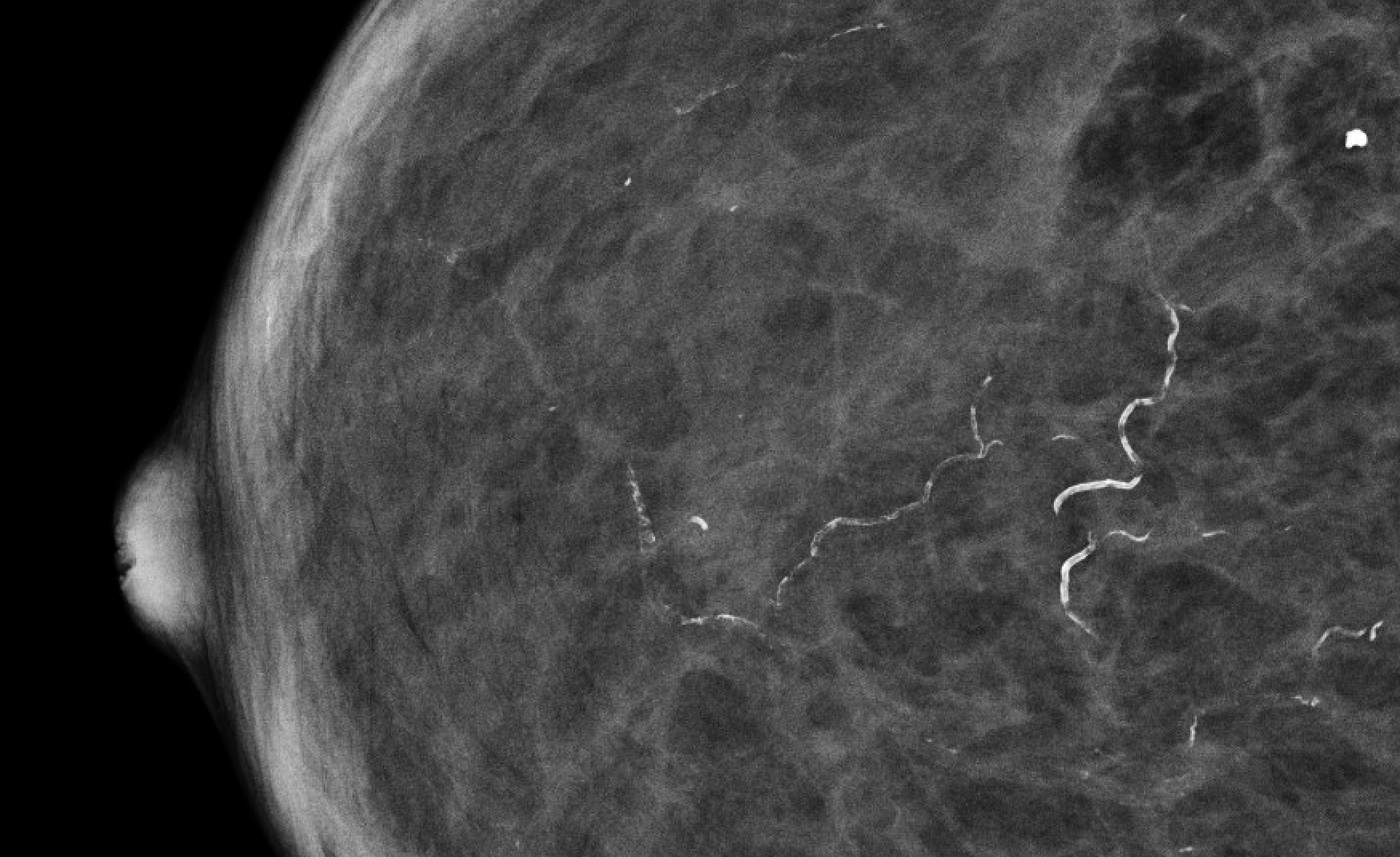 | Parallel tracks or tram-tracks along vessels | BI-RADS 2 |
| Coarse/popcorn | 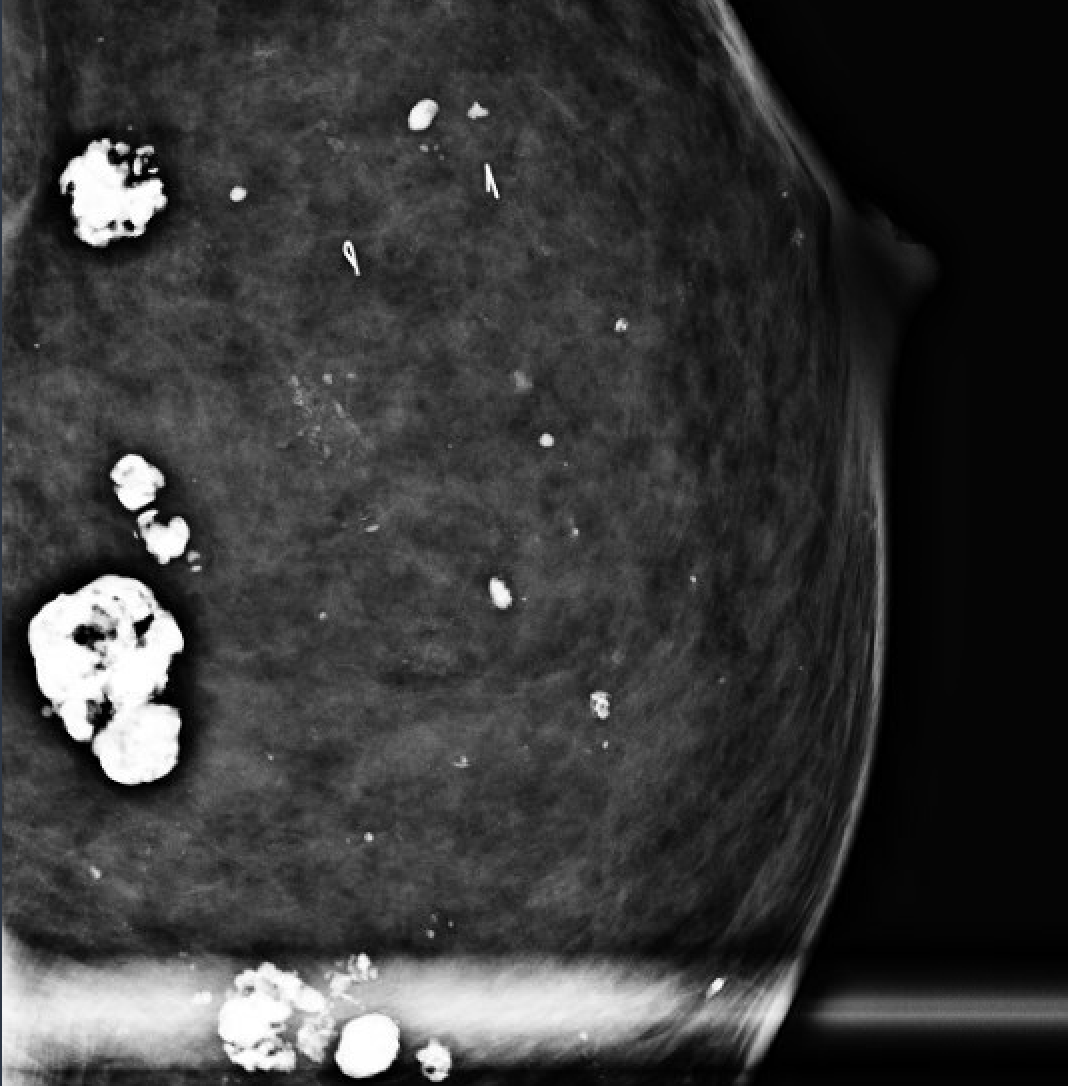 | Involuting fibroadenoma; >1mm, dense | BI-RADS 2 |
| Large rod-like | 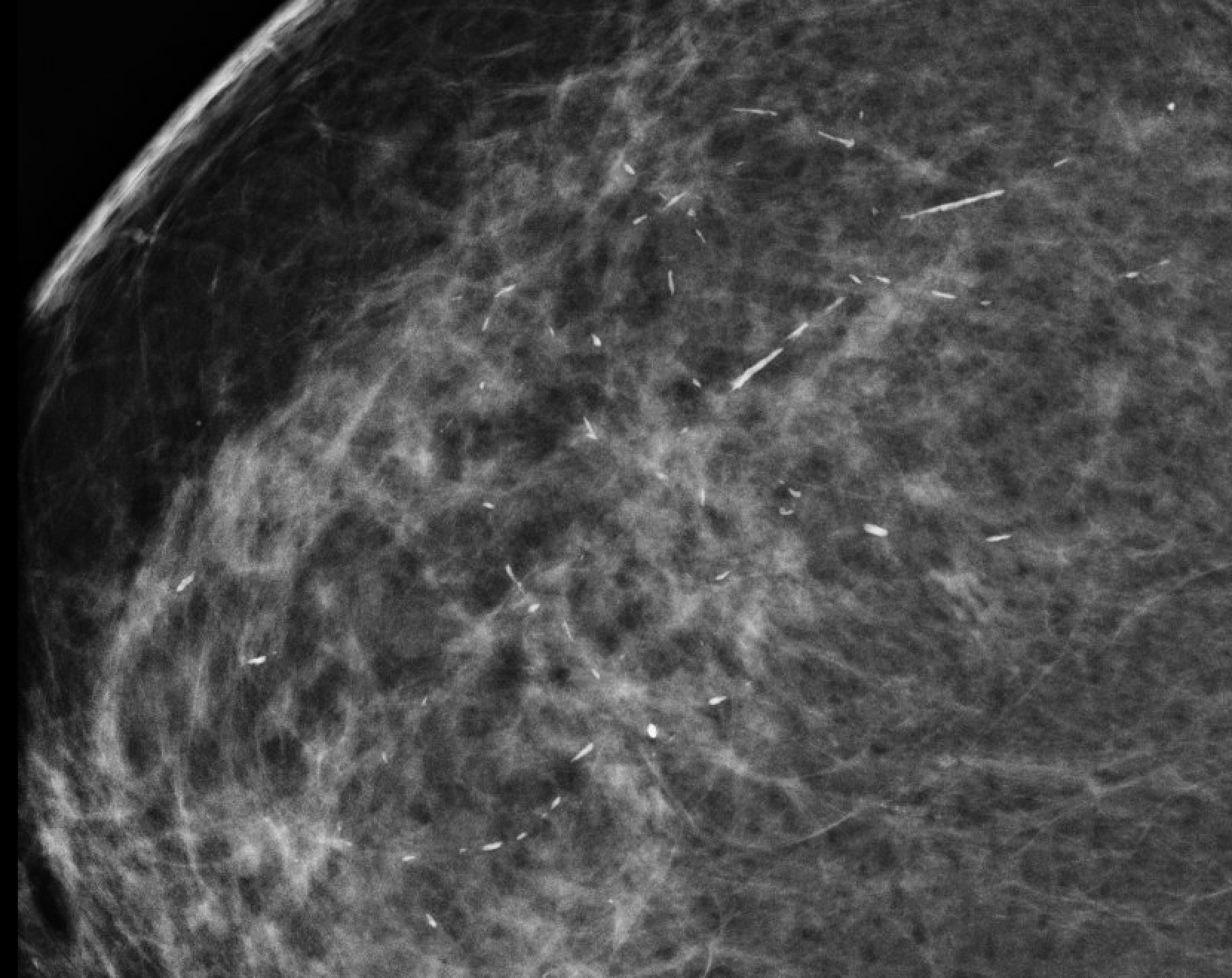 | Plasma cell mastitis; >1mm rods, may branch | BI-RADS 2 |
| Round |  | ≥1mm spheres, uniform | BI-RADS 2 or 3 |
| Lucent-centered |  | Round/oval with lucent center | BI-RADS 2 |
| Eggshell/rim |  | Thin calcification on sphere surface (oil cyst) | BI-RADS 2 |
| Milk of calcium |  | Tea-cup on lateral; smudgy on CC | BI-RADS 2 |
| Dystrophic | 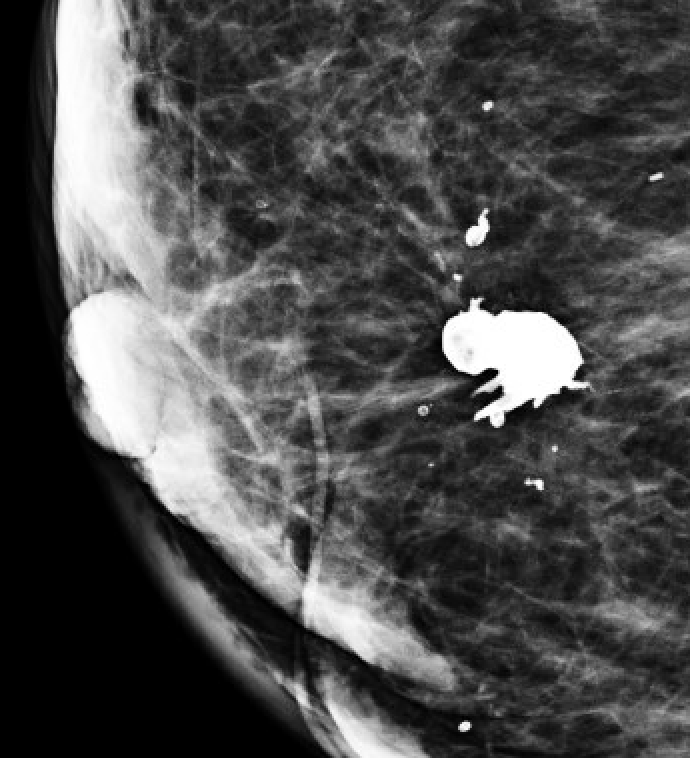 | Irregular, >0.5mm; post-trauma/radiation | BI-RADS 2 |
| Suture | 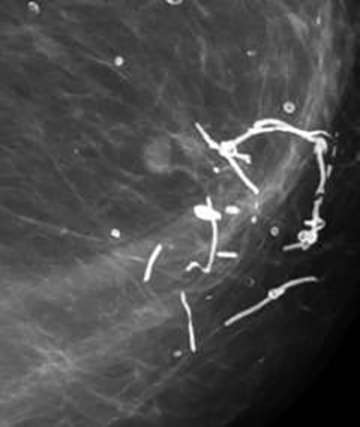 | Tubular/linear; post-radiation | BI-RADS 2 |
| Intermediate Concern | |||
| AmorphousRA | 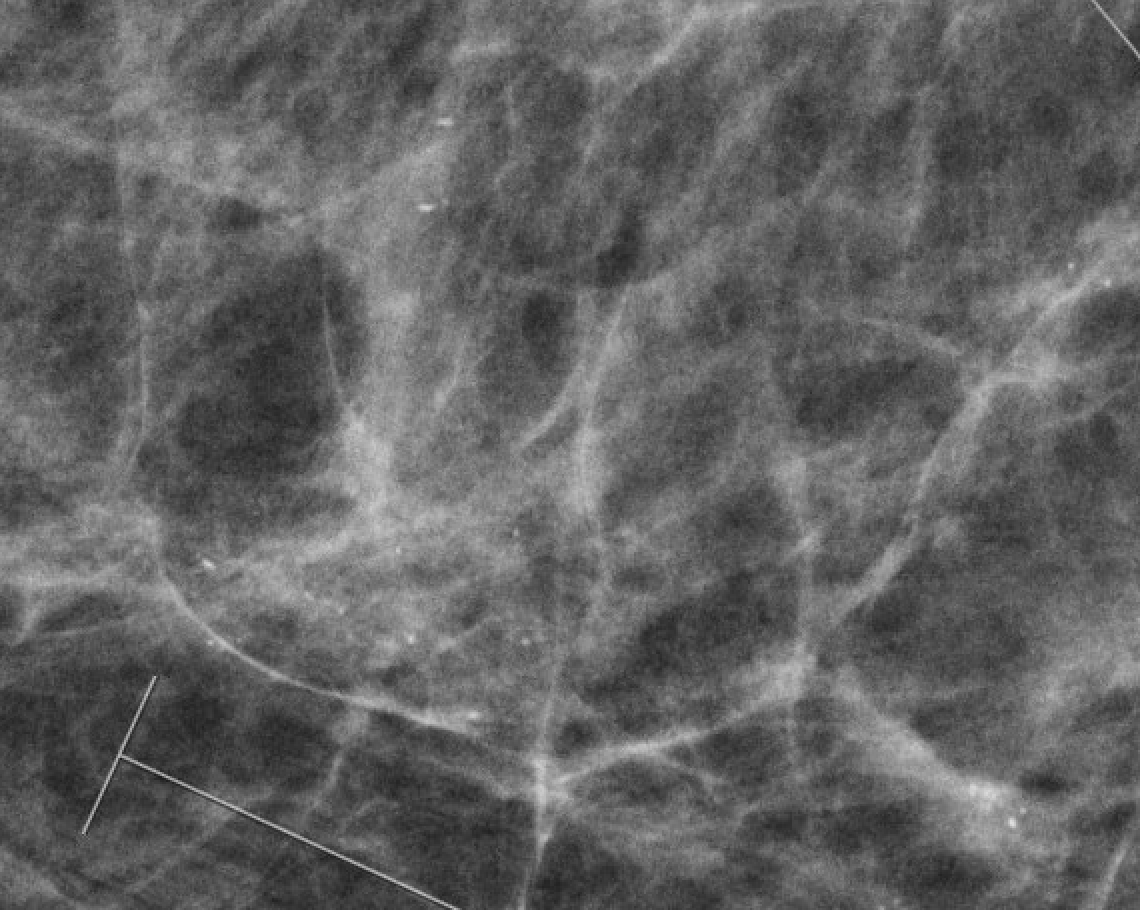 | Indistinct, cloudlike; too small to characterize | BI-RADS 4B |
| Coarse heterogeneousRA | 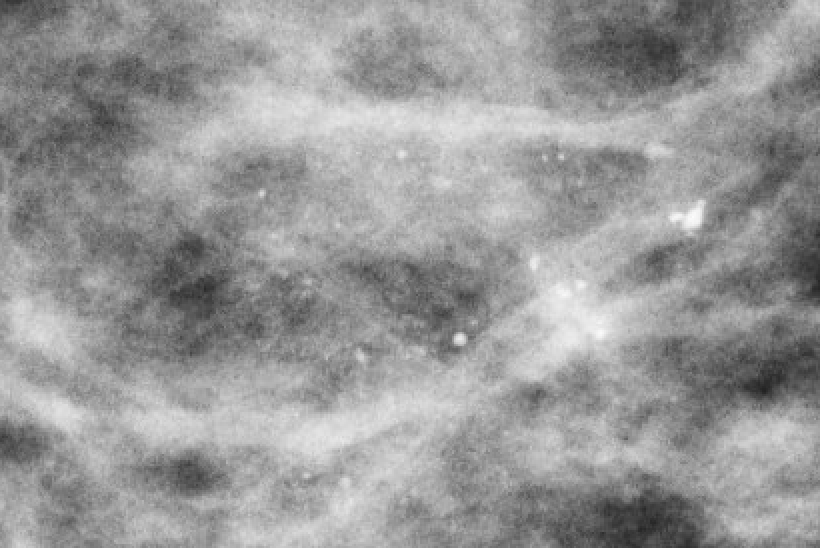 | Irregular, >0.5mm but not benign morphology | BI-RADS 4A–4B |
| Higher Suspicion | |||
| Fine pleomorphicRA | 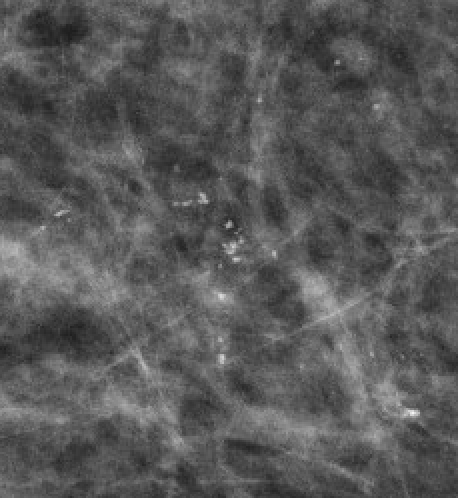 | Irregular, <0.5mm, varied shapes | BI-RADS 4B–4C |
| Fine linear/branchingRA | 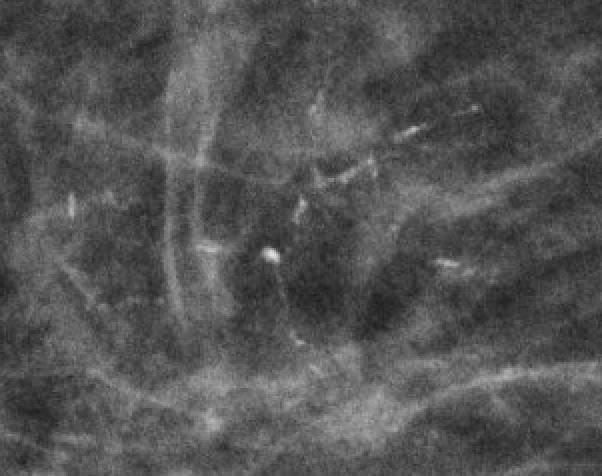 | Thin irregular lines; filling ductal lumen | BI-RADS 4C–5 |
| Term | Definition | Significance |
|---|---|---|
| DiffuseRA | Random throughout breast | Typically benign (bilateral) |
| RegionalRA | Large portion of breast tissue (not ductal) | May be benign or suspicious |
| Grouped (clustered)RA | ≥5 calcifications in <2cm | Suspicious if morphology suspicious |
| LinearRA | In a line (may branch); suggests ductal | Suspicious |
| SegmentalRA | Wedge-shaped; ductal/lobular distribution | Highly Suspicious |
Mammography — Architectural Distortion
Spiculation without a definite mass center
| Cause | Features | Management |
|---|---|---|
| Radial scar/CSL | Mimics cancer; central lucency on 2D; may be invisible on one view | Biopsy (upgrade rate 0–8%) |
| Post-surgical scar | History of surgery; stable over time | Compare to prior; stable = BI-RADS 2 |
| Invasive carcinoma | New, increasing, associated mass/calcs | BI-RADS 4–5; biopsy |
| Sclerosing adenosis | May appear as distortion | Biopsy to exclude malignancy |
| Fat necrosis | Post-trauma/biopsy history; may have oil cyst | Clinical correlation |
Asymmetries & Associated Features
Asymmetric findings and secondary signs of malignancy
| Type | Definition | Assessment |
|---|---|---|
| Asymmetry | Seen on one view only | Likely summation; needs confirmation |
| Global asymmetry | Occupies ≥1 quadrant; no mass, distortion, or calcs | Usually benign if stable; new = BI-RADS 0/3 |
| Focal asymmetry | <1 quadrant; seen on 2 views; no central mass | New/growing = BI-RADS 4; stable = BI-RADS 3 |
| Developing asymmetry | New or enlarging focal asymmetry vs. prior | BI-RADS 4 — high PPV (~15%) |
| Feature | Significance |
|---|---|
| Skin retraction | Suspicious — Cooper's ligament involvement |
| Nipple retraction | Suspicious if new |
| Skin thickening | Inflammatory cancer vs. benign skin change |
| Trabecular thickening | Lymphedema, inflammatory cancer, or benign |
| Axillary adenopathy | Suspicious if enlarged/dense — biopsy indicated |
| Architectural distortion | Secondary sign when associated with mass |
| Skin lesion | Mark with BBs if applicable |
Special Cases — Mammography
Specific entities with established imaging features
| Entity | Features | BI-RADS | Notes |
|---|---|---|---|
| Simple cyst | Round/oval; circumscribed; may resolve | 2 | Confirm with US |
| Clustered microcysts | ≤3mm anechoic spaces; thin septa | 2 | If purely simple on US |
| Complicated cyst | Homogeneous low-level echoes | 3 | 6-mo follow-up if single |
| Skin lesion | With BB marker; identifiable as skin | 2 | Use tangential view |
| Intramammary lymph node | Reniform; fatty hilum; ≤1cm; upper-outer quadrant | 2 | Classic location and morphology required |
| Hamartoma (fibroadenolipoma) | Mixed density; pseudocapsule; "breast within breast" | 2 | Pathognomonic appearance |
| Vascular calcifications | Tram-track pattern along vessels | 2 | Calcified vessel walls |
| Fat necrosis | Oil cyst, dystrophic calcs, lipid-filled mass | 2 | History of trauma/surgery |
| Diabetic mastopathy | Ill-defined dense mass; US shows posterior shadowing | 3–4 | Needs biopsy |
| Mondor disease | Thrombophlebitis of superficial vein | 2 | Clinical and US diagnosis |
Digital Breast Tomosynthesis (DBT)
3D mammography — technical considerations and clinical applications
DBT vs 2D Comparison
| Parameter | 2D FFDM | DBT |
|---|---|---|
| Cancer detection rate | Baseline | +1–2.5/1000 improvement |
| Recall rate | Baseline | Reduces by 15–40% |
| Architectural distortion detection | Limited | Significantly improved |
| Calcification evaluation | Standard | May need 2D for calcification morphology |
| Radiation dose | 1× | ~2× (DBT + synthetic 2D); similar if C-View used |
| Insurance coverage | Standard | Now widely covered |
Synthetic 2D (C-View / Insight 2D)
Reconstructed 2D from DBT data eliminates need for acquired 2D. Comparable sensitivity to FFDM. Reduces total dose to ~1.25× FFDM. Standard of practice in most centers.
DBT-Specific Findings
- Architectural distortion: much more conspicuous on DBT; responsible for most incremental cancers detected
- Masses: better margin characterization; reduced superimposition
- Calcifications: BI-RADS 2025 recommends reviewing calcifications on both DBT and 2D (synthetic); fine morphology better on acquired 2D
- Asymmetries: many 2D asymmetries resolve (summation artifact) on DBT
Ultrasound — Mass Characteristics
ACR BI-RADS US lexicon
| Term | Description | Suspicion |
|---|---|---|
| RoundRA | Spherical | Low if circumscribed |
| OvalRA | Elliptical; up to 3 gentle lobulations | Low |
| IrregularRA | Neither round nor oval | Suspicious |
| Parallel (wider-than-tall)RA | Long axis parallel to skin | Benign orientation |
| Not parallel (taller-than-wide)RA | AP dimension > transverse | Suspicious — violates tissue planes |
| Term | Notes | Suspicion |
|---|---|---|
| CircumscribedRA | Abrupt transition; no angular margin | Benign |
| Not circumscribed | Any of: indistinct, angular, microlobulated, spiculated | Suspicious |
| IndistinctRA | No clear edge | Suspicious |
| AngularRA | Sharp angles; infiltrating growth | Suspicious |
| MicrolobulatedRA | Short-cycle undulations | Intermediate |
| SpiculatedRA | Radiating lines | Highly Suspicious |
| Term | Appearance | Association |
|---|---|---|
| Anechoic | No internal echoes | Simple cyst (BI-RADS 2) |
| Hyperechoic | Echogenicity > fat | Usually benign (lipoma, fat lobule) |
| Complex cystic & solid | Mixed solid/cystic | Biopsy if symptomatic or new |
| HypoechoicRA | Less echogenic than fat | Variable — needs full characterization |
| Isoechoic | Same as fat | Variable |
| Heterogeneous | Mixed echo pattern | Indeterminate |
| Term | Significance |
|---|---|
| No posterior features | Indeterminate |
| Enhancement (posterior acoustic enhancement) | Benign — fluid-filled structure |
| ShadowingRA | Suspicious — fibrous or malignant tissue |
| Combined pattern | Indeterminate |
| Calcifications in mass | Highly suspicious if coarse |
| Architectural distortion | Suspicious |
| Duct changes | Suspicious |
| Skin changes | Thickening/retraction = suspicious |
| Edema | Inflammatory process |
| Vascularity | Internal vascularity increases suspicion |
Ultrasound — Cysts & Special Cases
Classification and management
| Type | US Features | BI-RADS | Management |
|---|---|---|---|
| Simple cyst | Anechoic; circumscribed; posterior enhancement; no internal echoes; thin wall | 2 | Routine screening; no follow-up needed |
| Clustered microcysts | Multiple ≤3mm anechoic spaces; thin (<0.5mm) septa; no solid component | 2 | Routine follow-up |
| Complicated cyst | Homogeneous low-level internal echoes; circumscribed; no thick wall/solid component | 3 | 6-month follow-up if single; if multiple = BI-RADS 2 |
| Complex cystic & solid | Thick wall (>0.5mm), thick septation, intracystic solid mass, or mixed solid/cystic | 4 | Biopsy |
| Abscess | Thick irregular wall; internal debris; tender; clinical fever | — | Clinical management; aspiration |
| Galactocele | Complex cyst in lactating/post-lactating woman | 2–3 | Clinical |
| Oil cyst (fat necrosis) | Circumscribed; anechoic to complex; post-trauma history | 2 | No follow-up if classic |
| Sebaceous/epidermal inclusion cyst | Skin origin; posterior shadowing | 2 | No action if classic |
Ultrasound — Lymph Nodes
Axillary, intramammary, and regional assessment
| Feature | Normal | Abnormal/Suspicious |
|---|---|---|
| Shape | Reniform (kidney-shaped) | Round |
| Hilum | Echogenic fatty hilum present | Absent fatty hilum |
| Cortex | ≤3mm uniform cortical thickness | Focal cortical thickening >3mm |
| Size | Variable; cortex thickness more important than size | Entire node >2cm without fatty hilum |
| Vascularity | Hilar flow pattern | Peripheral/internal vascularity |
| Number | Multiple normal-appearing | Single enlarged abnormal node |
Management
| Finding | Action |
|---|---|
| Normal axillary LN | BI-RADS 1/2 — no action |
| Abnormal (focal cortical thickening) | BI-RADS 4 — consider biopsy (FNA or CNB) |
| Intramammary LN | BI-RADS 2 if classic reniform with fatty hilum in upper outer breast |
| Metastatic workup | Staging CT or PET-CT if biopsy-proven axillary mets |
Vascularity & Elastography
Doppler and strain/shear-wave assessment
| Finding | Interpretation | Action |
|---|---|---|
| Absent vascularity | Simple cyst; or avascular solid mass | Not independently suspicious |
| Internal vascularity | More common in malignancy | Increases suspicion |
| Peripheral vascularity | Circumferential rim vessels | More common in benign lesions |
| Increased hilar flow | Normal LN | Reassuring |
| Absent hilar flow | Metastatic LN | Suspicious |
| Technique | Principle | Malignancy Feature |
|---|---|---|
| Strain elastography | Compression; relative stiffness | Stiff (blue in standard colormap) = suspicious |
| Shear-wave elastography (SWE) | Acoustic push; quantitative kPa | >80 kPa highly suspicious; <30 kPa reassuring |
MRI — Background Parenchymal Enhancement
BPE assessment and clinical relevance
Optimal exam; highest conspicuity for enhancement.
Generally adequate for interpretation.
May limit lesion detection; consider timing (days 7–14 of cycle).
Significantly limits interpretation; consider repeating in follicular phase (days 7–14).
Symmetry
| Type | Pattern | Interpretation |
|---|---|---|
| Symmetric BPE | Bilateral, similar distribution | Expected finding — document, continue interpretation |
| Asymmetric BPE | Unilateral or focal | Warrants attention — consider pathologic enhancement |
Clinical Factors
| Factor | Effect on BPE | Action |
|---|---|---|
| Premenopausal | Higher BPE (luteal phase) | Schedule days 7–14 of cycle |
| Postmenopausal | Lower BPE (no HRT) | Optimal timing |
| HRT use | Elevated BPE | Document; may limit sensitivity |
| Tamoxifen/AIs | Reduce BPE | Associated with treatment response |
| Post-RT (treated breast) | Reduced/absent BPE | Expected; compare with contralateral |
MRI — Masses & Enhancement Kinetics
Morphology, internal enhancement pattern, and kinetic curve analysis
Shape
| Shape | Description | Suspicion |
|---|---|---|
| Round | Spherical | Lower suspicion |
| Oval | Elliptical; ≤3 undulations | Lower suspicion |
| Irregular | Neither round nor oval | Suspicious |
Margin
| Margin | Interpretation |
|---|---|
| Circumscribed | Benign-leaning — sharp, well-defined |
| Irregular | Suspicious — uneven, jagged |
| Spiculated | Highly Suspicious — radiating projections |
| Pattern | Description | PPV for Malignancy |
|---|---|---|
| Homogeneous | Uniform; confluent | Lower (~15–20%) |
| Heterogeneous | Non-uniform | Intermediate |
| Rim enhancementRA | Peripheral > central | Higher (~40–50%) |
| Dark internal septations | Low-signal septa within enhancing mass | Benign (fibroadenoma feature) |
| Enhancing internal septations | Enhancing septae | More suspicious |
| Central enhancement | Center more than periphery | Variable |
- Slow (<50% signal increase) — Benign/indeterminate
- Medium (50–100% increase) — Indeterminate
- Rapid (>100% increase) — Suspicious
Signal continues to rise. Benign pattern. PPV ~6%
Signal stabilizes. Indeterminate. PPV ~25%
Signal decreases after peak. Suspicious. PPV ~60–70%
MRI — Non-Mass Enhancement (NME)
Enhancement without a discrete 3D mass — the most important MRI section
| Distribution | Definition | Suspicion Level |
|---|---|---|
| FocalRA | <25% of a quadrant; may be single focus | Low–Intermediate |
| LinearRA | In a line; may branch; suggests ductal | Suspicious |
| SegmentalRA | Wedge-shaped; apex toward nipple; ductal/lobular unit | Highly Suspicious — PPV ~65–75% |
| RegionalRA | Large area; not ductal distribution; ≥25% of quadrant | Intermediate |
| Multiple regions | ≥2 regions separated by fat | Usually benign (bilateral BPE) or multifocal DCIS |
| Diffuse | Throughout most of breast | Usually BPE or treatment effect |
| Pattern | Description | Suspicion |
|---|---|---|
| Homogeneous | Uniform enhancement | Lowest suspicion (may be BPE) |
| Heterogeneous | Variable enhancement | Intermediate |
| ClumpedRA | Cobblestone/confluent foci in non-mass area | Suspicious — PPV ~45% |
| Clustered ringRA | Ring-enhancing foci clustered together | Highest Suspicion — PPV ~60–79% |
Key Clinical Scenarios
| Scenario | Interpretation / Action |
|---|---|
| Segmental NME + clumped | DCIS until proven otherwise → BI-RADS 4C–5 |
| Linear NME | Consider DCIS → BI-RADS 4B–4C; biopsy |
| Regional homogeneous NME bilateral | BPE → BI-RADS 1–2 |
| Focal NME new | BI-RADS 3–4A; consider 6-month follow-up vs. biopsy |
MRI — Associated Features & Clinical Scenarios
Secondary signs, implant assessment, and high-risk indications
| Feature | Significance |
|---|---|
| Nipple retraction | New: suspicious; correlate with clinical exam |
| Skin thickening | >3mm; inflammatory carcinoma vs. benign skin change |
| Skin enhancement | Direct dermal involvement — T4d staging |
| Pectoralis muscle invasion | T4a staging; contact is not invasion |
| Chest wall invasion | Requires muscle/rib involvement |
| Axillary adenopathy | Abnormal morphology: round, no hilum, cortex >3mm |
| Peri-tumoral edema | Common in IDC; not independently suspicious |
| Satellite lesions | Additional foci near index cancer — affects surgical planning |
| Duct extension | Ductal carcinoma pathway toward nipple |
| Finding | Implant Type | Significance |
|---|---|---|
| Intracapsular rupture | Silicone | Linguini sign (collapsed shell); keyhole sign |
| Extracapsular rupture | Silicone | Free silicone outside capsule; snowstorm on US |
| Gel bleed | Silicone | Normal finding; subclinical silicone permeation |
| Capsular contracture | Any | Clinical; Baker grade I–IV |
| Normal saline implant | Saline | Valve or fold artifact; no rupture assessment needed |
- BRCA1/2 mutation carrier or untested first-degree relative of carrier
- Lifetime risk ≥20% by Tyrer-Cuzick or other validated model
- History of chest/mediastinal RT between ages 10–30
- Li-Fraumeni, Cowden, Bannayan-Riley-Ruvalcaba syndrome
- TP53, PTEN, STK11, or CDH1 pathogenic variant
- Personal history of breast cancer + dense breasts OR diagnosed ≤50 (individualized)
Reporting — Required Elements
MQSA-compliant breast imaging report structure
| Element | Requirement |
|---|---|
| Patient information | Name, DOB, date of exam |
| Indication | Screening vs. diagnostic; clinical history |
| Breast composition | Breast density category A–D (lay language required) |
| Findings | Description using ACR BI-RADS lexicon |
| Comparison | Prior study date and findings if available |
| Impression | Final BI-RADS category (0A, 0B, 1, 2, 3, 4A, 4B, 4C, 5, 6) |
| Recommendation | Specific management recommendation matching category |
| Dense breast notification | Required for C/D; lay language; per MQSA 2024 |
| Radiologist signature | Required; interpreting physician credentials |
| Parameter | Screening | Diagnostic |
|---|---|---|
| Indication | Asymptomatic; routine | Symptom, palpable finding, abnormal screening, high-risk |
| Immediate read | Not required (batch reading) | Real-time read required |
| Additional views | Not performed at time of exam | May be performed during exam |
| Category 0 | Acceptable | Avoid — resolve to final category |
| Radiologist on-site | Not required | Required (per most protocols) |
- Imaging-pathology concordance must be assessed for all biopsy results
- Concordant benign: imaging features match benign pathology → routine follow-up
- Concordant malignant: imaging features match malignancy → proceed with treatment
- Discordant: pathology does not explain imaging findings → repeat biopsy or excision
- Upgrade at excision: CNB shows high-risk lesion (ADH, ALH, LCIS, radial scar, papilloma) and excision shows carcinoma
Audit & Quality Metrics
ACR BI-RADS 2025 — outcome monitoring and benchmarking
| Metric | Definition | ACR Benchmark |
|---|---|---|
| Recall rate (screening) | % of screening exams recalled for additional imaging | 5–12% (avg ~10%) |
| Cancer detection rate (CDR) | Cancers detected per 1,000 screening exams | ≥2.5/1,000 (avg ~5/1,000 with DBT) |
| PPV1 (abnormal interpretation) | Cancer found / all recalled exams | 3–8% (avg ~5%) |
| PPV2 (biopsy recommended) | Cancer found / all biopsy recommendations | 20–40% (avg ~25–35%) |
| PPV3 (biopsy performed) | Cancer found / all biopsies performed | 25–40% |
| Sensitivity | True positives / (true positives + false negatives) | ≥75% (goal ~85%) |
| Specificity | True negatives / (true negatives + false positives) | ≥88% |
| Stage distribution | % cancers detected at Stage 0/I | Goal >50% Stage 0/I |
- All BI-RADS assessments must be tracked
- Mandatory linkage of outcomes (cancer yes/no) to imaging interpretation
- Annual audit report required by ACR accreditation
- Benchmark comparison against national norms
- Definition: Cancer diagnosed within 12 months of a negative or benign screening exam
- Target: <2 per 1,000 negative screens
- High interval cancer rate suggests: increased breast density, rapidly growing tumors, or interpretation errors
- DBT reduces interval cancer rate by ~15–25% compared to 2D mammography
Mammography — Lymph Nodes
Intramammary and axillary lymph node assessment on mammography
| Feature | Normal | Abnormal |
|---|---|---|
| Shape | Reniform (kidney-shaped) | Round, loss of normal contour |
| Fatty hilum | Present — echogenic/lucent center | Absent SUSPICIOUS |
| Size | Typically <1 cm; variable | >2 cm without fatty hilum |
| Location | Upper outer quadrant, along vessels | Any quadrant — raises suspicion |
| Number | Solitary or few | Multiple enlarged nodes |
| Cortex | Thin, uniform | Focal cortical thickening >3 mm SUSPICIOUS |
| Finding | Assessment | Action |
|---|---|---|
| Normal morphology | BI-RADS 1/2 | Routine |
| Enlarged, maintains fatty hilum | BI-RADS 2–3 | Correlate clinically |
| Dense/replaced hilum | BI-RADS 4 SUSPICIOUS | Ultrasound ± biopsy |
| Multiple dense nodes, bilateral | Consider systemic cause | Clinical correlation (lymphoma, reactive, sarcoid) |
| Unilateral dense nodes with known ipsilateral cancer | Staging N1 | FNA or CNB for staging |
Skin Lesions & Dilated Ducts
Cutaneous findings and ductal pathology on mammography
| Entity | Features | BI-RADS | Workup |
|---|---|---|---|
| Sebaceous/epidermal cyst | Round/oval; may have lucent center; skin surface origin | 2 | Tangential view to confirm skin origin |
| Neurofibromatosis lesion | Multiple skin nodules projecting over breast | 2 | Clinical history; mark with BB |
| Scar | Architectural distortion at prior surgery site; stable | 2 | Compare to prior; stable = benign |
| Skin thickening (diffuse) | >3 mm; unilateral or bilateral | 0–4 | Unilateral new: consider inflammatory CA; bilateral: CHF, lymphedema |
| Skin retraction/dimpling | Puckering at skin surface | 4–5 SUSPICIOUS | Workup for underlying malignancy |
| Nipple retraction (new) | Inverted nipple, new | 4 SUSPICIOUS | US of subareolar region; biopsy if mass found |
| Paget disease | Nipple/areola skin changes; associated DCIS | 4–5 | Skin punch biopsy; MRI for extent |
| Finding | Description | BI-RADS | Notes |
|---|---|---|---|
| Simple ductal ectasia | Tubular subareolar lucency or density; bilateral; rod-like calcifications | 2 | Benign — plasma cell mastitis pattern |
| Unilateral duct dilation | Single dilated duct; new or symptomatic | 0–3 | US to evaluate for intraductal mass |
| Duct with intraductal mass | Filling defect in dilated duct | 4 SUSPICIOUS | Galactography or MRI; biopsy |
| Bloody nipple discharge + duct dilation | High clinical concern | 4 SUSPICIOUS | US, galactography, or MRI; surgical consult |
Ultrasound — Tissue Composition
Background echotexture and its effect on lesion conspicuity
| Type | Description | Clinical Relevance |
|---|---|---|
| Homogeneous — fat | Uniformly echogenic; fatty tissue predominates | Excellent conspicuity; lesions easy to detect FAVORABLE |
| Homogeneous — fibroglandular | Uniformly echogenic with fibroglandular tissue | Good conspicuity for hypoechoic masses |
| Heterogeneous | Mixed echogenicity; uneven background | Reduced conspicuity; may obscure small lesions LIMIT |
| Consideration | Details |
|---|---|
| Echogenicity reference | Subcutaneous fat is the standard reference for echogenicity comparisons |
| Skin thickness | Normal: 0.5–2 mm; >3 mm = skin thickening (reportable) |
| Cooper's ligaments | Thin echogenic lines; thickening/distortion = associated feature of malignancy |
| Chest wall layers | Ribs (acoustic shadow), intercostal muscles, pleura visible — evaluate for chest wall involvement |
Ultrasound — Non-Mass Lesions
Findings that do not qualify as discrete masses
| Finding | Description | BI-RADS | Notes |
|---|---|---|---|
| Focal echotexture change | Area of altered echogenicity without discrete mass; no posterior features | 3–4 | New or symptomatic = biopsy; stable = follow-up |
| Architectural distortion | Distortion of normal tissue planes; radiating lines; no central mass | 4 SUSPICIOUS | Correlate with mammography; biopsy |
| Ductal changes | Dilated or abnormal-appearing ducts (intraductal solid component) | 4 SUSPICIOUS | Evaluate with Doppler; galactography or MRI |
| Skin thickening | Diffuse or focal skin >3 mm | Variable | Unilateral new: rule out inflammatory carcinoma |
| Edema | Increased echogenicity of fat; skin thickening; Lymphedematous change | Variable | Infection vs. malignancy vs. systemic cause |
| Pre-pectoral collection | Fluid anterior to pectoralis; post-procedure | Variable | Hematoma vs. seroma vs. abscess |
| Finding | Significance | Action |
|---|---|---|
| Normal duct | <2 mm; anechoic; subareolar; collapses with pressure | BI-RADS 1/2 |
| Dilated duct ≥3 mm | Duct ectasia; alone = may be benign | Evaluate for intraductal mass |
| Intraductal solid mass | Papilloma, DCIS, or invasive cancer SUSPICIOUS | BI-RADS 4; biopsy |
| Branching duct with solid component | Highly suspicious for DCIS/papillary carcinoma SUSPICIOUS | BI-RADS 4C–5; biopsy + MRI |
Ultrasound — Calcifications
US detection and characterization of breast calcifications
| Type | US Appearance | Significance |
|---|---|---|
| Macrocalcifications | Echogenic foci with posterior acoustic shadowing; >0.5 mm | Usually benign (fibroadenoma, vessel); correlate with mammography |
| Microcalcifications in mass | Punctate echogenic foci within solid mass; may shadow | Increases suspicion for malignancy SUSPICIOUS |
| Clustered microcalcifications | Group of punctate echogenic foci; may correspond to grouped mammo calcs | BI-RADS 4 if no mass; biopsy if no mammographic correlation |
| Calcifications in cyst wall | Echogenic rim; posterior shadow | Benign (dystrophic) if classic circumscribed cyst BENIGN |
| Milk of calcium | Echogenic layer with posterior shadowing; moves with position change | BI-RADS 2 — benign BENIGN |
| Calcifications in DCIS area | Ill-defined echogenic region ± calcifications; no discrete mass | BI-RADS 4; MRI or biopsy guided by mammography findings |
| Scenario | Approach |
|---|---|
| Mammo calcifications with no US correlate | Stereotactic biopsy if suspicious morphology (not US-guided) |
| Mammo calcifications + US mass | US-guided biopsy with calcium confirmation (specimen radiograph) |
| US calcifications + normal mammogram | Reassign based on mammographic morphology; avoid overdiagnosis |
| Post-biopsy clip calcification | BI-RADS 2 — benign procedure change |
Ultrasound — Associated Features
Secondary findings that modify the assessment of the primary lesion
| Feature | Description | Significance |
|---|---|---|
| Architectural distortion | Disruption of normal tissue planes adjacent to lesion | Highly suspicious SUSPICIOUS |
| Duct changes | Dilated ducts leading to or from a mass | Raises suspicion for ductal carcinoma SUSPICIOUS |
| Skin thickening | Skin >3 mm overlying lesion; local or diffuse | Suspicious if focal and adjacent to lesion SUSPICIOUS |
| Skin retraction | Puckering or dimpling of skin surface | Suspicious — ligamentous involvement SUSPICIOUS |
| Edema | Increased echogenicity of surrounding fat; fluid in tissue planes | Inflammatory process; may indicate malignancy |
| Vascularity | Internal or peripheral blood flow on Doppler | Internal vascularity in solid mass increases suspicion |
| Elasticity assessment | Tissue stiffness (qualitative or quantitative) | Stiff mass = more suspicious; soft = less suspicious |
| Calcifications in mass | Echogenic foci within the lesion | Increases suspicion if fine; may be benign if coarse SUSPICIOUS |
| Lymph nodes | Regional nodal assessment (axillary, intramammary) | Abnormal morphology → staging implications |
MRI — Lymph Nodes
Axillary, internal mammary, and intramammary nodal assessment
| Feature | Normal | Suspicious |
|---|---|---|
| Shape | Reniform | Round SUSPICIOUS |
| Fatty hilum | Present; T1 bright central hilum | Absent SUSPICIOUS |
| Cortical thickness | <3 mm, uniform | Focal or diffuse >3 mm SUSPICIOUS |
| Enhancement pattern | Rim or hilar pattern | Heterogeneous, diffuse SUSPICIOUS |
| Size | Variable; rely on morphology | Replacement of fatty hilum at any size |
| Finding | Notes |
|---|---|
| Normal internal mammary LN | Small (<5 mm), smooth, along internal mammary vessels; BI-RADS 1 |
| Enlarged internal mammary LN | >1 cm, enhancing, or round — staging implication (N3b) SUSPICIOUS |
| Bilateral internal mammary LN | May be reactive; bilateral symmetry favors benign |
| Associated with medial cancer | Medial tumors have higher rate of internal mammary drainage — document |
MRI — Typically Benign Findings
Entities with well-established benign MRI characteristics
| Entity | MRI Features | BI-RADS | Key Differentiator |
|---|---|---|---|
| Simple cyst | T2 bright, T1 dark; no enhancement; circumscribed | 2 BENIGN | Absolutely no enhancement |
| Fibroadenoma (classic) | T2 bright; oval; circumscribed; homogeneous or dark internal septations; persistent kinetics | 2–3 BENIGN | Dark non-enhancing septations pathognomonic |
| Lymph node (intramammary) | T2 bright hilum; reniform; enhancing cortex | 2 BENIGN | Fatty hilum on T1, classic location |
| Hamartoma | Mixed signal; pseudo-capsule; encapsulated fat | 2 BENIGN | "Breast within a breast" |
| Fat necrosis | T1 bright (fat); T2 variable; rim enhancement possible; oil cyst | 2 BENIGN | History of trauma/surgery; T1 fat signal |
| Post-biopsy scar | Low T1/T2 signal; spiculated architecture; stable or decreasing; non-enhancing | 2 BENIGN | Stable over 18+ months; no new enhancement |
| Vascular malformation | T2 bright tubular/serpentine structures; flow voids | 2 BENIGN | Serpentine vessels; flow voids on spin echo |
| Diabetic mastopathy | T2 hypointense; irregular; no significant enhancement | 3 BENIGN | History of type I diabetes; keloid-like stroma |
| Descriptor | Definition | BI-RADS | Management |
|---|---|---|---|
| Focus | <5 mm enhancing dot; too small to characterize morphology | 2–3 | Single new focus in high-risk patient = 6-month MRI; stable = BI-RADS 2 |
| Multiple bilateral foci | Scattered foci both breasts; symmetric | 2 | Likely BPE or benign — routine follow-up |
| Solitary focus, new, high-risk | Single dot; no morphology characterizable | 3 | 6-month follow-up MRI |
| Focus with kinetics | Any focus with washout or persistent | 3–4A | Kinetics unreliable at <5 mm; rely on morphology/context |
MRI — Breast Implant Assessment
ACR BI-RADS implant lexicon and FDA-cleared protocols
| Type | MRI Signal | Notes |
|---|---|---|
| Silicone — single lumen | T2: bright; silicone seq: bright; T1: dark | Most common; assess for rupture |
| Saline — single lumen | T2: bright; silicone seq: dark (suppressed); T1: dark | Rupture clinically apparent (deflation); no MRI rupture assessment needed |
| Double-lumen (silicone inner, saline outer) | Both compartments visible; assess inner silicone | Complex; may show valve artifact |
| Expander (tissue expander) | Variable; metallic port artifact; saline fill | Port artifact common; limits evaluation |
| Sign | Description | Type | Significance |
|---|---|---|---|
| Linguini sign | Curvilinear low-signal lines within the implant (collapsed shell folded within silicone) | Intracapsular rupture RUPTURE | Pathognomonic for intracapsular rupture; most reliable sign |
| Subcapsular line sign | Thin line of silicone between shell and fibrous capsule | Intracapsular (early) | May precede full shell collapse |
| Keyhole / Teardrop sign | Small amount of silicone herniated through shell tear into capsule space | Intracapsular | Early finding; shell not fully collapsed |
| Extracapsular silicone | Silicone signal outside the fibrous capsule | Extracapsular rupture RUPTURE | May be in lymph nodes (snowstorm on US), axilla, or distant |
| Gel bleed | Tiny amount of silicone permeating through intact shell; not rupture | Normal variant | Expected; not a rupture; subclinical finding |
| Radial folds | Normal infolding of implant shell; low signal lines that contact implant edge | Normal NORMAL | Distinguish from linguini sign: radial folds contact edge; linguini sign = free-floating lines |
| Finding | Description | Notes |
|---|---|---|
| Capsular contracture | Thickened, calcified fibrous capsule; implant deformity on MRI | Baker grade I–IV (clinical); MRI shows capsule thickness and calcification |
| Periprosthetic fluid | Fluid between implant and fibrous capsule | Small amount = normal; large amount = seroma; ALCL concern if late-onset |
| BIA-ALCL (ALCL) | Breast implant-associated anaplastic large cell lymphoma; late seroma (often >1 year post-implant) | Textured implants highest risk; late periprosthetic fluid + mass = biopsy urgently |
| Implant position | Subglandular vs. submuscular vs. dual-plane | Document; affects coverage and mammographic appearance |
| Axillary silicone | Silicone signal in axillary lymph nodes | Extracapsular rupture with nodal migration; may mimic adenopathy on mammo/US |
Contrast-Enhanced Mammography (CEM)
Dual-energy technique combining morphologic and functional breast imaging
| Parameter | Detail |
|---|---|
| Contrast agent | Iodinated IV contrast (standard mammographic dose); 1.5 mL/kg at 2–3 mL/sec |
| Timing | Images acquired 2–10 minutes post-injection (peak enhancement window) |
| Energy levels | Low energy (~26–32 kVp, like standard mammo) + high energy (~45–50 kVp, above iodine K-edge at 33.2 keV) |
| Output images | Low-energy (standard mammo appearance) + recombined (subtraction) image showing iodine enhancement |
| Radiation dose | ~1.2–2× standard 2D mammography |
| Views | Standard CC + MLO (minimum); additional views as needed |
- No MRI contraindications (pacemaker, claustrophobia)
- Lower cost and wider availability
- Shorter exam time (~10–15 min)
- Better calcification assessment (low-energy image)
- Familiar mammographic anatomy for radiologists
- No BPE equivalent — background enhancement less problematic
- Iodine contrast required (nephrotoxicity, allergy risk)
- 2D only — no 3D spatial information
- No kinetic curve analysis
- Limited chest wall evaluation
- Less sensitive than MRI for DCIS
- No implant assessment capability
| Parameter | CEM | MRI |
|---|---|---|
| Sensitivity (invasive CA) | ~90–93% | ~90–95% |
| Sensitivity (DCIS) | ~75–80% | ~80–90% |
| Specificity | ~85–90% | ~72–85% |
| Background enhancement equivalent | Background iodine uptake (minimal interference) | BPE (can significantly limit interpretation) |
| Kinetics | Single time-point only | Multi-phase kinetic curves |
| Descriptor | Options |
|---|---|
| Background enhancement | Minimal / Mild / Moderate / Marked (same as MRI BPE) |
| Enhancement morphology (mass) | Shape (round/oval/irregular) + margin (circumscribed/not circumscribed) |
| Enhancement morphology (non-mass) | Distribution: focal / linear / segmental / regional / multiple regions / diffuse |
| Internal pattern (non-mass) | Homogeneous / Heterogeneous / Clumped / Clustered ring |
| Enhancement intensity | Low / Medium / High — relative to background tissue |
| Indication | Evidence |
|---|---|
| Supplemental screening (dense breasts, intermediate risk) | ACRIN 6688: CDR comparable to MRI; fewer recalls than abbreviated MRI in some series |
| Problem-solving (equivocal mammography/US) | Can upgrade or downgrade BI-RADS 3–4 findings |
| Preoperative staging (MRI contraindicated) | Comparable to MRI for lesion extent; accepted ACR alternative |
| Neoadjuvant chemotherapy response | Enhancement reduction correlates with pathologic response; emerging role |
| Post-treatment surveillance (MRI contraindicated) | Acceptable alternative when MRI not possible |
General BI-RADS FAQ
Frequently asked questions — practical clinical guidance
- Initial exam: BI-RADS 3 assigned
- 6 months: Ipsilateral targeted follow-up
- 12 months: Bilateral study (full mammogram or targeted US)
- 24 months: Bilateral study
- If stable at 24–36 months → downgrade to BI-RADS 2
- Concordant benign: Imaging features are consistent with the benign pathology result → routine follow-up per BI-RADS 3 or 6-month imaging
- Concordant malignant: Pathology confirms malignancy → proceed to treatment
- Discordant: Pathology does not explain the imaging (e.g., benign fat necrosis but the lesion was irregular and spiculated) → repeat biopsy or surgical excision
- PPV for malignancy: ~12–15% (range 6–27% in published series)
- Default assessment: BI-RADS 4A (suspicious, low) → biopsy recommended
- If workup (spot compression, US) shows it resolves → may downgrade to BI-RADS 1/2
- On DBT: many "asymmetries" seen on 2D resolve on tomosynthesis (summation artifact) — this is a major advantage of DBT
- Category A or B (not dense): "Your breast tissue is not dense. This is not unusual."
- Category C or D (dense): "Your breast tissue is dense. Dense breast tissue is common and is not abnormal. However, dense breast tissue can make it harder to detect cancer. It also appears to be associated with an increased risk of breast cancer. This information about the results of your mammogram is given to you to raise your awareness. Use this information to talk with your health care provider about your own risks for breast cancer."
- BI-RADS 3: <2% → surveillance, NOT biopsy
- BI-RADS 4A: 2–10% → biopsy; PPV in this range still relatively low
- BI-RADS 4B: 10–50% → biopsy; moderate suspicion
- BI-RADS 4C: 50–95% → biopsy; high suspicion
- BI-RADS 5: ≥95% → biopsy + initiate workup; treat as malignant until proven otherwise
- Architectural distortion — most commonly missed; subtle in dense or overlapping tissue; DBT dramatically improves detection
- Developing asymmetry — requires comparison with priors; often subtle one-view finding
- Masses obscured by density — especially in heterogeneous (C) or extremely dense (D) breasts
- Calcifications — fine linear/branching (DCIS) can be subtle; amorphous calcs easy to dismiss
- Contralateral breast neglect — attention focused on known abnormality; contralateral cancer missed
Radiology-Pathology Correlation
Concordance assessment after image-guided breast biopsy — ASBrS 2024 / ACR
Concordance vs. Discordance
| Finding | Definition | Required Action |
|---|---|---|
| Concordant — Benign | Pathology adequately explains the imaging finding (e.g., fibroadenoma for circumscribed oval mass; fibrocystic change for calcifications) | Routine follow-up per BI-RADS recommendation; imaging at 6, 12, 24 months if BI-RADS 3 |
| Concordant — High Risk | Pathology (e.g., ADH, LCIS, radial scar) explains the imaging and is an expected result for the lesion type and appearance | Management per high-risk lesion guideline (see High Risk Lesions tab); multidisciplinary discussion |
| Discordant | Pathology does not adequately explain the imaging finding (e.g., benign fibrocystic change for a spiculated mass; normal breast tissue for calcifications that were targeted) | Repeat biopsy with larger gauge or VAB, or surgical excision. Do not observe discordant results. |
| Technical Failure | No lesional tissue obtained; calcifications absent on specimen radiograph; post-biopsy clip not in lesion on post-procedure imaging | Repeat biopsy required; confirm targeting with specimen radiograph for calcifications |
Concordance Examples by Imaging Finding
| Imaging Finding | Concordant Pathology | Discordant — Consider Re-biopsy / Excision |
|---|---|---|
| Circumscribed oval/round mass | Fibroadenoma, cyst, lymph node, lipoma, hamartoma | Normal breast tissue only, fat necrosis without clinical history |
| Irregular / spiculated mass | Carcinoma, radial scar, fat necrosis (post-surgical), sclerosing adenosis | Fibrocystic change, normal breast tissue, benign stroma only |
| Grouped fine pleomorphic calcifications | DCIS, ADH, FEA, sclerosing adenosis with calcifications, fibrocystic change with calcifications | Normal breast tissue, fibroadenoma without calcifications, stromal tissue only |
| Segmental calcifications | DCIS, lobular neoplasia, extensive FEA | Focal fibrocystic change, benign findings without calcifications in specimen |
| Non-mass enhancement (MRI) | DCIS, invasive carcinoma, FEA, adenosis, fibrocystic change | Normal fibroglandular tissue only, stroma without lesional tissue |
| Architectural distortion | Invasive carcinoma, radial scar, post-surgical scarring, sclerosing adenosis | Normal breast tissue, fibrocystic change without sclerosing features |
Concordance Assessment Checklist
- Document lesion targeted and biopsy site confirmation (clip placement, post-procedure imaging)
- Confirm calcifications present on specimen radiograph (if targeted by calcifications)
- Assess whether pathology explains the imaging morphology, BI-RADS category, and location
- Communicate result and concordance assessment to ordering clinician
- Document recommendation (follow-up interval, re-biopsy, or excision) in report
- Discordant imaging-pathology: always
- No calcifications on specimen radiograph despite calcification target
- Technically inadequate sample (insufficient tissue, post-biopsy clip migration)
- High-risk lesion with additional concerning features (see High Risk Lesions tab)
- Patient preference for definitive diagnosis over surveillance
The biopsy report should explicitly state: (1) lesion targeted, (2) biopsy technique and gauge, (3) pathology result, (4) concordance assessment (concordant vs. discordant), and (5) management recommendation. Diagnostic imaging at 6, 12, and 24 months is standard follow-up when concordant benign or high-risk surveillance is chosen.
Radiology-Pathology Correlation — High-Risk Lesions
Surgical management recommendations — ASBrS 2024 Resource Guide + NCCN/SSO
| Lesion | Upgrade Rate (CNB) | Upgrade Rate (VAB) | ASBrS 2024 Recommendation | Exceptions / Key Factors | Source |
|---|---|---|---|---|---|
| Atypical Ductal Hyperplasia (ADH) | ~29% pooled (meta-analysis) | 5–15% | Surgical excision; low-risk criteria → observation acceptable | Lower risk: mammographic calcifications (not mass), small lesion, complete removal (≥50–95% of calc), small ADH volume, no other high-risk lesions. Low-risk cohort: 2% upgrade, 4.4% cancer at 5 yr without surgery. | ASBrS 2024; NCCN 2024 |
| Classic LCIS / ALH | 0–4% | 0–3% | No excision; observation with clinical and imaging follow-up | Excision if: discordant imaging; co-existing high-risk lesion (LN + ADH/non-classic LCIS → ≥25% upgrade); MRI-guided biopsy (higher upgrade rate). 1–2% annual breast cancer risk persists regardless of excision. | ASBrS 2024; ACR |
| Pleomorphic LCIS (PLCIS) | 25–60% | — | Surgical excision to negative margins (2mm recommended) | ER-negative, HER-2 positive, comedonecrosis, microcalcifications similar to DCIS. Recurrence at 2mm: 26.3%; at <1mm: 36.4%. Similar to DCIS margins guideline. | ASBrS 2024; SSO/ASTRO/ASCO |
| Florid LCIS (FLCIS) | 30–40% | — | Surgical excision to negative margins (2mm recommended) | Recognized by WHO 2019 as distinct variant. Mass-forming distention of TDLU acini; do not confuse with extensive CLCIS (involvement of multiple ducts without acinar expansion). | ASBrS 2024 |
| Columnar Cell Lesion (CCL) without atypia | <2% | <1% | No excision; return to screening | CCC and CCH without atypia. Excision not recommended if concordant. | ASBrS 2024 |
| Flat Epithelial Atypia (FEA) — pure, isolated | ~5% pooled (2021 meta-analysis, n=2484) | 0–3% | No excision; observation with clinical and imaging follow-up | Excision if: extensive residual calcifications or inadequately sampled. If ≥90% of calcifications removed → 0% upgrade. ADH found in 17% of excision specimens (impacts management). Observation reasonable if majority of target removed. | ASBrS 2024 |
| Papilloma without atypia | 1–5% (asymptomatic); slightly higher if symptomatic | 0–2% | No excision; observation with clinical and imaging follow-up | Consider excision if: symptomatic (palpable mass, nipple discharge); size >1cm; age >50; peripheral location; >50% residual lesion after CNB. Prospective multicenter data: 1.7% upgrade to DCIS; no invasive cancers in BI-RADS ≤4 asymptomatic papillomas (n=116). | ASBrS 2024; SSO |
| Papilloma with atypia | 20–30% | 10–20% | Surgical excision | Atypical papillary lesions upgraded at excision up to 20–30%. Surgical excision recommended regardless of location. | ASBrS 2024; ACR/SSO |
| Complex Sclerosing Lesion (CSL) without atypia | 1–5% (8–16G CNB) | ~1% (2019 meta-analysis, n=3163) | No excision; observation if concordant | Excision if: atypia present (→ see below); not adequately sampled; other concerning features. Incidental microscopic CSL or <5mm → no upgrades reported. Enhancement on MRI does not predict need for excision. | ASBrS 2024; SSO 2019 |
| Complex Sclerosing Lesion (CSL) with atypia | Up to 35% | — | Surgical excision | Routine excision recommended when atypia is present on CNB. | ASBrS 2024 |
| Mucocele-like Lesion (MLL) without atypia | 0–4% | 0–2% | No excision; observation if concordant | Routine surgical excision not recommended for concordant MLL without atypia. All studies are small/retrospective; careful concordance essential. | ASBrS 2024 |
| Mucocele-like Lesion (MLL) with atypia | Up to 31% | — | Surgical excision | Surgical excision recommended; upgrade to invasive cancer up to 31%. | ASBrS 2024 |
| Desmoid Tumor / Fibromatosis | N/A (benign; not malignant upgrade) | N/A | Observation; imaging every 3–6 months (mammogram, MRI, or CT as clinically indicated) | Excision if symptomatic or interval growth (aim for R0 resection). Associated with FAP/Gardner syndrome. Recurrence common with incomplete resection. Surgery deferred given variable behavior. | ASBrS 2024 |
| PASH (Pseudoangiomatous Stromal Hyperplasia) | N/A (benign; not malignant upgrade) | N/A | Clinical observation; no excision if concordant | Consider excision if symptomatic (palpable mass, enlarging). Myofibroblast proliferation mimicking vascular lesion. Recurrence after excision described. | ASBrS 2024 |
| Phyllodes (borderline/malignant) | N/A — requires excision | N/A | Wide local excision (≥1cm margins for malignant; negative for borderline) | Stromal overgrowth, increased mitoses, infiltrative border. Cellular fibroepithelial lesion on CNB → excision to exclude phyllodes (10–20% are phyllodes at excision). | SSO; NCCN 2024 |
- CNB = core needle biopsy (typically 14G spring-loaded or 9–11G VAB). VAB achieves more complete lesion sampling and consistently lower upgrade rates.
- Upgrade = finding of DCIS or invasive carcinoma at surgical excision after a high-risk CNB diagnosis.
- VAE (vacuum-assisted excision) using 7–11G is an emerging alternative to surgical excision for select lesions; European 2024 guidelines suggest VAE for lesions ≤15mm — not yet standard in the U.S.
- Discordance always mandates repeat biopsy (larger gauge or VAB) or surgical excision regardless of lesion type.
- All patients with high-risk lesions should undergo comprehensive breast cancer risk assessment and be considered for risk-reducing medication and high-risk screening (NCCN). High-risk lesion diagnosis is not itself an indication for genetic testing, but personal/family history should be evaluated.
- All high-risk lesions should be presented at multidisciplinary tumor board or high-risk conference.
Recommendations assume radiologic-pathologic concordance has been established. Follow-up imaging at 6, 12, and 24 months (ACR guidelines) is standard when surveillance is chosen. Strongly consider excision if lesion progresses during follow-up. Multidisciplinary input from breast radiologist, breast surgeon, and breast pathologist improves upgrade risk estimates.
Ask BI-RADS AI
Powered by Claude — ACR BI-RADS 2025 knowledge
Gynecomastia Case ↗
Imaging approach, mammographic patterns, and causes — Mannix et al. RadioGraphics 2024
| Feature | Details |
|---|---|
| Definition | Benign proliferation of glandular breast tissue in males; most common male breast condition. Bilateral in up to 50%. |
| Clinical Presentation | Palpable subareolar mass or tenderness; may be unilateral or bilateral. Usually distinguished from pseudogynecomastia (fatty enlargement without glandular tissue). |
| Age-Based Imaging Triage |
Age ≥ 25 yr: bilateral diagnostic mammography first (two views per side); US as adjunct if mammography inconclusive or mass is focal/eccentric Age < 25 yr: ultrasound first (lower radiation concern, denser tissue); mammography added if US inconclusive or findings atypical for gynecomastia |
| US Role | Reserve for focal/eccentric masses, inconclusive mammography, or guiding biopsy. ⚠ Avoid routine US of classic mammographic gynecomastia — the subareolar glandular tissue appears as an irregular, hypoechoic, angulated mass on US and can closely mimic malignancy, adding confusion rather than clarity when the mammographic pattern is already diagnostic. |
| MRI Indications | Not routine. Reserved for: problem-solving after equivocal mammo/US, staging known MBC, high-risk surveillance (BRCA1/2 carriers), or implant evaluation. |
| Pattern | Mammographic Appearance | Clinical Correlation | Management |
|---|---|---|---|
| Nodular (Florid) | Fan-shaped or flame-shaped subareolar density with ill-defined posterior border; radiates from nipple | Active, early phase; elevated estrogen or androgen stimulation; often tender | BI-RADS 2; clinical correlation; address underlying cause |
| Dendritic (Fibrous) | Irregular spiculated density extending into fat; prominent fibrous stroma; may mimic malignancy | Late/chronic phase; fibrosis predominates; usually painless and longstanding | US correlation to confirm benign; biopsy if atypical features (eccentric, mass-forming) |
| Diffuse Glandular | Extensive bilateral glandular tissue mimicking female breast; often symmetric | Estrogen excess (cirrhosis, exogenous hormones, testicular tumors); Klinefelter syndrome | BI-RADS 2; address underlying hormonal cause |
| Category | Examples |
|---|---|
| Physiologic | Neonatal (maternal estrogen), pubertal (transient, resolves 6–18 mo), senescent (>65 yr; ↓ testosterone) |
| Hypogonadism | Klinefelter syndrome (47,XXY), orchitis, cryptorchidism, castration, Kallmann syndrome, androgen insensitivity |
| Increased Estrogen Production | Testicular tumors (Leydig cell, Sertoli cell, germ cell), adrenal tumors, obesity (peripheral aromatization of androgens to estrogens), congenital adrenal hyperplasia |
| Liver Disease | Cirrhosis → ↑ sex hormone-binding globulin (SHBG) → ↑ estrogen; also ↓ hepatic androgen metabolism |
| Renal Disease | Chronic renal failure, hemodialysis → ↑ LH → ↑ aromatization |
| Thyroid Disease | Hyperthyroidism → ↑ SHBG, ↑ peripheral aromatization |
| Malnutrition / Refeeding | Refeeding after starvation: rapid ↑ in insulin/gonadotropins → transient estrogen surge |
| Drugs | See drug table below |
| Idiopathic | ~25% of cases; no identifiable cause after full workup |
| Category | Drugs | Mechanism | Evidence Level |
|---|---|---|---|
| Well-Established (Definitive Evidence) | |||
| Antiandrogens | Bicalutamide, flutamide, enzalutamide, abiraterone, cyproterone acetate | Block androgen receptor or ↓ androgen synthesis → unopposed estrogen | Definitive; dose-dependent; up to 70% incidence with bicalutamide monotherapy |
| Estrogens / Anabolic Steroids | Exogenous estrogens (HRT, DES), anabolic-androgenic steroids, testosterone supplementation | Direct estrogenic effect or peripheral aromatization | Definitive |
| Digoxin | Digoxin | Structural similarity to estrogen; binds estrogen receptor | Definitive; classic association |
| Spironolactone | Spironolactone | Anti-androgen (androgen receptor antagonist) + weak estrogenic activity | Definitive; dose-dependent |
| Ketoconazole | Ketoconazole | Inhibits testicular and adrenal androgen synthesis (CYP17) | Definitive |
| Metronidazole | Metronidazole | Weak estrogenic activity | Definitive |
| Probable Evidence | |||
| H2-Blockers | Cimetidine (strongest), ranitidine | Androgen receptor antagonism; ↑ prolactin | Probable; cimetidine strongest association; PPIs much lower risk |
| Calcium Channel Blockers | Verapamil, diltiazem, amlodipine, nifedipine | ↑ prolactin; unclear mechanism | Probable; case reports and series |
| ACE Inhibitors | Enalapril, captopril | Mechanism unclear | Probable; case reports |
| Antipsychotics | Haloperidol, risperidone, quetiapine, olanzapine | ↑ prolactin (dopamine antagonism) → ↓ testosterone; ↑ estrogen | Probable; risperidone highest risk among atypicals |
| Tricyclic Antidepressants | Amitriptyline, imipramine | ↑ prolactin | Probable |
| Opioids | Heroin, methadone, chronic opioid analgesics | ↓ hypothalamic GnRH → ↓ LH/FSH → ↓ testosterone | Probable; particularly with chronic use |
| GnRH Agonists | Leuprolide, goserelin | Initial flare then testosterone suppression → ↑ estrogen ratio | Probable; common in prostate cancer treatment |
| Possible / Uncertain Evidence | |||
| Proton Pump Inhibitors | Omeprazole, lansoprazole | Possible weak estrogen-like or anti-androgen activity | Possible; low risk |
| Statins | Atorvastatin, simvastatin | ↓ cholesterol (androgen precursor) → possible ↓ androgen synthesis | Possible; low risk; case reports only |
| Chemotherapy | Alkylating agents (cyclophosphamide), methotrexate, vinca alkaloids | Gonadotoxicity → ↓ testosterone | Possible; primary hypogonadism mechanism |
| Antiretrovirals | HAART regimens (efavirenz, stavudine) | ↑ estrogen, altered metabolism | Possible; multifactorial |
| Amphetamines / MDMA | Amphetamines, MDMA | Unclear; possible estrogen-like effects | Possible |
Treatment note: Address underlying cause first. Medical therapy (tamoxifen 10–20 mg/day or raloxifene) effective in symptomatic florid gynecomastia. Surgical excision (liposuction ± subcutaneous mastectomy) for longstanding fibrous/dendritic gynecomastia or cosmetic concern. Radiation prophylaxis used in prostate cancer patients starting antiandrogen therapy.
Male Breast Cancer & Benign Entities Case ↗
Distinguishing gynecomastia from malignancy; MBC overview; benign masses; high-risk screening
| Feature | Gynecomastia | Male Breast Cancer |
|---|---|---|
| Location | Subareolar, centered behind nipple; bilateral in ~50% | Eccentric — not centered on nipple; almost always unilateral |
| Mammographic shape | Fan-shaped / flame-shaped subareolar density (nodular pattern) or irregular fibrous density (dendritic) — posterior border often ill-defined but lacks true spiculation from a discrete mass | Irregular or oval mass; spiculated margins common; may have associated microcalcifications |
| Calcifications | Absent or benign-type | Suspicious microcalcifications (pleomorphic, linear branching) in ~30% |
| Skin / nipple changes | Usually absent | Nipple retraction, skin thickening, ulceration — important red flags |
| Axillary adenopathy | Absent | Present in ~40–50% at diagnosis |
| US appearance | Hypoechoic subareolar tissue, fan-shaped, no posterior acoustic shadowing or internal vascularity | Irregular hypoechoic mass with angular/spiculated margins, posterior shadowing, internal vascularity on Doppler |
| BI-RADS | BI-RADS 1 or 2 (classic nodular/dendritic pattern); BI-RADS 0 if atypical | BI-RADS 4 or 5; tissue sampling required |
| Feature | Details |
|---|---|
| Incidence | <1% of all breast cancers; ~2,800 cases/yr in US. Median age at diagnosis: ~67 yr (older than female BC). |
| Histology | Invasive ductal carcinoma (IDC) ≥ 90%. Invasive lobular carcinoma rare (<2%; males lack lobular units). DCIS in ~10%. |
| Receptor Profile | ER+ in ~90%, PR+ in ~80%, HER2+ in ~10%. Luminal A subtype predominates. Triple-negative MBC is rare (~3%). |
| Presentation | Painless eccentric palpable mass (most common); nipple discharge, retraction, or ulceration; skin changes. Often presents at later stage due to delayed recognition and lower clinical suspicion. |
| Prognosis | Stage-for-stage similar to female BC; overall slightly worse due to older age at presentation and higher stage at diagnosis. 5-yr survival: stage I ~100%, stage II ~85%, stage III ~65%, stage IV ~25%. |
| Treatment | Surgery (modified radical mastectomy or breast-conserving surgery), adjuvant chemotherapy/radiation per oncology guidelines; endocrine therapy (tamoxifen first-line for ER+ disease; aromatase inhibitors less effective in males due to incomplete estrogen suppression). |
| Risk Factor | Notes |
|---|---|
| BRCA2 mutation | Strongest genetic risk; lifetime risk ~6–8% (vs ~0.1% baseline). MBC is a BRCA2 sentinel malignancy. BRCA2 accounts for ~10–15% of MBC. |
| BRCA1 mutation | Modest increased risk (~1–2% lifetime); much less than BRCA2 for males. |
| Klinefelter syndrome (47,XXY) | 20–50× increased risk due to hyperestrogenism and hypogonadism. |
| Family history | First-degree female relative with BC; bilateral female BC; Jewish ancestry (BRCA founder mutations). |
| Radiation exposure | Prior chest wall radiation (e.g., Hodgkin lymphoma treatment). |
| Hyperestrogenism | Obesity (peripheral aromatization), cirrhosis, exogenous estrogen, testicular disease. |
| Other genes | PALB2, CHEK2, ATM mutations associated with moderately elevated risk. |
| Entity | Imaging Features | Notes |
|---|---|---|
| Epidermal Inclusion Cyst | Well-circumscribed anechoic to complex cystic mass on US; may have calcified wall; may be palpable | Most common benign palpable lesion in males. Subareolar or skin-related. BI-RADS 2 if classic; aspiration if symptomatic. |
| Lipoma | Well-circumscribed, compressible, isoechoic to fat on US; isoattenuating to fat on mammo | Common; BI-RADS 2. Angiolipoma may be tender. |
| Abscess | Complex cystic/solid mass with posterior acoustic enhancement, perilesional edema on US; rim enhancement on MRI | Associated with skin changes, erythema, tenderness; may have sinus tract. Aspiration + antibiotics; imaging-guided drainage if large. |
| Hemangioma | Heterogeneous or lobulated mass; internal vascularity on Doppler; T2 bright on MRI | Rare; biopsy for definitive diagnosis if atypical. Cavernous hemangioma most common type. |
| Myofibroblastoma | Well-circumscribed oval mass; homogeneously hyperechoic on US; rare mammo calcifications | Benign spindle cell tumor; equal male-female incidence. BI-RADS 3–4A; core biopsy confirms diagnosis. Wide local excision curative. |
| Pseudoangiomatous Stromal Hyperplasia (PASH) | Non-specific; well-circumscribed mass on US; may be invisible on mammo; BI-RADS 3–4A | Rare in males; typically incidental finding on core biopsy. No malignant potential; excision if enlarging or symptomatic. |
| Papilloma | Intraductal mass on US; dilated duct; may cause nipple discharge | Rare in males. Benign but may harbor atypia on pathology; excision recommended for definitive diagnosis. |
| Fibroadenoma | Well-circumscribed oval mass; parallel orientation; hyperechoic on US | Very rare in males (requires functioning lobular tissue). Usually in males with longstanding gynecomastia or Klinefelter syndrome. |
| Population | Recommendation | Source |
|---|---|---|
| BRCA2 carriers | Annual clinical breast exam starting age 35; consider annual mammography starting age 40 (NCCN); MRI if mammographically dense or strong family history | NCCN 2024; ASCO 2016 |
| BRCA1 carriers | Annual clinical breast exam; consider mammography if significant family history of MBC; individualize based on risk | NCCN 2024 |
| Klinefelter syndrome | Annual clinical breast exam; consider baseline mammography; MRI if gynecomastia obscures mammographic assessment | ASCO 2016; expert consensus |
| Prior chest radiation | Annual mammography ± MRI starting 8–10 yr after radiation or at age 25 (whichever is later), per female BC screening protocols for radiation-exposed patients | ACS 2023; NCCN 2024 |
| General male population | No routine screening recommended; evaluate symptomatic males promptly | ACR; USPSTF |
Breast Cancer Screening — Guidelines Summary
Risk-stratified recommendations from major guideline bodies (2024–2025)
| Risk Category | Age to Start | Modality | Frequency | Source |
|---|---|---|---|---|
| Average risk | 40 | Mammography ± tomosynthesis | Annual (ACR/SBI) or Biennial (USPSTF 2024) | ACR 2023, USPSTF 2024 |
| BRCA1/2 | 25–30 | Annual MRI; add mammography at 30 | Annual MRI + annual mammo (alternating q6mo per NCCN) | NCCN 2025 |
| TP53 (Li-Fraumeni) | 20 | Annual MRI (preferred over mammo due to radiation risk) | Annual | NCCN 2025 |
| PTEN (Cowden) | 30 | Annual MRI + mammography | Annual | NCCN 2025 |
| PALB2/CHEK2/ATM | 30–35 | Annual MRI + mammography | Annual | ACR 2023, NCCN 2025 |
| Lifetime risk ≥20% (model-based) | 25–40 (no earlier than 25) | Annual MRI + mammo (mammo no earlier than 30) | Annual | NCCN 2025, ACR 2023 |
| Prior chest RT ≥10 Gy, age 10–30 | 25 or 8 yrs post-RT | Annual MRI + mammography | Annual | ACR 2023, NCCN 2025 |
| Personal Hx breast CA, dx <50 | At diagnosis | Annual MRI + mammography | Annual | ACR 2023 |
| Extremely dense (C4/D) | 50 (consider 40) | Annual MRI after negative mammo | Annual | NCCN 2025 |
| Heterogeneously dense (C3) | 40 | Mammography; consider supplemental | Annual + shared decision | NCCN 2025, ACR 2023 |
Average vs. High Risk — Definitions and Workup
Risk stratification determines screening modality, starting age, and interval
- No first-degree relatives with breast cancer
- No prior breast biopsy showing high-risk lesion
- Lifetime risk <15% by validated model
- No prior chest radiation
- No known genetic mutation (BRCA1/2, TP53, PTEN, PALB2, CHEK2, ATM, CDH1, STK11)
| Criterion | Details |
|---|---|
| BRCA1/2 PV | Carrier or untested 1st-degree relative of carrier |
| TP53/PTEN/STK11/CDH1 PV | Pathogenic variants — annual MRI from age 20–30 depending on syndrome |
| PALB2/CHEK2/ATM | Moderate-risk genes; MRI from 30–35; updated NCCN 2025 includes these |
| Model-based ≥20% | BRCAPRO, Tyrer-Cuzick, BOADICEA, CanRisk, BCSC — must use validated model |
| Prior chest RT ≥10 Gy | Hodgkin lymphoma or similar; radiation received before age 30 |
| Li-Fraumeni / Cowden / BRR syndrome | Syndrome diagnosis in patient or first-degree relative |
- May benefit from supplemental screening — individualized decision based on patient preference and access
- Consider annual mammography + shared decision regarding MRI
- Referral to high-risk program or genetic counseling recommended
- No definitive guideline recommendation for supplemental MRI at this threshold
| Lesion | MRI Recommendation |
|---|---|
| ADH or LCIS with ≥20% lifetime risk | Annual MRI — NCCN/ACR recommend strongly |
| ADH or LCIS alone | Consider annual MRI, especially with other risk factors; individualized |
| ALH with ≥20% lifetime risk | Annual MRI |
| Radial scar + atypia | Consider MRI; individual assessment based on complete risk profile |
Landmark Screening RCTs
Key randomized controlled trials that established mammography screening evidence
Digital Breast Tomosynthesis (DBT) Trials
Evidence supporting tomosynthesis over 2D mammography for screening
Dense Breast Supplemental Screening Trials
Evidence for and against supplemental screening in women with dense breasts
High-Risk MRI Screening Trials
RCTs and prospective studies establishing MRI for high-risk surveillance
Contrast-Enhanced Mammography (CEM) Trials
Emerging evidence establishing CEM as an MRI alternative
Palpable Breast Lesion — Workup Protocol
Age-stratified imaging algorithm and management — NCCN 2025, ACR Appropriateness Criteria
Age-Stratified Imaging Algorithm
| Age | First-line Imaging | If High Clinical Suspicion | Additional Notes |
|---|---|---|---|
| ≥30 years | Diagnostic mammogram with tomosynthesis + targeted ultrasound | Tissue sampling even if imaging negative | US may be omitted only if mammo shows definitively benign finding at palpable site |
| <30 years | Targeted ultrasound (preferred) | Add diagnostic mammogram; proceed to biopsy | If low suspicion: observe 1–2 cycles, then US if persistent |
| Pregnant / lactating | Targeted ultrasound first | Add mammogram if needed (minimal fetal dose with shielding) | Pump before imaging; CNB safe during lactation (milk fistula rare) |
Triple Test
| Component | Tool | Notes |
|---|---|---|
| Clinical breast exam | Physical examination | High clinical suspicion overrides all imaging findings |
| Imaging | Mammography ± ultrasound | Geographic correlation with palpable site is mandatory |
| Tissue sampling | Core needle biopsy (preferred) | Sensitivity/specificity approach 100% when all three are concordant |
Whole Breast Ultrasound — Evidence Summary
Supplemental screening ultrasound: benefits vs. harms
Performance Metrics — Mammography Alone vs. Mammography + WBUS
| Metric | Mammo Alone | Mammo + WBUS | RR (95% CI) |
|---|---|---|---|
| Cancer detection rate | Baseline | +2–4/1000 | RR 1.14 (0.76–1.68) NS |
| Interval cancers | Baseline | No significant reduction | RR 0.67 (0.33–1.37) NS |
| False-positive biopsy rate | 22.2/1000 | 52.0/1000 | RR 2.23 (1.93–2.58) |
| Short-interval follow-up rate | 1.1% | 3.9% | RR 3.10 (2.60–3.70) |
| PPV (biopsy recommendation) | 21.4% | 9.5% | RR 0.50 (0.35–0.71) |
| False-positive recalls per 1000 | Baseline | +48/1000 | — |
Source: Lee JM et al. JAMA Intern Med. 2019;179(5):658-667 (PROSPR Consortium)
Guideline Comparison — Major Organizations
Side-by-side comparison: USPSTF 2024, ACS 2023, ACR/SBI 2023, NCCN 2025
| Recommendation | USPSTF 2024 | ACS 2023 | ACR/SBI 2023 | NCCN 2025 |
|---|---|---|---|---|
| Average-risk start age | 40 | 45 (optional 40–44) | 40 | 40 |
| Average-risk interval | Biennial | Annual 45–54; biennial or annual 55+ | Annual | Annual |
| Upper age limit | 74 | Until life expectancy <10 yr | Until life expectancy <10 yr | Individualized |
| DBT vs. 2D | Not specified | Not specified | DBT preferred | DBT preferred |
| Dense breast notification | No recommendation | No recommendation | Strongly endorses | Endorsed |
| Supplemental US | Insufficient evidence | No recommendation | MRI preferred; WBUS last resort | WBUS only if CEM/MBI/MRI unavailable |
| High-risk MRI start | Not addressed | Not addressed | ≥20% lifetime: annual MRI + mammo from 30 | ≥20%: annual MRI from 25–30 + mammo from 30 |
| BRCA1/2 | Not addressed | Not addressed | Annual MRI from 25, mammo from 30 | Annual MRI from 25, mammo from 30; alternate q6mo |
Key References
Primary literature cited throughout this clinical trials reference
- 1Shapiro S et al. Periodic screening for breast cancer: the Health Insurance Plan project and its sequelae, 1963–1986. J Natl Cancer Inst. 1988;69(2):349-55. First large RCT demonstrating mammography screening benefit.HIP Trial — foundational screening evidence
- 2Tabár L et al. Swedish two-county trial: impact of mammographic screening on breast cancer mortality during 3 decades. Radiology. 2011;260(3):658-63. 20-year follow-up; 31% mortality reduction.Swedish Two-County — primary evidence for annual screening
- 3Miller AB et al. Twenty-five year follow-up for breast cancer incidence and mortality of the Canadian National Breast Screening Study. Oncology. 2016;30(12):1084-8. Controversial trial showing no mortality benefit; criticized for randomization flaws.Canadian NBSS — widely debated negative trial
- 4Henderson JT et al. Screening for Breast Cancer: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2024;331(22):1931-1946. Meta-analysis supporting USPSTF 2024 recommendation to start screening at 40.USPSTF 2024 — basis for biennial-at-40 recommendation
- 5Pisano ED et al. TMIST trial design. N Engl J Med. 2022. Design paper for the largest breast cancer screening trial; primary results pending.TMIST — definitive DBT vs. 2D RCT; results expected 2030s
- 6Lång K et al. Performance of one-view breast tomosynthesis as a stand-alone breast cancer screening modality. Lancet Oncol. 2016;17(8):1201-8. MBTST — 27% more cancers detected with DBT vs. 2D in Swedish population.MBTST — Swedish DBT superiority trial
- 7Skaane P et al. Comparison of digital mammography alone and digital mammography plus tomosynthesis in a population-based screening program. Radiology. 2013;267(1):47-56. Oslo trial — DBT + synthetic 2D increases invasive cancer detection by 27%.Oslo Tomosynthesis — key paired design DBT trial
- 8Ciatto S et al. Integration of 3D digital mammography with tomosynthesis for population breast-cancer screening. Lancet Oncol. 2013;14(7):583-9. STORM trial — DBT adds 2.4/1000 incremental cancer detection in Italian screening.STORM — Italian multicenter DBT trial
- 9Berg WA et al. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography. JAMA. 2012;307(13):1394-404. ACRIN 6666 — US adds 4.2 cancers/1000 but doubles false-positive biopsy rate.ACRIN 6666 — supplemental US in high-risk women
- 10Bakker MF et al. Supplemental MRI Screening for Women with Extremely Dense Breast Tissue. N Engl J Med. 2019;381(22):2091-102. DENSE trial — supplemental MRI adds 16.5/1000 cancers; interval cancer rate RR 0.33.DENSE Trial — definitive MRI evidence for BI-RADS D breasts
- 11Comstock CE et al. Comparison of Abbreviated Breast MRI vs Digital Breast Tomosynthesis for Breast Cancer Detection Among Women With Dense Breasts Undergoing Screening. JAMA. 2020;323(4):369-377. EA1141 — abbreviated MRI detects 15.2 vs. 6.2/1000 cancers vs. DBT in dense breasts.EA1141 — abbreviated MRI superior to DBT for supplemental screening
- 12Jochelson MS et al. Comparison of Screening CEDM and Breast MRI for Women at Increased Risk for Breast Cancer. Radiology. 2021;298(1):30-37. ACRIN 6688 — CEM established as viable alternative to abbreviated MRI.ACRIN 6688 — CEM vs. DBT for supplemental screening
- 13Lee JM et al. Performance of Screening Ultrasonography as an Adjunct to Screening Mammography. JAMA Intern Med. 2019;179(5):658-667. PROSPR — WBUS doubles false-positive biopsy rate without significant cancer detection benefit.PROSPR — real-world WBUS harms vs. benefits
- 14Saadatmand S et al. FaMRIsc: MRI + mammo sensitivity 84.7% vs. 40% mammo alone in BRCA/high-risk women. Lancet Oncol. 2019;20(8):1136-1147.FaMRIsc — first RCT of MRI in high-risk surveillance
- 15Leach MO et al. Screening with magnetic resonance imaging and mammography of a UK population at high familial risk of breast cancer. Lancet. 2005;365(9473):1769-78. MRISC — MRI sensitivity 71% vs. mammography 37% in BRCA/high-risk cohort.MRISC — Swedish foundational high-risk MRI study
- 16Phi XA et al. Systematic review and meta-analysis of radiological investigations for the detection of breast cancer in women with a familial risk. Eur J Cancer. 2015;51(16):2225-68. Pooled MRI sensitivity ~80%, mammography 16–40% in BRCA carriers.Phi et al. — definitive meta-analysis of MRI in BRCA surveillance
- 17Kuhl CK et al. Abbreviated biparametric prostate MR imaging. J Clin Oncol. 2014;32(22):2304-10. Abbreviated MRI — cancer detection 18.2/1000, equivalent to full protocol; 3-min scan.Kuhl — abbreviated MRI concept validation
- 18Sung JS et al. Breast Cancer Detection Using Combined MR Imaging and Mammography. Radiology. 2019;290(1):44-53. CEM multicenter — sensitivity 93%, AUC 0.87; CEM comparable to abbreviated MRI.Sung — CEM as problem-solving and screening tool
- 19Simmons RM et al. Cryoablation of Breast Cancer. Ann Surg Oncol. 2012;19(4):1179-85. ACOSOG Z1072 — 92.7% complete ablation for ≤1cm tumors; 20% for >1cm.ACOSOG Z1072 — key size threshold for cryoablation feasibility
- 20Manahan ER et al. Ice3 Trial: Interim results. Ann Surg Oncol. 2022. Ice3 — interim 3-year recurrence <3%; feasibility established for definitive cryoablation.Ice3 — definitive cryoablation pilot, no surgery
- 21Fukuma E et al. Cryoablation as a primary treatment for early breast cancer. Breast Cancer. 2021;28(2):473-81. Cryo-FIRST Japan — 3-year local recurrence 5%; OS 100%; MRI complete ablation 87%.Cryo-FIRST — Japanese phase 2 definitive cryoablation trial
- 22Hubbard JL et al. FROST trial: Phase 3 RCT of cryoablation versus lumpectomy. Ann Surg Oncol. 2024; NCT03801369. FROST — only phase 3 RCT comparing cryo vs. surgery; primary results expected ~2028–2030.FROST — pivotal NCI-sponsored randomized trial
- 23NCCN Clinical Practice Guidelines in Oncology: Breast Cancer Screening and Diagnosis. Version 1.2025. Current standard for risk-stratified screening, high-risk MRI, and palpable lesion management.NCCN 2025 — current guideline standard
- 24Monticciolo DL et al. Breast Cancer Screening in Women at Higher-Than-Average Risk. J Am Coll Radiol. 2023;20(7):902-914. ACR 2023 high-risk screening recommendations.ACR 2023 — high-risk and supplemental screening guidelines
- 25US Preventive Services Task Force. Breast Cancer Screening: Final Recommendation Statement. JAMA. 2024;331(22):1918-1930. USPSTF 2024 final recommendation — biennial screening starting at age 40 for all women.USPSTF 2024 — federal recommendation standard
| Factor | Preferred Modality | Key Point |
|---|---|---|
| Obesity (>300–400 lbs) | US first; upright stereo chair if needed | Prone table limit 300–400 lbs; upright chair up to 1,199 lbs with hydraulic lift |
| Limited mobility | US | Can be done supine, seated, or on stretcher; most positioning flexibility |
| Vasovagal history | US or prone stereo | Upright chair increases vasovagal risk; prone keeps patient unaware of procedure |
| Anticoagulation | Any (low-risk per SIR) | No proven benefit to routine hold; select smallest adequate needle gauge |
| Anxiety | US or prone | Real-time feedback reassuring; pre-procedural anxiolytics per institutional policy |
| Lesion Type | Preferred Modality |
|---|---|
| US-visible mass | Ultrasound-guided — fastest, no radiation, real-time |
| Calcifications (US-visible) | US-guided; confirm with specimen radiograph |
| Calcifications (US-occult) | Stereotactic / tomosynthesis-guided |
| MRI-only finding | MRI-guided biopsy |
| One-view mammographic finding | Stereotactic (tomosynthesis preferred) |
| Architectural distortion | Tomosynthesis-guided stereotactic |
| Axillary lymph node | Ultrasound-guided (first line) |
- Indication and alternatives (observation, surgical excision, short-interval follow-up)
- Bleeding/hematoma: ~1–5%; higher risk with larger needle gauges
- Infection: <1%; sterile technique throughout
- Vasovagal reaction: more common with upright positioning or anxiety
- Pneumothorax: rare; prevented by parallel-to-chest-wall technique
- Incomplete sampling / missed lesion: ~1–2%; may require repeat biopsy
- Tissue marker placement: routine after all image-guided biopsies
- Pathology turnaround: typically 2–3 business days
- Post-biopsy follow-up plan and result callback contact
| Agent | SIR Recommendation |
|---|---|
| Aspirin / NSAIDs | Continue — no hold required for standard CNB |
| Clopidogrel / prasugrel | Case-by-case; hold 5–7 days if clinically feasible |
| Warfarin | Continue if INR in therapeutic range |
| DOAC (apixaban, rivaroxaban) | Continue for standard CNB |
| Heparin infusion | Consider timing relative to procedure |
| Item | Details |
|---|---|
| Local anesthetic | 1% lidocaine ± 1:100,000 epinephrine; 9 cm spinal needle for deep lesions (>3 cm) |
| Scalpel | #11 blade for skin nick at needle entry site |
| Antiseptic | Chlorhexidine gluconate or betadine; sterile 4×4 gauze |
| Transducer sleeve | Sterile (US-guided procedures) |
| Biopsy device | 14G SLD for CNB; 9G VABB (standard or petite trough) |
| Tissue marker | Selected by modality and lesion location (see Tissue Markers) |
| Specimen containers | Formalin-filled; label with patient ID and laterality |
| Specimen radiograph | Mandatory for all calcification biopsies |
| Dressing | Steri-strips, gauze, elastic bandage |
| Material | Examples | Notes |
|---|---|---|
| Titanium | Star, coil, O-ring shapes | Standard; visible on mammo, US, MRI |
| Stainless steel / Nitinol | Various shapes | Shape variety differentiates multiple markers in same breast |
| Ceramic | Non-metal | For documented nickel or metal allergy |
| Collagen-embedded (CorMARK) | Hydrates after deployment | Improved sonographic visibility |
| Hydrogel-embedded (HydroMARK) | Self-expands in cavity | Best US visibility in fatty tissue; may migrate up to 2 cm along mesh length |
| Scenario | Preferred Marker |
|---|---|
| Superficial location | Small bare metal — less likely to be palpable |
| US follow-up planned (posterior, axillary, peri-implant) | Spherical/rectangular with embedded material — best US visibility in fatty tissue |
| Stereotactic or MRI-guided biopsy | Bare metal or bioabsorbable mesh (self-expands in biopsy cavity) |
| Near skin surface | Bare metal only — bioabsorbable mesh may protrude or be palpable |
| Post-NAC MRI evaluation planned | Marker with smallest MRI susceptibility artifact (avoid Magseed, RFID) |
- Report: include marker shape, clock position, quadrant, and depth from skin/nipple
- Post-procedure mammogram documents marker in 2 planes (CC and MLO) — same or next day
- If marker migrated: document actual location and note displacement from intended biopsy site
- Displaced markers require careful annotation for accurate presurgical localization planning
- Review prior imaging; confirm target lesion and plan approach
- Obtain informed consent; review anticoagulation status and allergies
- Position patient optimally
- Identify target by US; document depth from skin and distance from chest wall
- Prepare skin with antiseptic; apply sterile transducer sleeve
- Administer local anesthesia: dermal wheal with 25G needle, then inject along tract
- For deep lesions (>3 cm): use 9 cm spinal needle; inject small bolus (~0.1 cc) and confirm tip by hypoechoic fluid collection on US
- Wait 2–3 minutes for full anesthetic effect
- Create skin nick with #11 scalpel blade at planned entry site
- Advance biopsy device under real-time US guidance, parallel to chest wall
- Obtain 4–5 cores (SLD) or targeted vacuum passes (VABB)
- Place tissue marker after final sample
- Apply manual pressure ×10 minutes; sterile dressing
- Obtain bilateral post-procedure mammograms to document marker location
- Provide written post-procedure instructions and pathology callback plan
- Inject small bolus (~0.1 cc); confirm tip position by hypoechoic fluid collection on US
- Lateral decubitus + skin incision at breast periphery creates a flatter approach angle
- For large breasts: insert at steep angle with tip just posterior to lesion, then gradually flatten to parallel
- Tilt US probe to form right angle with biopsy device — improves needle tip visualization
- Account for SLD dead space (0.5–0.8 cm): ensure room deep to lesion for device tip
- Hydrodissection: inject lidocaine or sterile saline between lesion and pectoralis/implant to create safe zone
- Open-trough coaxial technique: allows sampling without firing toward implant or chest wall
- For implant capsule biopsy (rule out BIA-ALCL): hydrodissect fibrous capsule away from implant envelope before sampling
- FNA (25G) may be adequate for peri-implant lesions when space is very limited
- Skin-sparing mastectomy with reconstruction: hydrodissect both superficial and deep to create saline pool
| Gauge | Application |
|---|---|
| 14G | Standard CNB — minimum recommended for adequate histology |
| 16G | Anticoagulated patients, superficial lesions |
| 12G | Larger cores; some specialized devices |
| 9G | Vacuum-assisted biopsy (VABB) — largest cores, multiple passes |
- Advance device (trough closed) to just posterior to the target lesion
- Deploy trough needle (Stage 1): verify on real-time US that trough is within target
- Fine adjustment: apply gentle downward torque on device handle to bring more lesion into trough
- Optional: rotate transducer 90° for orthogonal view — confirms trough position in 3D
- Deploy cutting cannula (Stage 2): shears core without further forward needle movement
- Withdraw device; transfer specimen directly to formalin
- Repeat steps 1–6 for total of 4–5 cores
- Place tissue marker after final sample
- Advance blunt coaxial introducer (with inner stylet) to target lesion
- Remove stylet; hollow cannula provides re-entry channel for each SLD pass
- Insert SLD through cannula for each biopsy pass; reinsert cannula after each sample
- Advantage: no re-targeting between passes; reduces discomfort in dense tissue; ideal for trainees
- Disadvantage: small air introduced with each exchange may obscure small lesions
- Empty or friable cores: confirm target still visible; retarget to a new tract through adjacent tissue
- Needle persistently follows prior tract: switch to 2-stage technique; rotate device 90° between passes
- Continue rotating to sample different quadrants of the lesion ("open the pie")
- Alternative: switch to VABB (vacuum pull replaces trough-fill mechanism)
- Small lesions (<1 cm): complete or near-complete removal desired
- Complex cystic and solid masses: maximizes capture of solid component
- Suspected papillary or intraductal lesions: near-complete removal may obviate surgery
- Subdermal masses (combine with tandem-needle technique)
- Lesions difficult to visualize after local anesthetic injection
- Calcification sampling requiring multiple rotational passes through a single insertion
| Trough | Length | Best Use |
|---|---|---|
| Standard | 2.0 cm | Standard masses, calcifications |
| Petite | 1.2 cm | Superficial lesions, thin breasts, peri-implant, subareolar |
- Position VABB device POSTERIOR to target — lesion will be vacuumed into aperture
- US confirmation: two echogenic lines flanking trough = correct prebiopsy positioning
- Ensure transducer footprint captures both sides of trough
- Activate foot-pedal: vacuum → cut → secure-specimen cycle runs automatically
- Keep switch depressed for continuous sequential passes
- To increase pull: release switch briefly, then re-press — repeated cycling creates cumulative pull
- Rotate device 45–90° between passes for circumferential sampling
- Always direct trough away from skin, nipple, chest wall, and implant
- Place tissue marker through device after adequate sampling
- Remove device; apply manual pressure ×10 minutes
- Place all samples in specimen container on radiograph cassette
- If no calcifications in first 6–8 passes: recheck targeting; acquire new scout images
- Minimum 8–12 rotational samples recommended for calcification VABB
- Document calcification adequacy in radiology report; save specimen radiograph
- Advance blunt coaxial introducer to POSTERIOR margin of lesion — account for 8 mm SLD dead space
- Remove inner obturator; hollow cannula remains as access port
- Insert SLD with sampling trough OPEN through cannula
- Advance SLD to end of coaxial cannula
- Pull coaxial cannula back, exposing open biopsy notch within lesion
- Apply upward torque on device handle to fill notch with target tissue
- Fire outer cutting cannula (Stage 2 only) — no forward needle movement
- Advance coaxial cannula over biopsy needle; withdraw SLD; repeat
- Reduce transducer downward pressure during injection to facilitate fluid spread
- Sterile saline substitutes for lidocaine when additional anesthesia not needed
- Deep lesions near pectoralis: bolus injection creates 5–10 mm safe zone
- Peri-implant lesions: hydrodissect to move lesion surface away from implant
- Cryoablation: sterile saline maintains distance between ice margin and skin
- Position VABB device posterior to target in standard orientation
- Insert hydrodissection needle superficial to mass but deep to dermis
- When VABB vacuum cycle begins: simultaneously inject saline/lidocaine through hydrodissection needle
- Dual effect: (a) physical barrier between dermis and VABB aperture; (b) active fluid layer protecting adjacent structure
- Repeat injection during each vacuum cycle as needed
- Wait 5–10 minutes with gentle massage — anesthetic diffuses into surrounding tissue
- Keep transducer FIXED on surrounding anatomic landmarks (fatty lobule, Cooper ligament intersection)
- Never move transducer off the injection site — losing landmarks means losing your mental map to target
- Resume biopsy when lesion returns to view (typically within 5–10 minutes)
- Confident lesion visualization takes priority over minimizing skin-to-lesion distance
- Available approaches: CC (superior entry), lateral (lateral/medial entry), oblique
- Prone table: breast dependent through table opening; minimizes vasovagal risk
- Upright unit: preferred for obese patients; lateral decubitus option available
- Upright unit: higher success rate than prone in obese patients for posterior lesions
- Position patient on prone table or upright chair in selected projection
- Compress breast; confirm lesion visible on scout images
- Acquire stereotactic pair or tomosynthesis scout
- Click target — X, Y, Z coordinates auto-calculated and sent to biopsy control module
- Confirm coordinates on control module
- Advance stage to calculated coordinates (motorized)
- Note skin-to-target depth on procedure screen — guides anesthesia needle depth
- Administer local anesthesia along needle tract
- Create skin nick with #11 scalpel blade
- Advance biopsy device to calculated depth
- Acquire pre-fire stereo/tomosynthesis images — confirm needle at target
- Fire biopsy device; acquire post-fire images
- Perform 6–12 rotational vacuum passes (VABB), rotating 30–45° between each
- Place tissue marker; acquire post-marker images
- Remove device; apply manual pressure; dress wound; remove compression
- Obtain bilateral CC and MLO post-procedure mammograms to document marker
- Place all cores in specimen container on radiograph cassette
- Perform specimen radiograph after every 2–3 VABB passes
- If no calcifications in specimen: recheck targeting; acquire new scout images
- Document adequacy in report; save specimen radiograph
| Device | Min. Compressed Thickness | Rationale |
|---|---|---|
| Standard VABB (2.0 cm trough) | ~2.8 cm | 2.0 cm trough + 0.8 cm tip dead space |
| Petite VABB (1.2 cm trough) | ~2.0 cm | 1.2 cm trough + 0.8 cm tip |
| Lateral arm approach | ~1.0 cm | Needle travels parallel to compression plate; minimal depth requirement |
- Reduce compression slightly — breast pillows out more centrally, increasing effective thickness
- Inject additional subcutaneous lidocaine — lifts skin away from posterior plate
- Roll or bolster breast with gauze, tape, or foam padding
- Double-paddle technique — apply second compression plate between breast and detector (air-gap)
- Position lesion near center of compression window — maximum tissue pillows out centrally
- Switch to lateral arm approach — requires only ~1.0 cm minimum (most effective solution)
- Pre-pectoral implants reduce available compressible breast thickness
- Implant must be displaced posteriorly by compression — further reduces effective thickness
- Petite device preferred for all implant patients undergoing stereotactic biopsy
- Superficial lesions: manually advance needle until sampling well is just below skin before firing
- Additional subcutaneous lidocaine pushes skin away, increasing skin-to-lesion distance
| Indications | Limitations |
|---|---|
| Thin breasts (min ~1 cm vs. ~2 cm standard) | Central lesions more difficult to access |
| Superficial and peripheral lesions | Larger breasts: 9 cm spinal needle may not reach deep targets |
| Lesions near chest wall | 4th coordinate (Lat X) required on upright units — additional learning curve |
| Lesions obscured by lidocaine conventionally | Not yet widely adopted despite availability since 2007 |
| Reduced radiation dose (fewer required views) | — |
| Feature | Standard Vertical | Lateral Arm |
|---|---|---|
| Needle direction | Z-axis (into compression plane) | X-axis (parallel to compression paddle) |
| Min compressed thickness | ~2.8 cm | ~1.0 cm |
| Key coordinate | Z-depth | Lat X (manual insertion distance) |
| Breast thickness needed | In Z direction | Only marginally greater than needle width |
- Confirm standard vertical approach is NOT amenable
- Remove needle guide holder from upright unit
- Install lateral arm compression paddle (system auto-targets for lateral approach)
- Connect adaptor and needle guide to carriage; slide device mount onto lateral arm and lock
- Install lateral arm stand on image receptor to elevate breast from detector
- Position breast: align paddle EDGE with breast edge at entrance side (reduces coordinate error)
- Acquire tomosynthesis scout
- Click target — X, Y, Z, and Lat X coordinates auto-calculated and transmitted to control module
- Note Lat X (circled red on screen) = manual insertion distance for biopsy device
- Push motor-enable buttons to advance Z rail; adjust Z knob until all differential lines are green
- Note safety margins to superior/inferior skin surfaces (blue circles on screen)
- Attach sterile needle guide; load biopsy device
- Administer local anesthesia with 18G 9 cm SPINAL NEEDLE (standard needles too short for lateral approach); use skin-to-needle-tip distance shown in green oval on screen
- Manually insert biopsy device until X-stop (red arrow indicator) is reached
- Verify needle location number matches X-stop value; lock carriage
- Acquire pre-fire image; confirm needle at target
- Fire device; acquire post-fire image; perform directional sampling
- Place tissue marker; acquire post-marker images; remove device
- Unlock biopsy arm; swing to access breast from side
- Position: CC view = lateral or medial access; ML or LM = superior or inferior access
- Acquire tomosynthesis scout; click target (no separate Lat X coordinate needed)
- Push motor-enable buttons — arm moves to Y and Z coordinates
- Administer local anesthesia; manually advance needle to X coordinate
- Confirm on pre-fire and post-fire images; perform biopsy; place marker
- Review diagnostic MRI: confirm lesion present; determine approach (medial vs. lateral)
- Choose approach = shortest distance from target to skin surface
- Bilateral same-day biopsies: lateral approach for BOTH (medial inaccessible with lateral coil in place)
- Verify MRI-compatible biopsy equipment and tissue marker for planned field strength (1.5T vs. 3T)
- Confirm no MRI contraindications; verify eGFR if gadolinium concern
- Position patient prone; ipsilateral breast in dedicated biopsy coil
- Apply gentle compression paddle: stabilizes breast, reduces motion, allows adequate perfusion for enhancement
- Administer IV gadolinium contrast
- Acquire dynamic post-contrast sequences (3–5 phases)
- Identify target; calculate grid coordinates (row, column, depth)
- Select grid hole corresponding to target coordinates
- Remove patient from bore; mark skin entry 'X' with sterile marker
- Prepare skin; administer local anesthesia
- Advance MRI-compatible obturator through grid hole to calculated depth
- Return patient to bore; acquire post-obturator confirmation images
- Confirm obturator position at or adjacent to target
- Remove inner obturator; insert biopsy device through outer sheath
- Fire device; obtain 6–10 samples (rotate between passes)
- Place MRI-compatible tissue marker
- Acquire post-marker MRI; remove device; apply pressure; dress wound
- Obtain bilateral post-procedure mammograms (documents marker in 2 planes)
- Radiology-pathology concordance review within 24–48 hours
- Reduce compression — excessive compression may reduce perfusion and prevent enhancement
- Obtain additional delayed sequences before cancelling
- ~40–50% of MRI-only lesions are benign; non-visualization may mean lesion resolved
- Skin 'X' mark: if patient moved mid-procedure, misaligned X alerts to retarget
- If still not visualized: cancel procedure; reassess with second-look US or short-interval MRI
- Use petite MRI biopsy device (smaller trough)
- Target off-center in trough — place lesion near leading edge away from skin
- Inject additional lidocaine into superficial tissue to displace skin from biopsy path
- Displace implant posteriorly with compression before grid placement
- Peri-implant lesions: place device adjacent to implant; directionally sample AWAY from implant
- Anterior breast: add foam padding or saline bag between grid and anterior breast for adequate compression
- Confirm adequate enhancement on post-obturator images before sampling
- Posterolateral: prone oblique (elevate contralateral side); ipsilateral arm at side; remove table padding to maximize posterior tissue in coil; freehand angled technique to sample beyond grid edge
- Posteromedial: prone oblique (elevate ipsilateral shoulder; roll breast toward contralateral coil) — allows medial breast access from lateral direction
- If repositioning cannot access lesion: place MRI-compatible marker at obturator site for subsequent surgical localization
| Wire | Features |
|---|---|
| Kopans spring hookwire | External needle removable post-deployment; most widely used |
| Homer J-wire | J-shaped tip; repositionable before deployment |
| Frank hookwire | Original design; 25G spinal needle preloaded |
| Pigtail wire | Pigtail anchor; various gauges and lengths |
- Review imaging; confirm guidance modality (mammo, US, DBT, or MRI) and approach
- Verify patient NPO per OR requirements (same-day surgery)
- Position patient; administer local anesthesia
- Advance introducer needle to target — tip 1–2 cm DEEP to lesion
- Confirm needle position by imaging (spot views / real-time US / post-obturator MRI)
- Advance internal flexible wire until midpoint (thickest part) is AT lesion; tip 1–2 cm beyond
- Confirm wire position with orthogonal imaging
- Remove external introducer needle — flexible wire remains
- Coil externally protruding wire against skin; secure with tape and dressing
- Obtain orthogonal mammographic views documenting wire location vs. lesion
- Communicate to surgeon: depth from skin to lesion, wire type, mammographic documentation
- Patient proceeds directly to OR
| Metric | Data |
|---|---|
| Migration rate | 0–1.8% |
| Positive margin rate | 5.5–57% (higher for DCIS regardless of method) |
| Average surgical time | 6–62 minutes |
| Device cost | ~$20 |
- Same-day surgery required — excludes first OR case; patient must be NPO
- Wire migration: rare but reported — pneumothorax, mediastinal perforation, implant rupture
- Axillary nodes: not preferred — arm movement may damage axillary vessels/brachial plexus
- Higher patient anxiety vs. NWL devices; fasting + anxiety increases vasovagal risk
- Indicated for nonpalpable lesions spanning 2.5–5.0 cm
- No minimum distance between wires — multiple wires do not interfere (unlike NWL devices)
- Pre-plan with surgeon for complex bracketing cases — discuss desired margins and cosmetic outcome
- Obtain orthogonal views of all wires to document spatial relationship to lesion
- I-125-coated material in 4.5 × 0.8 mm titanium seed
- Dose: 3.7–11.1 MBq (0.1–0.3 mCi) per seed
- Half-life: 59.4 days; 27 keV gamma photon peak
- Preloaded into sterile 18G needle with bone wax plug at tip
- Standard needle: 10 cm; seeds effective for 90 days from preloading
- Check seed needle with Geiger-Müller (GM) meter — confirm seed present
- Advance needle to target under image guidance (US, mammo, or MRI)
- Deploy: remove rubber stopper; simultaneously push internal stylet forward WHILE pulling needle hub backward
- Rotate needle hub to ensure seed and bone wax fully expelled from tip
- Withdraw needle; check biopsy site and needle with GM meter — confirm implantation
- Obtain orthogonal imaging to document seed location
- Bracketing: maintain ≥2 cm between seeds for differential intraoperative GM detection
- Explantation standard: within 5 days (NRC license allows up to 14 days)
- Surgeon uses intraoperative gamma probe to confirm activity and guide excision
- Check surgical specimen AND cavity with gamma probe after excision
- Seed returned to radiology for shielded decay and disposal per NRC license
| Metric | Data |
|---|---|
| Migration rate | 0.1–2% |
| Positive margin rate | 5–32% |
| Cost | Comparable to WL after NRC infrastructure amortized |
| Patient preference | Superior to wire — lower anxiety, flexible scheduling |
- Two nitinol antennae attached by resistor — antennae anchor device and prevent migration
- Housed in sterile 16G needle in 3 lengths: 5 cm, 7.5 cm, 10 cm
- Approved depth: ≤6 cm from skin surface (measured supine by US)
- Advance needle tip ~6 mm DISTAL to center of target
- Deploy by withdrawal release: pull needle back while reflector deploys at center of target
- Antennae self-expand to anchor device; confirm by orthogonal imaging
- Bracketing: maintain ≥2 cm between devices
- Handpiece transmits infrared signal; reflector responds with audible + visual feedback
- Detection impaired: depth >6 cm, within hematoma, posterior to dense calcified mass
- Signal interference: direct contact with electrocautery or older halogen OR lights
- Contains nickel — contraindicated in documented nickel allergy
- MRI: minimal artifact at 1.5T and 3T (advantage over Magseed and RFID)
| Metric | Data |
|---|---|
| Migration rate | 4.5–7.0% (highest of all NWL — mostly from post-procedural hematoma) |
| Positive margin rate | 0–16.8% |
| Device cost | ~$450 (plus console and handpiece) |
| MRI artifact | Minimal at 1.5T and 3T |
- Non-radioactive; 5 mm × 1 mm cylindrical; preloaded in sterile 18G stainless steel needle
- Bone wax plug stabilizes seed before deployment
- Manufacturer-specified sensing depth: ~4 cm
- Advance needle tip to center of target
- Deploy: advance steel obturator forward while pulling needle backward
- Confirm placement by orthogonal imaging
- Bracketing: maintain ≥2 cm between seeds for independent detection
- Sentimag probe generates alternating magnetic field; audio frequency + numerical value increase near seed
- Non-ferromagnetic intraoperative instruments required to eliminate magnetic interference
| Metric | Data |
|---|---|
| Migration rate | 0–4.5% |
| Positive margin rate | 9–22% |
| Device cost | ~$400 |
| MRI artifact | ~4 cm (largest of all NWL) |
- Ferrite rod + copper + microprocessor in glass casing with polypropylene antimigratory sheath
- Preloaded in sterile 12G needle; unique RFID identification number per tag
- May require small skin incision — 12G is the largest of all NWL applicators
- Advance needle tip to center of target
- Deploy: advance steel obturator pushing tag from needle tip
- Confirm placement by orthogonal imaging
- Critical: if two tags placed <1.8 cm apart, unique IDs will NOT appear on reader — use wire localization for close-proximity bracketing
- Handheld reader: audible signal + visual distance indicator (up to 60 mm)
- Displays unique tag ID — critical when multiple tags present
- Glass casing may fracture during surgical excision — alert surgical team
- 12G applicator difficult in dense breast tissue
| Metric | Data |
|---|---|
| Migration rate | 0–0.6% (lowest of all NWL devices) |
| Positive margin rate | 0–27% |
| Device cost | ~$550 |
| MRI artifact | 2–5 cm |
| Feature | Wire | RSL (I-125) | SAVI SCOUT | Magseed | RFID |
|---|---|---|---|---|---|
| Gauge | 16–20G | 18G | 16G | 18G | 12G |
| Decouples scheduling | ✗ Same-day only | ✓ ≤14 days | ✓ | ✓ | ✓ Long-term |
| MRI artifact | None | None | Minimal | ~4 cm ⚠ | 2–5 cm ⚠ |
| Migration rate | 0–1.8% | 0.1–2% | 4.5–7.0% | 0–4.5% | 0–0.6% |
| Cost/device | ~$20 | Low after setup | ~$450 | ~$400 | ~$550 |
| Regulatory | None | NRC license required | None | None | None |
| Axillary nodes | Limited | Not standard | FDA approved | Yes | Yes |
| Nickel allergy concern | No | No | ⚠ YES | No | No |
| Electrocautery interference | No | No | ⚠ Older halogen | No | No |
| Patient satisfaction | Lower | Higher | 97% recommend | Higher | Higher |
Technical Parameters
| Parameter | Details |
|---|---|
| Probe | Single cryoprobe (FROST protocol); 17G ProSense (IceCure Medical, liquid nitrogen) |
| Guidance | Real-time ultrasound (primary); CT or MRI feasible for special cases |
| Freeze cycles | Double freeze-thaw-freeze cycle; typically 8–10 min per freeze cycle |
| Ice ball monitoring | Ultrasound — hyperechoic leading edge visualized in real time |
| Target margin | Ice ball must extend ≥1 cm beyond tumor edge on all sides |
| Anesthesia | Local anesthesia ± IV sedation; outpatient procedure |
| Confirmatory biopsy | Post-ablation core biopsy mandatory (FROST: 6-month biopsy); confirms complete ablation |
| Recovery | Return to activity within 24–48 hours; no incision, no drain, no wound care |
Patient Selection Criteria (FROST / ICE3 / FDA Parameters)
- Age: ≥50 (Stratum 1: ≥70; Stratum 2: 50–69 with radiation); ICE3 required ≥60
- Histology: Biopsy-proven invasive ductal carcinoma (IDC) — non-lobular histology
- Receptor status: HR+ (ER/PR+) / HER2-negative
- Grade: Grade 1–2 (well to moderately differentiated)
- Tumor size: Unifocal, US-visible, ≤1.5 cm (FROST median 9mm; ICE3 ≤1.5 cm)
- Nodal status: Node-negative (cN0) on clinical imaging
- DCIS component: No significant intraductal (DCIS) component on core needle biopsy
- Multifocality: Unifocal disease only; no multicentric cancer
- Ultrasound visibility required for real-time guidance
Contraindications
- Multifocal or multicentric disease
- Significant DCIS component
- Lobular histology (diffuse growth pattern, difficult to define margins)
- Tumor not visible on ultrasound
- HR-negative or HER2-positive tumor
- Node-positive disease (cN1+)
- Locally advanced disease (T3/T4)
- Prior radiation to ipsilateral breast
Ideal Candidate Criteria
- Age: ≥50–60 (strongest data for ≥70 in FROST Stratum 1)
- Histology: Unifocal invasive ductal carcinoma (IDC) — non-lobular
- Receptor: HR+ (ER/PR+) / HER2-negative, grade 1–2
- Size: ≤1.5 cm, US-visible (median tumor size in FROST: 9 mm)
- Nodes: Node-negative (cN0) by clinical imaging
- DCIS: No significant intraductal component on core biopsy
- Genetics: No known BRCA1/2 pathogenic variant
- Prior Tx: No prior ipsilateral breast radiation
Summary Evidence Table
| Parameter | Evidence | Current Status |
|---|---|---|
| Complete ablation (FROST) | 82/83 patients (99%); Phase II | Established feasibility |
| Complete ablation (ACOSOG Z1072) | 75.9% overall; 92% within ablation zone | Size-dependent; excludes >25% DCIS |
| 5-yr IBTR — FROST overall | 3.64% (Holmes et al. 2026) | Phase II; no randomized comparison |
| 5-yr IBTR — FROST Stratum 1 (≥70, ET only) | 2.08% | Best outcomes subgroup |
| 5-yr IBTR — FROST Stratum 2 (50–69, ET+RT) | 5.80% | Higher; RT required in this group |
| 5-yr IBTR — ICE3 (Fine 2024) | 4.3%; n=194, age ≥60 | Phase II; completed |
| Trial-ineligible patients (real-world) | 18.2% IBTR at 3 years (Oueidat 2024) | Critical — strict selection required |
| FDA approval for breast cancer Tx | IceCure ProSense — FDA approved 2025 | First-ever approval for cancer (not just FA) |
Head-to-Head: Cryoablation vs Lumpectomy + RT
| Factor | Cryoablation | Lumpectomy + RT |
|---|---|---|
| Procedure time | 30–60 min (outpatient) | 60–90 min (OR) + weeks of RT |
| Recovery | 24–48 hours; no wound, no drain | 2–4 weeks post-op + 3–6 weeks RT |
| 5-yr local recurrence | 3.64% overall / 2.08% (age ≥70, FROST) | ~1–2% at 5 years (standard of care) |
| Margin confirmation | Not possible (ablation zone, no specimen) | Pathologic margins confirmed in specimen |
| Cosmesis | Excellent (no incision); 95–98% satisfaction (ICE3) | Good to excellent (breast-conserving) |
| Cost (Khan 2023) | $2,221.70 mean procedure cost | $16,896.50 mean procedure cost |
| FDA status (breast cancer) | FDA-approved 2025 (IceCure ProSense) | Standard of care |
Post-Ablation Follow-Up Protocol
- FROST protocol: Mandatory core biopsy at 6 months + regular clinical exams and imaging
- Khan et al.: Mammogram + US at 6 months; MRI at baseline then annually
- CEM: Emerging as a practical MRI alternative for post-ablation surveillance
Expected Post-Ablation Imaging Findings (Benign)
Breast Cancer — Definitive Cryoablation
Palliative & Metastatic Breast Cancer
Patient Selection Criteria
- Biopsy-proven fibroadenoma (core needle biopsy) — strict rad-path concordance required
- Lesion size <3.0 cm (ASBrS/SBI 2025 cutoff)
- Ultrasound-visible lesion (required for real-time guidance)
- Symptomatic (pain, cosmetic concern, anxiety) or patient preference to avoid open excision
- Exclude: phyllodes tumor, atypia, or malignancy on CNB
Trial Data
Mechanism of Immunogenic Cell Death
- Cell death via intracellular ice crystal formation + osmotic injury — necrotic, immunogenic (contrast with apoptosis, which is immunologically silent)
- Tumor cell necrosis releases DAMPs, tumor antigens, and pro-inflammatory cytokines (IL-1β, HMGB1, ATP)
- Activates innate immune cells (NK cells, dendritic cells) and initiates antigen cross-presentation to T cells
- May prime systemic anti-tumor immune response capable of targeting distant metastases (abscopal effect)
Phase II Clinical Trial — NCT03546686
Treatment: Perioperative cryoablation → Ipilimumab 1 mg/kg IV + Nivolumab 240 mg IV → standard surgical excision → adjuvant nivolumab or pembrolizumab ± capecitabine/olaparib.
Primary endpoint: 3-year event-free survival (EFS).
FES PET/CT — Overview & Indications
¹⁸F-Fluoroestradiol (Cerianna) · FDA-approved 2020 · Functional ER imaging adjunct to biopsy
SNMMI Appropriate Use Criteria (2023)
All four current Appropriate indications target patients with known recurrent or metastatic ER+ breast cancer. Primary staging, routine T/N staging, and response assessment are Rarely Appropriate.
| Appropriateness | Clinical Scenario | Score (1–9) |
|---|---|---|
| Appropriate | ER status assessment when biopsy is unfavorable or nondiagnostic | 8 |
| ER detection when prior imaging findings are equivocal or suspicious | 8 | |
| Initial diagnosis of metastatic disease — for consideration of endocrine therapy | 8 | |
| Progression of metastatic disease — for consideration of 2nd-line endocrine therapy | 8 | |
| May Be Appropriate | Unknown primary when biopsy not feasible | 5 |
| Routine staging of extra-axillary nodes and distant metastases | 5 | |
| Staging invasive lobular carcinoma (ILC) or low-grade IDC | 5 | |
| ER status in lieu of biopsy for accessible lesions | 5 | |
| Lesion detection in suspected/known recurrent or metastatic disease | 5 | |
| Rarely Appropriate | Diagnosing primary breast cancer | 2 |
| Routine T-staging of primary tumor | 1 | |
| Routine staging of axillary nodes | 3 | |
| Initial diagnosis of primary cancer — for endocrine therapy | 1 | |
| Measuring response to therapy | 1 |
Drug Interactions — Must Withhold Before Imaging
ER-blocking drugs competitively inhibit FES uptake and cause false-negative results
| Drug | Class | FDA-Required Washout | Clinical Notes |
|---|---|---|---|
| Elacestrant | Oral SERD | ≥ 11 days | 5 × t½ (~2.2 days) |
| Tamoxifen | SERM | ≥ 8 weeks | Active metabolites (endoxifen, 4-OH-tamoxifen) have prolonged half-lives; full washout extended well beyond parent drug clearance |
| Fulvestrant | IM SERD | ≥ 28 weeks | t½ ~40 days; also degrades ER (not just blocks it) — FES imaging during fulvestrant therapy is essentially infeasible in routine clinical practice. Oncologists are often reluctant to withhold therapy this long |
| Aromatase inhibitors letrozole, anastrozole, exemestane |
AI | No washout required | Work peripherally (suppress estrogen synthesis); do not bind ER directly. FES imaging proceeds normally during AI therapy |
| CDK4/6 inhibitors palbociclib, ribociclib, abemaciclib |
CDKi | No washout required | Do not interact with ER. FES imaging proceeds normally |
Imaging Protocol & Normal Biodistribution
Patient preparation, administration, and expected physiologic distribution
Protocol
| Parameter | Details |
|---|---|
| Dose | 222 MBq (6 mCi); acceptable range 111–222 MBq (3–6 mCi) |
| Administration | IV push over 1–2 min via syringe pump + ≥10 mL normal saline flush |
| Uptake time | 60–80 min post-injection (feasible as early as 20 min to limit bowel tracer accumulation) |
| Coverage | Vertex/skull base to mid-thigh; arms above head, supine |
| Patient prep | Hydrate well; void immediately before scanning to reduce bladder activity adjacent to pelvic structures; no fasting or dietary restrictions required; no pregnancy test unless childbearing potential |
| SUV windowing | Default: 0–5. Increase to 0–20 for areas adjacent to liver/bowel. Narrow to 0–2.5 for precise assessment of hepatic/abdominal lesions |
| Radiation dose | ~4.9 mSv effective dose from 222 MBq; critical organs: liver, gallbladder, uterus |
Normal Biodistribution
- Liver — highest background; the radiotracer's critical organ
- Biliary system / common bile duct
- Small bowel — reabsorbed radiolabeled metabolites via enterohepatic circulation
- Kidneys / ureters / bladder — excretion
- Uterus — high ER expression; serves as internal control confirming adequate FES tissue binding
- Injection site vein — residual tracer in draining vein is expected
- Reactive / inflammatory lymph nodes
- Stimulated marrow / marrow repopulation after G-CSF
- Degenerative osseous disease
- Sites of active infection or inflammation
- Brain parenchyma (no physiologic uptake → brain metastases are conspicuous on FES)
- Brown fat / symmetric mediastinal/hilar uptake
Image Interpretation
FES positivity thresholds, windowing strategy, and diagnostic performance
| Finding | Threshold / Definition | Implication |
|---|---|---|
| FES-positive lesion | SUVmax ≥ 1.5 or qualitatively above local background and blood pool | Functionally ER-positive disease. Predicts likelihood of response to endocrine-targeted therapy |
| FES-negative lesion | SUVmax < 1.5 / at or below local and blood pool background | ER-negative, functionally ER-negative, or endocrine-refractory. In landmark data, 0/15 patients with all-site SUV <1.5 responded to endocrine therapy |
| Hepatic & bowel lesions | High physiologic liver background may mask ER+ disease; lesional SUV may be below hepatic parenchyma even at SUV >1.5 | Greatest limitation of FES PET. Correlate with MRI or consider biopsy. Window images down to 0–2.5 and consider delayed imaging or early acquisition before bowel excretion |
Factors Affecting FES Uptake
| Factor | Effect | Note |
|---|---|---|
| SHBG (sex hormone-binding globulin) | Inversely associated — higher SHBG → lower FES SUV | ~45% of circulating FES is SHBG-bound; the only independent predictor of lean body mass–adjusted FES uptake in multivariate analysis |
| Endogenous estradiol (premenopausal) | No significant effect on diagnostic accuracy | Physiologic estrogen levels do not interfere with FES uptake. FES can be used in pre- and postmenopausal women and in men |
| ER-blocking drugs | Markedly reduces or eliminates FES uptake → false-negative | See Drug Interactions section — the single most important pre-scan check |
| Tumor size | Small lesions (<1 cm) may fall below PET resolution threshold | Partial volume effect limits sensitivity for sub-centimeter disease; uptake may not exceed background |
False Positives & False Negatives
Pitfalls in FES PET/CT interpretation
| Category | Entity | Mechanism / Practical Note |
|---|---|---|
| False Positive | Uterine fibroids / endometrium | ER-expressing benign tissue; uterus is expected to be FES-avid — do not call uterine uptake pathologic in patients with a uterus |
| Other ER+ malignancies (uterine, ovarian, endometrial) | May produce unexpected FES-avid lesions outside of breast cancer distribution; clinical correlation required in patients with dual malignancies | |
| Meningiomas | May show FES uptake; absence of physiologic brain uptake makes these conspicuous — do not mistake for intracranial metastasis. Brain MRI recommended for clarification | |
| Post-radiation pulmonary/nodal changes | FES uptake in irradiated lung seen in >50% of cases in one series. Draining lymph nodes of irradiated fields may also be FES-avid. Likely reflects vascular permeability/extravasation rather than ER binding — correlate with timing of radiation and FDG PET | |
| False Negative | Liver metastases | High hepatic physiologic background; even large lesions may have a SUV below hepatic parenchyma. The greatest limitation of FES PET — do not exclude liver metastases based on negative FES scan alone. Correlate with MRI or CECT |
| Peritoneal / bowel metastases | High bowel background from biliary/enteric excretion of radiolabeled metabolites limits detection of peritoneal disease close to bowel | |
| Small lesions (<1 cm) | Below PET resolution threshold; partial volume averaging reduces apparent SUV. May still show uptake qualitatively above background in some cases | |
| Truly ER-negative / ER-converted disease | Triple-negative breast cancer, and ER+ primaries that have lost ER expression at metastatic sites, will not show FES uptake — this is the expected and meaningful finding, not a scan failure. ER conversion occurs in 8–33% of patients over disease course |
Clinical Applications
ILC staging, ER heterogeneity, FDG complementarity
ILC is nearly always ER+ (>90%) but is poorly detected by FDG PET due to low cellularity and glucose metabolism. FES PET/CT demonstrates particular promise:
- Changed clinical stage in 18% of ILC patients in a prospective study
- Detected metastases not seen on standard imaging in 24% of patients
- Head-to-head: FES detected 254 vs. 111 total lesions compared to FDG PET in one ILC series (71% of patients)
- ILC metastasizes to unusual locations (peritoneum, GI tract, bone marrow) where FDG sensitivity is lower; FES can identify these ER+ sites
- SNMMI rates ILC staging as "May Be Appropriate" (score 5)
15–45% of metastatic breast cancer patients have FES-heterogeneous disease (a mix of FES+ and FES− sites at a single time point). Temporal ER conversion (ER loss at a previously positive site) occurs in 8–33% over the disease course.
- Patients with 100% FES-avid disease had longest PFS on endocrine therapy combined with CDK4/6 inhibition (73 wk vs. 20 wk for FES-heterogeneous and 15 wk for 100% FES-negative)
- FES-heterogeneous patients treated with endocrine therapy had shorter PFS than those treated with chemotherapy (median 4.6 vs. 7.1 months in one retrospective study)
- FES negativity in an IHC-positive tumor: zero of 15 patients with all-site SUV <1.5 responded to endocrine therapy in a landmark study — the strongest negative predictive signal in the literature
FDG and FES image different aspects of disease biology and are best viewed as complementary. Across four studies, FES PET led to treatment change in 50% of patients with equivocal imaging findings.
- FDG+, FES+: ER+ metabolically active metastasis — supports endocrine therapy + CDK inhibition
- FDG+, FES−: Metabolically active but ER-negative or functionally ER-negative — endocrine therapy less likely to succeed; biopsy recommended; may support chemotherapy-based regimen
- FDG−, FES+: Indolent ER+ disease (e.g., sclerotic bone mets) — supports endocrine therapy; FDG alone would have underdetected these
- Both negative: Broad differential — benign finding vs. non-ER, non-FDG-avid disease; biopsy if clinically indicated
Dual-tracer imaging on separate days is the current standard (¹⁸F t½ = 110 min — next-day imaging is sufficient). Research protocols using long axial field-of-view (LAFOV) PET scanners to acquire both tracers in a single session are in development.
Safety, Adverse Reactions & Precautions
Excellent safety profile — key precautions are clinical, not toxicologic
| Category | Details |
|---|---|
| Adverse reactions | Extremely rare: in 1,207 patients studied, only injection-site pain and dysgeusia (altered taste) were reported. In a prospective safety study (n=90), procedural pain occurred in 10% of patients — attributable to mechanical needle insertion, not the radiopharmaceutical. Only 1 patient (1%) had a drug-related adverse event (injection site pain). No serious adverse events recorded in any study. |
| Contraindications | None listed in FDA prescribing information |
| Not a biopsy substitute | FES PET should not replace biopsy when biopsy is indicated. A negative FES scan should not override clinical or pathologic evidence supporting endocrine therapy. Biopsy confirmation is still recommended when FES PET results will change management. |
| Radiation safety | ~4.9 mSv effective dose from 222 MBq (6 mCi). Standard radiation safety precautions apply. Critical organs: liver, gallbladder, uterus. |
| Pregnancy | No human or animal studies available. Confirm negative pregnancy test before administration in women of childbearing potential. All radiopharmaceuticals carry potential for fetal harm. |
| Lactation | Interrupt breastfeeding for at least 4 hours after administration (per Cerianna prescribing information). NRC guidance: pumped milk from mothers given most radiopharmaceuticals can be stored safely for 10 physical half-lives of the radionuclide (¹⁸F t½ = 110 min → ~18 hours for 99.999% radioactive decay). Some experts recommend 12-hour avoidance of close infant contact after injection, though this is not specifically stated in the Cerianna label. |